G D’Arcangelo et al. J Pediatr Gastroenterol Nutr. 2026;82:495–502. Open Access! Vedolizumab for extraintestinal manifestations in pediatric inflammatory bowel disease: Results from the VedoKids study
Background: “Since vedolizumab is a gut-selective anti-α4β7 integrin, its effect on EIMs has been a matter of debate, with relevant data lacking in pediatric IBD. A systematic review, which included three interventional studies, five non-interventional studies, and three case series, concluded that there is insufficient evidence supporting the efficacy of vedolizumab for treating pre-existing EIMs in adults.3“
Methods: This was a subgroup analysis of the pediatric VedoKids cohort, a multicenter, prospective “real-life” study of children (aged 0–18 years) with IBD treated with vedolizumab and followed through 54 weeks.
Key findings:
- EIMs were identified in 18/142 (12.6%) children at baseline
- Children with EIMs had an average age of diagnosis of 9 yrs compared to 12 yrs in those without EIMs
- Children with EIMs had higher rate of pancolitis in UC and ileocolonic distribution in CD
- Prior anti-TNF medication was noted in 16 (89%) of EIM cohort compared to 74 (60%) of non-EIM cohort
- Concomitant medications were administered in 72% of EIM cases and to a similar number of non-EIM patients. For EIM patients, ASA were given in 7, steroids in 10, thiopurines in 4 and methotrexate in 2
- Children with EIMs had more active disease (see below)

- EIM resolution rate of 89%, mainly occurring within the early weeks of vedolizumab treatment
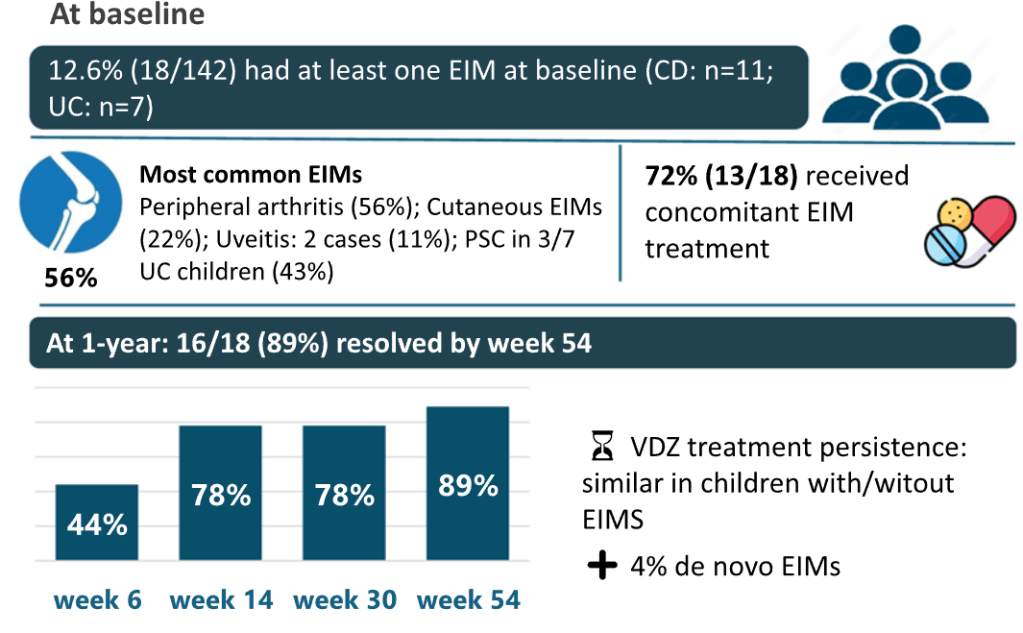
My take: While this study has several limitations, including the high rate of concomitant medications, it shows that most patients receiving vedolizumab had resolution of their EIMs. In addition, it shows that patients with EIMs had a more severe IBD phenotype.
Related blog posts:

