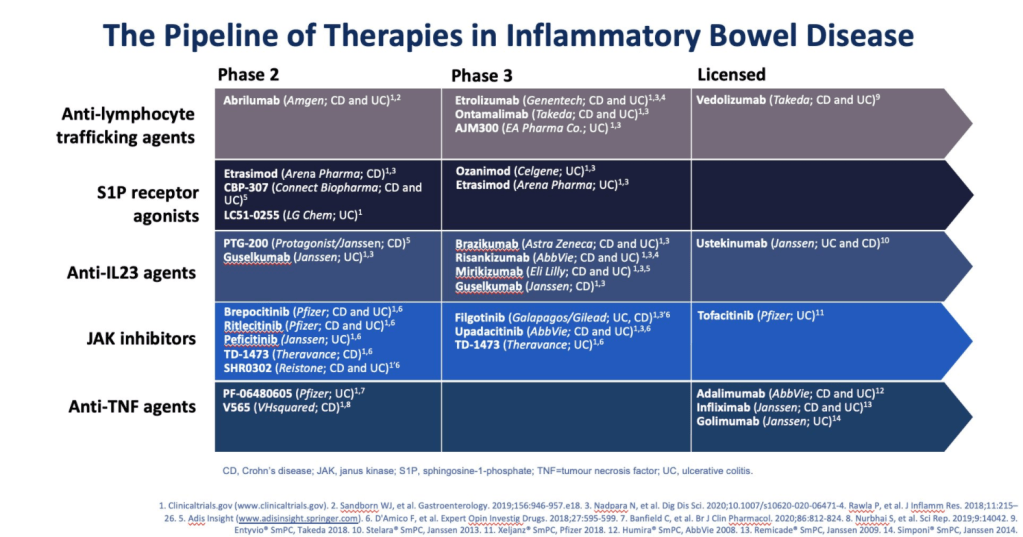
From Gastroenterology and Endoscopy News: New Anti–IL-23 Therapy Shows Benefit in Crohn’s Disease
An excerpt:
Two phase 3 placebo-controlled trials with the immune modulator risankizumab demonstrated control of Crohn’s disease whether or not patients had previously received a biologic agent.
Rates of clinical remission at 12 weeks with the interleukin (IL)-23 inhibitor risankizumab (Skyrizi, AbbVie), were about 48% in patients without prior exposure to biologic therapy and more than 40% in those with prior exposure…
The two trials, ADVANCE and MOTIVATE were presented together at the 2021 Digestive Disease Week (abstract 775a)…
Only 12% of patients in the placebo group achieved endoscopic remission versus 40.3% of those on the 600-mg dose of risankizumab (P<0.001). [Rates of endoscopic remission were higher in the biologic-naive (50.5%)]
My take: In addition to ustekinumab (already approved), a number of other therapeutic agents that target IL-23 are likely to be available soon to help manage Crohn’s disease. This includes risankizumab but others with phase 3 studies include brazikumab, mirikizumab, and guselkumab..