ES Dellon et al. NEJM 2022; 387; 2317-2330. Dupilumab in Adults and Adolescents with Eosinophilic Esophagitis
This study was pivotal for receiving FDA approval of dupilumab (dupixent) for the treatment of EoE (the only FDA approved therapy). Background: “Dupilumab, a fully human monoclonal antibody, blocks interleukin-4 and interleukin-13 signaling, which have key roles in eosinophilic esophagitis..Standard-of-care treatments for eosinophilic esophagitis include food elimination diets, proton-pump inhibitors (PPIs), swallowed topical glucocorticoids (applied to the esophagus by swallowing), and, in the case of strictures, esophageal dilation.11,12 However, the rates of response are variable (30 to 40% of patients may not have a response to first-line treatments).”
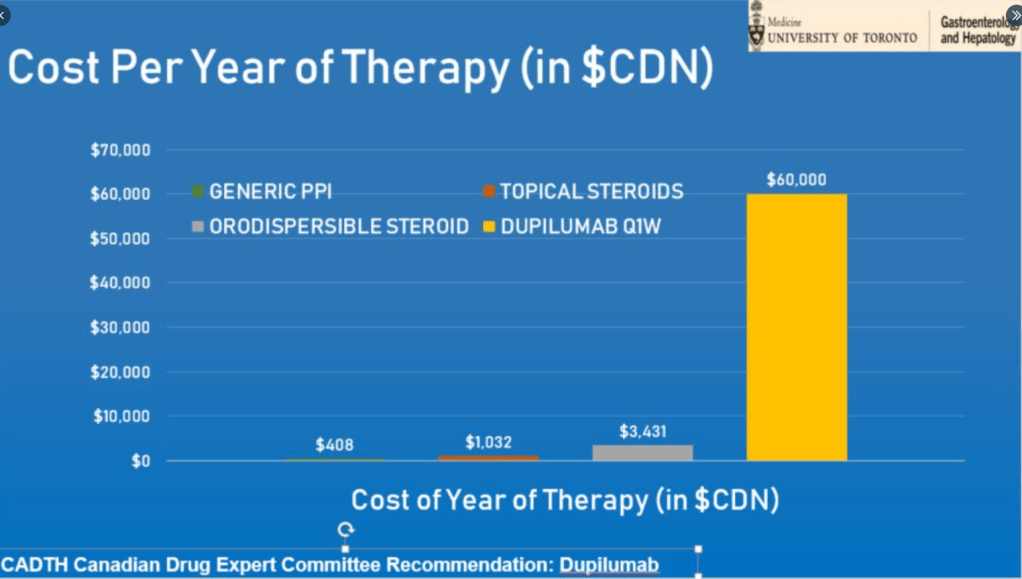
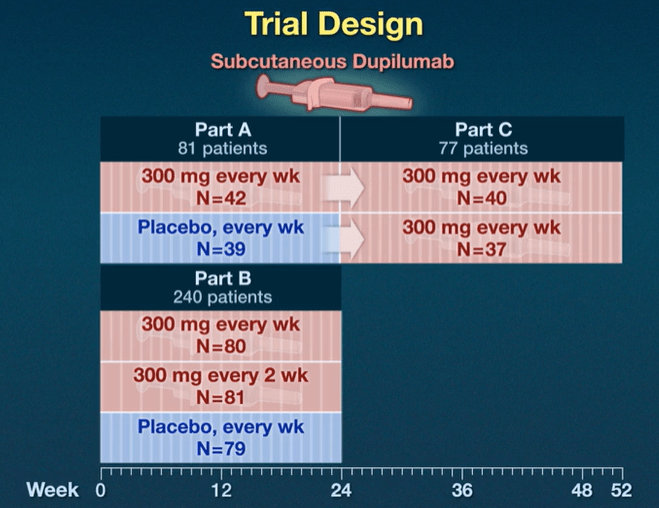
Key findings from three-part study (see images below):
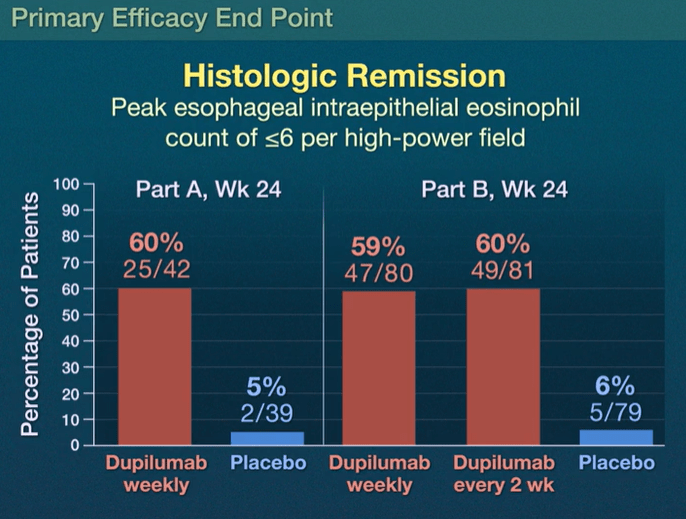
- Part A: Histologic remission occurred in 25 of 42 patients (60%) who received weekly dupilumab and in 2 of 39 patients (5%) who received placebo
- Part B: Histologic remission occurred in 47 of 80 patients (59%) with weekly dupilumab, in 49 of 81 patients (60%) with dupilumab every 2 weeks, and in 5 of 79 patients (6%) with placebo
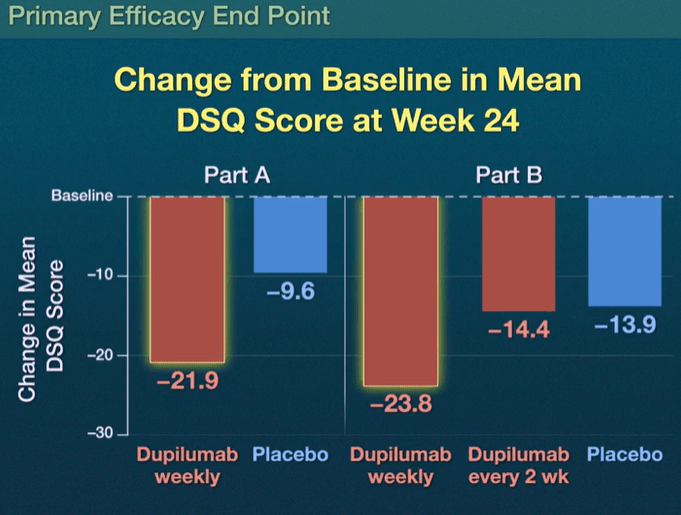
- Dysphagia Symptom Questionnaire (DSQ) scores: the scores improved with weekly dupilumab as compared with placebo, with differences of –12.32 (95% CI, –19.11 to –5.54) in Part A and –9.92 (95% CI, –14.81 to –5.02) in Part B (both P<0.001) but not with dupilumab every 2 weeks (difference in Part B, –0.51; 95% CI, –5.42 to 4.41)
- The most common adverse effect was injection site reactions. There were 10 serious adverse events; none of these “were considered by the trial investigators to be related to the trial regimen.”
In the associated commentary, (pg 2379-2380), Dr. Alex Straumann notes that since EoE is localized to the esophagus, whether a patients should be treated with a systemically acting medication, “particularly in light of the fact that topical glucocorticoids have been shown to be as efficacious as systemically acting prednisone.”
My take (borrowed in part from editorial): It remains unclear whether dupilumab “is better than the good old topical glucocorticoids in improving disease outcomes, particularly in light of considerable costs associated with this treatment.” Due to its cost (see below), dupillumab is likely best situated as a 2nd line treatment at this time for most patients.
Related blog posts:
- Dupilumab: FDA Approval for Eosinophilic Esophagitis
- New Therapy for Eosinophilic Esophagitis
- Promising Biologic for Eosinophilic Esophagitis
- How Genetics Influence Response Rate to PPIs in EoE
- Association and Causation: Early Life Risk Factors for Eosinophilic Esophagitis
- Negligible Effect of Eosinophilic Esophagitis Treatment on Longitudinal Growth
- Increased Risk of EoE with TEF/EA
- What is EoE?

Cost comparison (from Laura Targownwik on twitter):