K Manwani et al. JPGN 2022; 75: 252-256. Long-Term Growth in Children and Young People with Autoimmune Liver Disease Treated with Daily Steroids
This retrospective study of patients (n=74) diagnosed with autoimmune hepatitis (AH) prior to 16 years of age examined the safety of low dose steroids. Median age of patients with 12.8 yrs and median followup was 12.6 years. Typically, after induction, patients were tapered over ~2 months to 5 mg per day (& 2.5 mg per day if <12 years). Key findings:
- Growth of patients with AILD on a daily maintenance dose of steroids remains stable and within normal range during long-term follow up. At all time-points, the mean z-scores for weight, height and BMI were within the normal range, indicating normal nutritional status.
- Small, daily doses are effective in maintaining disease control and minimize the need for high-dose steroid pulses during relapses.
- In this cohort, there were 14 patients in which prednisolone was utilized as monotherapy; the majority received cotherapy with azathioprine (n=44), mycophenolate (n=12); triple-therapy was utilized tacrolimus (n=4).
- Prednisolone was stopped in 17 patients (23%) after a median time of 9.5 years (range 3 years-14 years)
M Biewenga et al. Clin Gastroenterol Hepatol 2022; 20: 1776-1783. Open access! Aminotransferases During Treatment Predict Long-Term Survival in Patients With Autoimmune Hepatitis Type 1: A Landmark Analysis
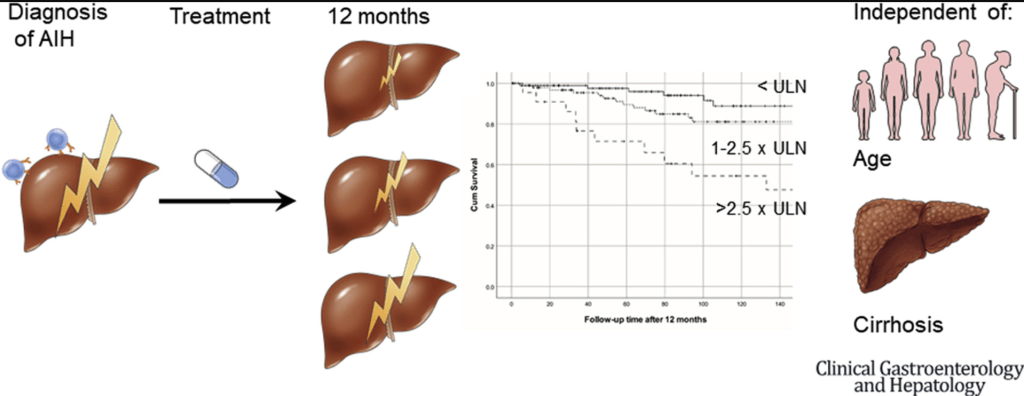
In this multicenter cohort study (n=301), it was shown that higher aminotransferases during treatment were independent of baseline risk factors associated with liver transplantation–free survival in patients with AIH type 1. Median followup was 99 months. Key finding:
- During follow-up, 15 patients required liver transplantation and 33 patients died
- In multivariate analysis AST at 12 months (HR, 2.13; P < .001) was predictive for survival independent of age, AST at diagnosis and cirrhosis, while IgG was not associated with survival (HR, 1.30; P = .53)
- There was a trend toward a worse survival in patients with mildly elevated aminotransferases (1–1.5× upper limit of normal) compared with patients with normal aminotransferases (P = .097)
My take: Normalization of AST (aminotransferases), especially during the first year of treatment, is associated with better long-term outcomes. The study by Manwani et al suggest that long-term low dose steroids are associated with low risks.
Also: Y Li et al.Hepatology 2022; 76: 564-575. Genome-wide meta-analysis identifies susceptibility loci for autoimmune hepatitis type 1 This study with 1622 Chinese patients identified two novel loci (CD28/CTLA4/ICOS and SYNPR) and may provide potential targets for additional treatments.
Related blog posts:
- Aspen Webinar 2021 Part 5 -Autoimmune Liver Disease & PSC
- Mortality Risk with Autoimmune Hepatitis
- Predicting Outcomes in Childhood Autoimmune Hepatitis
- Trends in Liver Diseases: Autoimmune Liver Diseases and Fatty Liver | gutsandgrowth
- Why It Is Hard to Stop Immunosuppression with Autoimmune Hepatitis and Lower Bone Density with Fatty Livers | gutsandgrowth
- Diagnosing autoimmune hepatitis which includes simplified scoring system