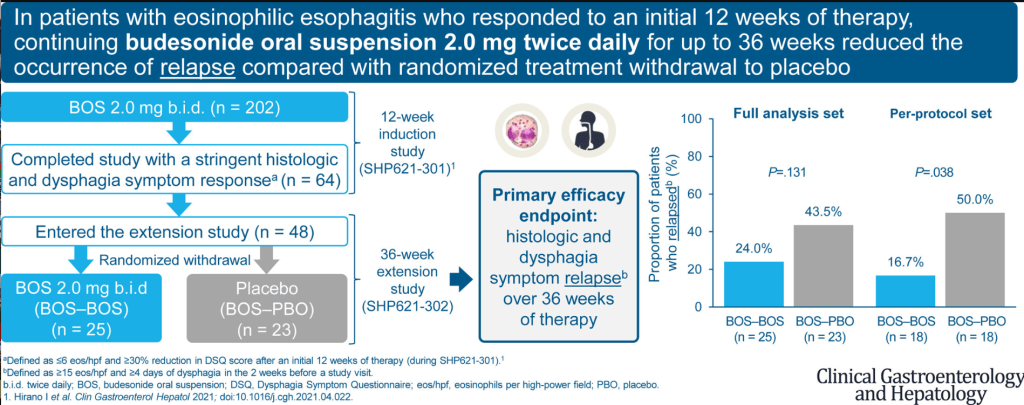
ES Dellon et al. Clin Gastroenterol Hepatol 2022; 1488-1498. Open access: Long-Term Treatment of Eosinophilic Esophagitis With Budesonide Oral Suspension
Methods: 48 patients who had fully responded to a 12-week induction course of budesonide 2 mg BID oral suspension were randomized to continuation of therapy or to placebo, for 36 weeks.
Key findings:
- Patients randomized to placebo experienced relapse at a numerically higher rate than those who continued budesonide (43.5% vs 24.0%; p=.13). This reached statistical significance in a per-protocol analysis
- In a separate arm, 13% of the 106 patients with previous partial or no response did subsequently fully respond to budesonide
- Budesonide therapy was well-tolerated; candidiasis-related events occurred in 17 patients overall and were mild to moderate, and abnormal adrenocorticotropic hormone stimulation tests were reported in 5%
My take: Most patients who respond to induction with budesonide will continue to respond to ongoing treatment. A high rate of relapse is seen in those randomized to placebo.
Related blog posts:
- I-SEE for Eosinophilic Esophagitis
- New 2020 Eosinophilic Esophagitis Guidelines | gutsandgrowth
- Dupilumab: FDA Approval for Eosinophilic Esophagitis
- What Happens When Topical Steroids Are Stopped in Eosinophilic Esophagitis | gutsandgrowth
- Orodispensable Budesonide Tablets for Eosinophilic Esophagitis | gutsandgrowth
- Head-to-Head: Budesonide vs Fluticasone for Eosinophilic Esophagitis | gutsandgrowth
- Phase 3 Trial of Budesonide for Eosinophilic Esophagitis & COVID-19 Deaths in U.S. | gutsandgrowth
- How Effective Are PPIs for Eosinophilic Esophagitis? | gutsandgrowth
- Eosinophilic Esophagitis -Up to Date Dietary Management Review | gutsandgrowth