L Peyrin-Biroulet et al. NEJM 2024; 391:213-223. Risankizumab versus Ustekinumab for Moderate-to-Severe Crohn’s Disease
Background: “Interleukin-23 is a heterodimeric proinflammatory cytokine comprising a p40 subunit shared with interleukin-12 and a unique p19 subunit that plays a key role in skin, joint, and gastrointestinal inflammation.16 Ustekinumab and risankizumab are humanized IgG1 monoclonal antibodies; ustekinumab selectively binds p40, and risankizumab selectively binds p19…In head-to-head trials directly comparing their efficacy in psoriasis, risankizumab was superior to ustekinumab, which suggests greater efficacy with p19 blockade than with p40 blockade.”
This “SEQUENCE” trial was a phase 3b, multicenter, open-label, randomized controlled trial with 527 patients with moderate-to-severe Crohn’s disease who either had an inadequate response or had intolerance to anti-TNF agents, received either risankizumab or ustekinumab.
Key Findings:

- A higher percentage of patients in the risankizumab group than in the ustekinumab group completed all the assigned treatment (90.2% [230 of 255 patients] vs. 72.8% [193 of 265 patients]). The primary reason for discontinuation of risankizumab was an adverse event (3.1% [8 of 255 patients]), and the primary reason for discontinuation of ustekinumab was lack of efficacy (13.2% [35 of 265 patients]
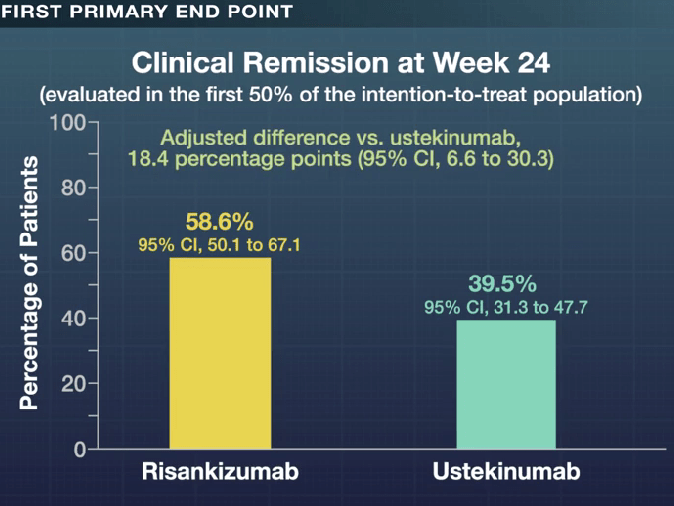
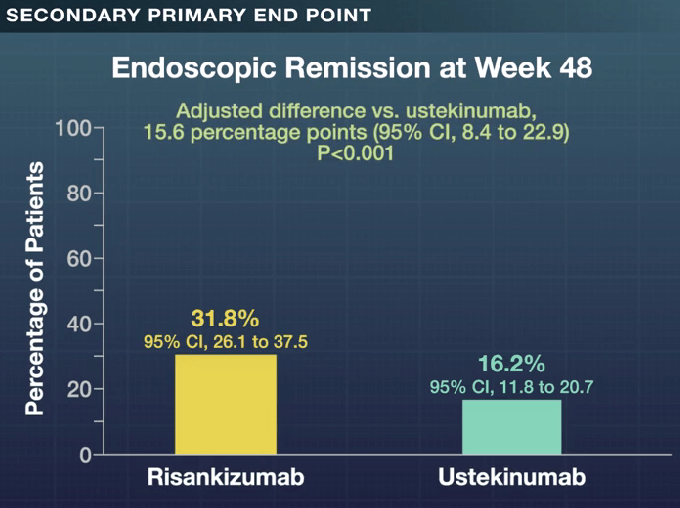
- Clinical remission at 48 weeks was 60.8% with risankizumab and 40.8%% with ustekinumab (P<0.001); there were similar rates of glucocorticoid-free clinical remission, 60.8% vs 40.4% respectively. Endoscopic response at 48 weeks was 45.1% and 21.9% respectively.
My take: These head-to-head results showed the superiority of risankizumab over ustekinumab across numerous clinical and endoscopic end points, including glucocorticoid-free clinical remission and endoscopic remission. However, it is still concerning to me that endoscopic remission rates were only 32% at 1 year and that less than half had an endoscopic response.
Related blog posts:

Pingback: Risankizumab Outperforms Ustekinumab | gutsandgrowth - reviewer4you.com
Oof. So, when/if TNF blockers do fail, there’s only a 30% to achieve clinical remission with either Risankizumab or Ustekinumab. A bit bleak. Is there any study on the interraction effect since those two are binding to different receptors.
You are not interpreting study correctly. With Risankizumab, the clinical remission rate was 61% at 1 yr. The endoscopic remission rate is lower (close to 30%) but this is a much higher standard than clinical remission. Even a clinical remission (not having symptoms, improved labs) is quite good. True healing of the mucosa is much better. There are lots of studies that have looked at how these medications work.