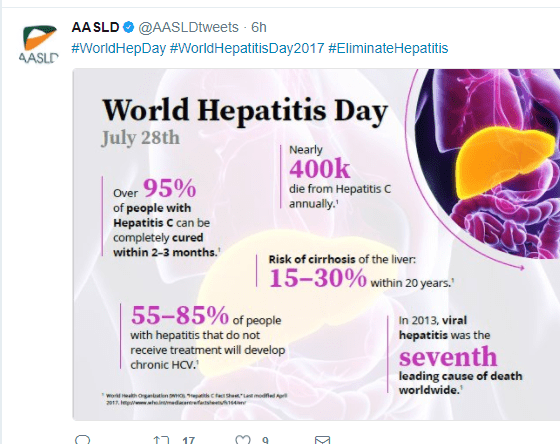Caution with hemoglobin A1c interpretation: MM Kelsey et al. J Pediatr 2020; 216: 232-5. In the HEALTHY Study (n=8814), the authors note that a hemoglobin A1c was ≥5.7% in 2% of normal weight youth. “This suggests need for cautious interpretation of prediabetes hemoglobin A1cs in youth”
Daily aspirin for NAFLD: TG Simon et al. Clin Gastroenterol Hepatol 2019; 17: 2776-84. In this prospective cohort of 361 adults with biopsy-proven NAFLD, the use of daily aspirin (in 151) was associated with lower odds of NASH (aOR.68) and reduced risk of fibrosis (aOR 0.54). “The greatest benefit found with at least 4 years or more of aspirin use” (aHR =0.50). The associated editorial (pages 2651-3) recommends controlled studies to determine if potential benefits outweigh the known risks (eg. bleeding).
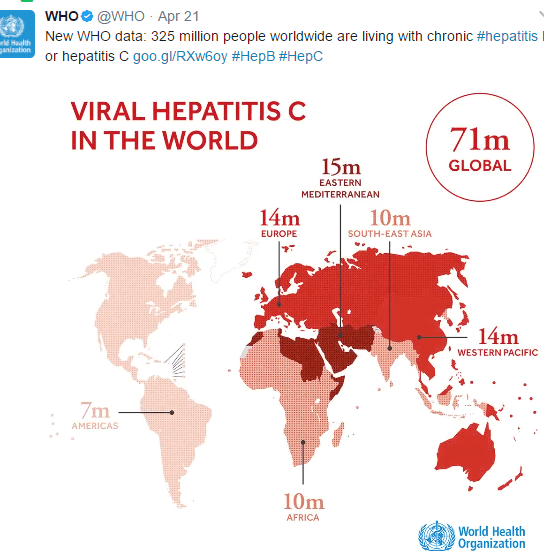
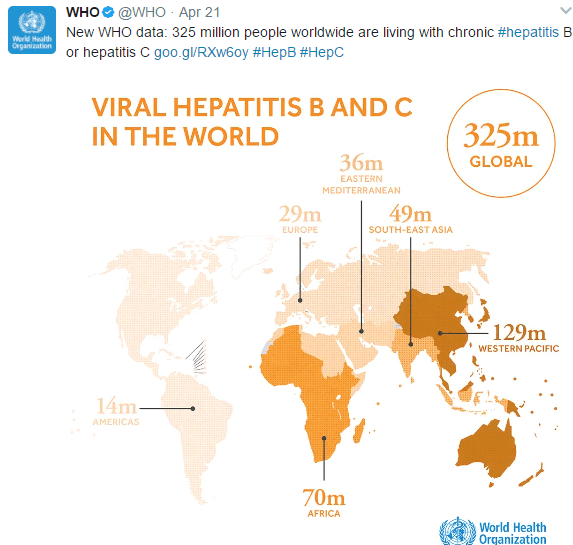
Glecaprevir/pibrentasvir for HCV Treatment Failure: AS Lok et al. Gastroenterol 2019; 157: 1506-17. This randomized study with 177 patients showed that 16 weeks of glecaprevir and pibrentasvir was effective in retreatment of patients with genotype 1 hepatitis C viral infection (after prior failure with sofosbuvir plus an NS5A inhibitor). The sustained virologic response 12 weeks after treatment was >90%.
Liver transplantation for Niemann-Pick Disease, type B: YLY Luo et al. Liver Transplantation 2019; 25: 1233-40. This report analyzed 7 children receiving liver transplantation for Niemann-Pick disease, type B. The authors report survival in the entire cohort and with normalized liver function within 3 weeks. In addition, they noted improvement in psychomotor ability ( 10 months after transplantation) and resolution of insterstitial lung disease. They state that developmental delay still existed in 4 patients during follow-up. The editorial (1140-1) notes that these findings need to be confirmed but open a new window in improving the phenotype. “A similar experience occurred with LT in maple syrup urine disease (MSUD), in which the liver is considered to host only 12-15% of the defective enzyme responsible for the disease…in MSUD, liver replacement is able to counteract 85% of extrahepatic expression of the disease and to completely correct the phenotype.”
Increased Abdominal-Surgery Risk in Patients with Idiopathic Noncirrhotic Portal Hypertension: L Elkrief et al. Hepatology 2019; 70: 911-24. Among 44 patients (median age 44 years) with noncirrhoitic portal hypertension, 16 (33%) had one or more portal hypertension-related complication within 3 months after surgery. 4 (9%) died within 6 months. “An unfavorable outcome (i.e. either liver or surgical complication or death) occurred in 22 (50%) patients” and was more likely in those with ascites, creatinine >100 micromol/L, or other extrahepatic complications related to portal hypertension.