M Mendizabal et al. Liver Transplantation 2025; 31: 412-416. Have CFTR modulators changed the need for liver and lung transplantation among patients with cystic fibrosis? An analysis of the UNOS database
This article notes that there have been 146,851 waitlistings and 95,254 liver transplants in the U.S. between 2012 and 2023. This includes 194 waitlistings and 138 transplants in patients with cystic fibrosis.
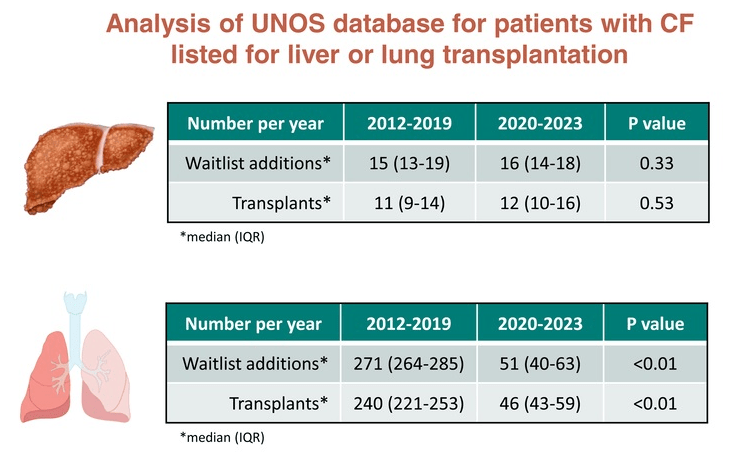

Key finding:

My take: This is great news for patients with cystic fibrosis. The drop in lung transplants is surely the tip of the iceberg. Think about your next breath! For patients with cystic fibrosis, these new medications make every single breath better. Longer followup is needed to determine if the long-term use of these agents may lower the rate of end-stage liver disease as well.
Related blog posts:
- CHOA Nutrition Support Lecture: Cystic Fibrosis Nutrition -Changing in the Age of ‘Miracle Drug’
- Aspen Webinar 2021 Part 7 -Cystic Fibrosis Liver Disease
- Intestinal Inflammation in Patients with Cystic Fibrosis
- Data on Immobilized Lipase Cartridge for Patients with CF
- Big Advance for Cystic Fibrosis -Who Will Benefit?






