J Colombel J et al. Gastroenterology, 2026. Open Access! Darvadstrocel in Patients With Crohn’s Disease With Complex Perianal Fistulas: The ADMIRE CD II Phase 3 Randomized Trial
Background: The ADMIRE CD II, a phase 3 trial of the efficacy and safety of darvadstrocel in patients with complex perianal fistulas (CPF) at weeks 24 and 52, conducted in more than twice as many sites and patients (n=568) as ADMIRE CD, from North America as well as Europe and Israel. This trial was conducted after the approval of darvadstrocel in Europe and Japan and therefore aimed to provide further confirmation of efficacy in patients with CPF.
Key findings:
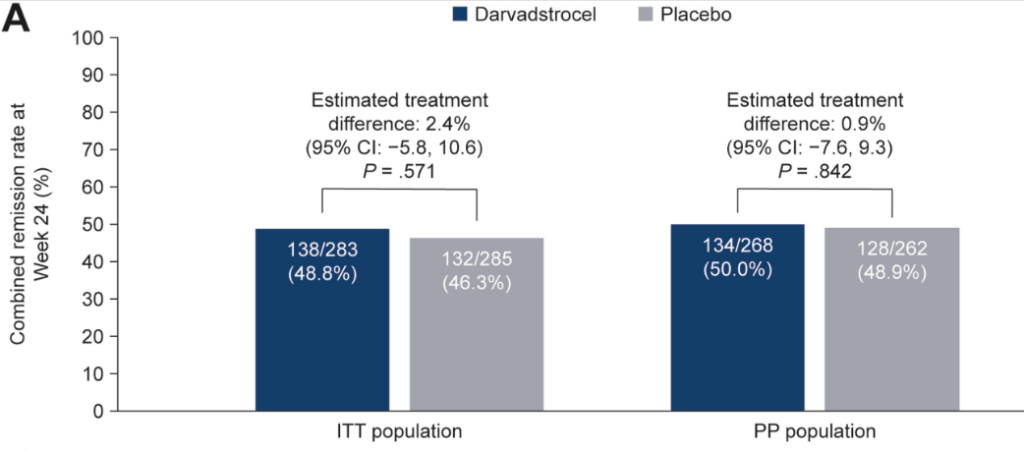
- At week 24, combined remission was achieved in 138 of 283 (48.8%) patients in the darvadstrocel group and 132 of 285 (46.3%) in the placebo group
- There were no significant differences in key secondary endpoints for darvadstrocel vs placebo (clinical remission at week 24 [P = .515] and time to clinical remission [P = .374])
- Treatment-emergent adverse events were infrequent and experienced by similar proportions of patients receiving darvadstrocel (203/278 [73.0%]) and placebo (201/274 [73.4%])
In the discussion, the authors speculate on why ADMIRE CD found a significant response to darvadstrocel whereas the current larger ADMIRE CD II did not.
- ” In ADMIRE CD II, all patients had seton placement (mandatory at least 2–3 weeks before treatment administration), whereas in ADMIRE CD, seton placement was as clinically indicated, and 10 patients (2 darvadstrocel, 8 placebo) did not have a seton. The fact that all patients, including those in the control group, underwent fistula preparation before treatment administration may have enhanced response rates in the placebo group in ADMIRE CD II”
- “In patients with CPF, it is well established that higher trough levels of concomitant systemic therapies are associated with improved fistula healing outcomes compared with lower trough levels. These data were not collected during ADMIRE CD II or ADMIRE CD, but it is acknowledged that the possibility of higher trough levels in ADMIRE CD II compared with ADMIRE CD could have contributed to the difference in placebo response rates between trials… also relevant to consider the higher proportion of patients using background immunosuppressants or a combination of immunosuppressants and monoclonal antibodies in ADMIRE CD II compared with ADMIRE CD.”
My take (borrowed in part from the authors): These results “challenge the relevance of stem cell therapy for perianal fistula healing.” It appears that good surgical management along with optimized medical therapy achieve the same results.
Related blog posts:
