JF Colombel. et al. Clin Gastroenterol Hepatol 2024; 22: 1668-1677. Open Access! Upadacitinib Reduces Crohn’s Disease Symptoms Within the First Week of Induction Therapy
This study was a post hoc analysis included pooled data from 2 phase 3, multicenter, double-blind, 12-week induction trials (U-EXCEL and U-EXCEED) and 1 maintenance trial (U-ENDURE). The study included 1021 patients with Crohn’s disease (CD) (n = 674 UPA45; n = 347 PBO).
Key findings:
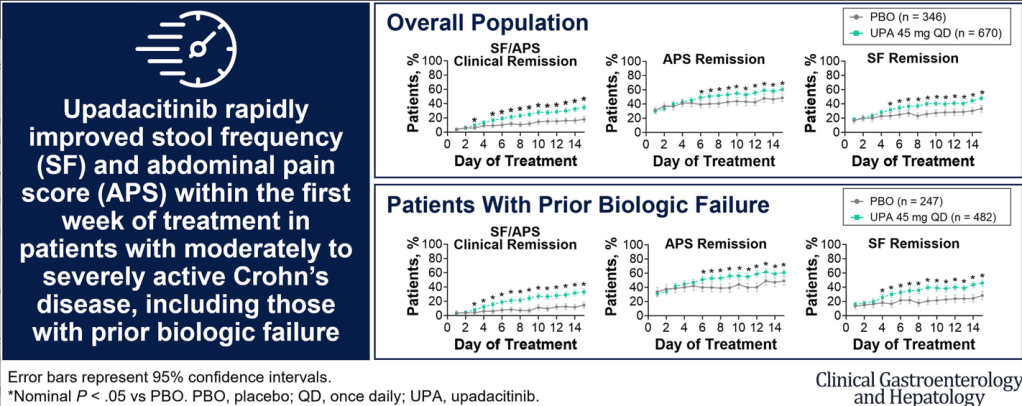
- Upadacitinib 45 mg taken once daily resulted in rapid relief from CD symptoms within 5 to 6 days of treatment initiation and improved clinical outcomes starting at week 2.
- The present analysis demonstrates symptomatic relief as early as day 5 to 6 for patients receiving UPA, with 16.7% of patients experiencing daily SF/APS clinical remission by day 5.
- The first achievement of daily stool frequency/abdominal pain score (SF/APS) clinical remission occurred earlier with UPA45 (median, 13 d) vs PBO (median, 32 d)
- Patients treated with UPA45 showed improved rates of SF/APS clinical remission (21.1% UPA45 vs 8.9% PBO) and clinical response (58.8% UPA45 vs 37.9% PBO) starting at week 2 (both P ≤ .01).
In their discussion, the authors note that time to response to treatment with upadacitinib compares favorably to other advanced therapies:
“Vedolizumab resulted in symptomatic improvement within 2 to 4 weeks of treatment initiation16; ustekinumab led to clinical response and remission at week 3 or 6, depending on the dose.17 Similarly, of the time points analyzed, clinical response and/or clinical remission was observed as early as week 2 for risankizumab, 5 infliximab,18 and certolizumab pegol,19 and as early as week 1 for adalimumab.20,21“
My take: The rapid response seen in many patients indicate that upadacitinib can be a steroid-sparing therapy in patients with Crohn’s disease.
Unrelated article: E Louis et al. JAMA 2024; doi:10.1001/jama.2024.12414. Risankizumab for Ulcerative Colitis: Two Randomized Clinical Trials
Key findings: Among the 975 patients with moderate to severe ulcerative colitis, analyzed in the induction trial, 1200 mg of risankizumab significantly increased the rates of clinical remission at 12-week follow-up compared with placebo (20.3% vs 6.2%, respectively). Among 548 patients included in the primary efficacy analysis for the maintenance trial, 180 mg of risankizumab and 360 mg of risankizumab significantly increased the rates of clinical remission (40.2% and 37.6%, respectively) compared with placebo (25.1%).
Related blog posts:
