B Kerur et al. JPGN 2022; 75: 64-69. Utilization of Antitumor Necrosis Factor Biologics in Very Early Onset Inflammatory Bowel Disease: A Multicenter Retrospective Cohort Study From North America
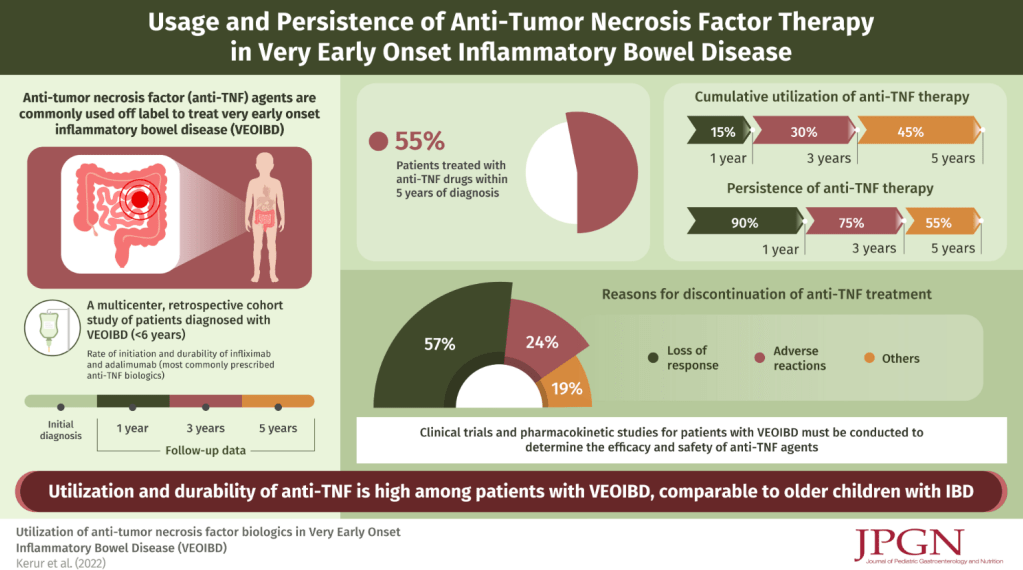
In this retrospective study, 120 of 294 children with VEO-IBD (diagnosed 2008 and 2013, PRO-KIDS network) received anti-TNF therapy (96% infliximab). 101 of these 120 had adequate data recorded. It is noted that additional data on this cohort has been previously published (IBD Updates: Outcomes of VEO-IBD, PIANO Study Update, and Insurance-Disparity Relationship). Key findings:
- Anti-TNF durability was 90% at 1 year, 75% at 3 years, and 55% at 5 years
- Patients with Crohn’s disease had better durability than those with UC/IBD-U (Hazard ratio 0.17)
- The most common reason for discontinuation of anti-TNF were loss of response in 24 (57%) children
- 67 (66%) received combined therapy with an immunomodulator and this was associated with improved anti-TNF durability (Hazard ratio 0.30). However, authors note this was in era preceding widespread therapeutic drug monitoring.
- The majority of children in the current study did not undergo testing for monogenic mutations
My take: Data for use of anti-TNF agents in this age group (< 6 yrs) has been limited. This study suggests similar effectiveness of anti-TNF agents in VEO-IBD compared to older groups. Given this groups increased risk for monogenic mutations, it is still a good idea, if feasible, to test for these disorders.
Related blog posts:
- Very Useful VEO-IBD Position Paper
- Patterns and Puzzles with VEO-IBD This is a very good review and the image on this day has one of my favorite patient t-shirts
- Targeted Therapy for Autoinflammatory Very Early Onset Inflammatory Bowel Disease