More from Aspen Webinar 2021. This blog entry has abbreviated/summarized several presentations. Though not intentional, some important material is likely to have been omitted; in addition, transcription errors are possible as well. Great lecture from Jim Squires.

Key points:
- Cystic Fibrosis Liver Disease (CFLD) is variably defined
- Risk factors include male patients, DeltaF508 mutations, meconium ileus and SERPINA1 Z allele
- Two main phenotypes: Classic “Focal Biliary Cirrhosis” and Obliterative Portal Venopathy (increasingly recognized)
- Intestinal microbiome and gut permeability/endotoxins may influence liver disease
- Treatments: ursodeoxycholic acid may be helpful but overall evidence is low quality. Cochrane review does NOT recommend its routine use
- Treatments: liver transplant (thorough review: Freeman et al. Liver Transpl 2019; 25: 640-657)
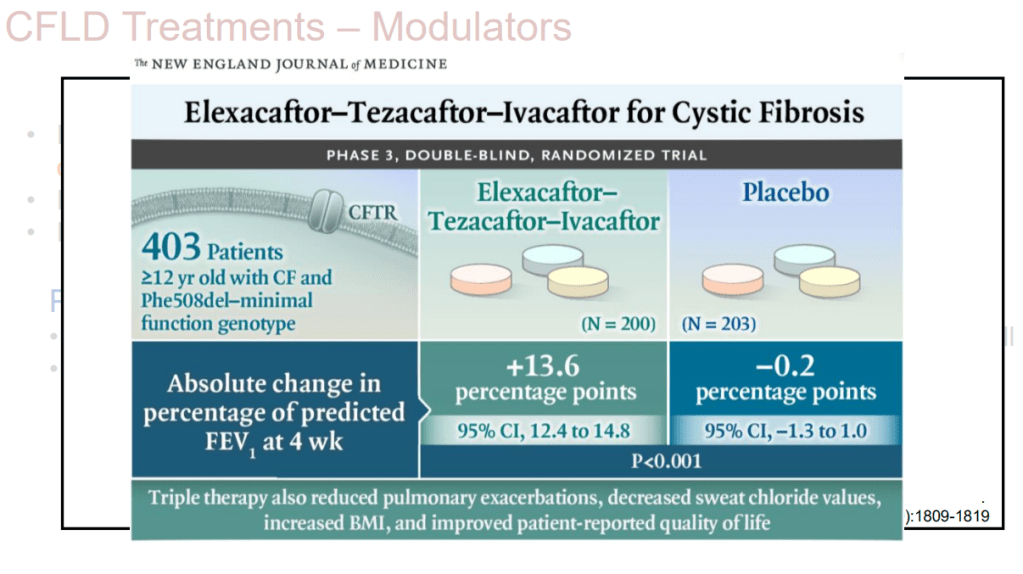
- Treatments: CF Modulators: potentiators (Ivacaftor), correctors (Lumacaftor, Elexacaftor, Tezacaftor)
- Treatment: Trikafta has been a game changer for CF lung disease. Its effects on the liver are not clear yet
Some of the slides:




Related blog posts:
- CHOA Nutrition Support -CF in the age of “Miracle Drug”
- Data on Immobilized Lipase Cartridge for Patients with CF
- Cystic Fibrosis Expert Update 2017
- Nutrition Pearls: Fiber and Cystic Fibrosis
- Big Advance for Cystic Fibrosis -Who Will Benefit?
- Nutrition Guidelines for Cystic Fibrosis
- Complex Family of CFTR-Associated Disorders | gutsandgrowth
SAVE THE DATE for next year’s conference: July 11-15, 2022 in Snowmass Village, CO