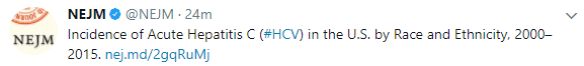With non-alcoholic steatohepatitis (NASH), there are currently no established medical therapies. However, several candidate medications look promising. However in recent years, many new medications have come with an impressive price tag and this has led to questions about whether emerging therapies for NASH will be affordable.
A recent article looked at the medication Obeticholic Acid, which was approved for treating primary biliary cholangitis. It is possible that it will be helpful for NASH. Yet, its cost , currently, is about $70,000 per year
GIHepNews: Despite clinical promise, obeticholic acid may be too expensive for treating NASH
Here’s an excerpt:
In the 72-week Phase II trial, called FLINT, 273 men and women with NASH were randomly assigned to receive OCA or placebo (Lancet 2015;385:956-965). Liver histology improved in 45% of those receiving OCA versus 21% in those receiving sham therapy (P=0.002). An increased risk for pruritus was the most notable adverse event among patients taking OCA (23% vs. 6% for placebo), according to the researchers. Based on the favorable benefit–risk results of the Phase II study, a Phase III trial is ongoing…
The expected benefit of OCA over lifestyle modifications for all the major long-term outcomes, such as decompensated cirrhosis (10% vs. 9.4%), liver-related mortality (9% vs. 8.1%) and transplant-free survival (72.2% vs. 71.5%), were relatively modest, the researchers reported. Those differences resulted in a cost per quality-adjusted life-year saved of $5.2 million with the assumption that 16% of patients would relapse…
“If the efficacy compared to placebo is of the same order found in the FLINT trial, the current cost of the drug would be prohibitive in a population-based context,” said Dr. Lavine, who was a co-investigator on the trial.
My take: Given the growing burden of NASH, new effective treatments are needed. In my view, though, cost-effectiveness has to be a consideration.

Prague Castle