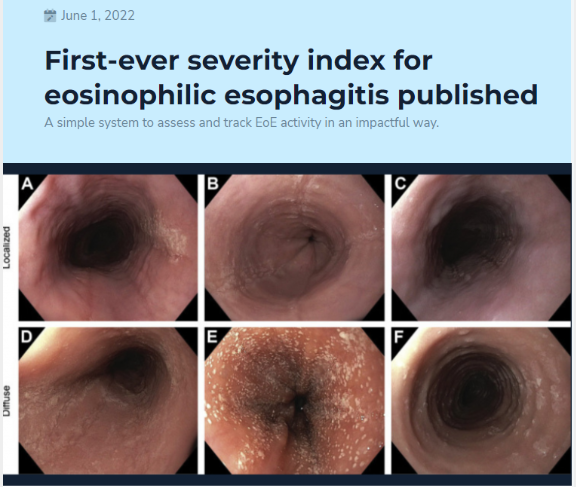
A Dickerson et al. Clin Gastroenterol Hepatol 2024; 22: 732-740. The Index of Severity for Eosinophilic Esophagitis (I-SEE) Reflects Longitudinal Clinicopathologic Changes in Children
This was a retrospective analysis on a prospectively enrolled cohort of children at a single center who were treated as part of routine clinical care. I-SEE was calculated at the diagnostic and follow-up endoscopies over a mean of 6.6 years.
Key findings:
- Of 67 children who met study criteria of at least 3 endoscopies over at least 2 years of follow-up time, 43%, 36%, and 21% had mild, moderate, and severe I-SEE scores at baseline, respectively.
- By the last instance, the overall I-SEE score dropped to 3.9 (P < .001). Body mass index <5% and poor feeding were more common in the children with severe I-SEE scores at baseline, and both improved by the last instance.
The discussion notes that I-SEE metric was developed to determine EoE severity and for tracking purposes to gauge effectiveness of therapy. They note that most patients improved but a score of 0, indicating deep remission, was difficult to achieve at the population level. They also anticipate further modifications to I-SEE “such as age or an assessment of symptoms that reflects inflammatory or fibrotic disease.”
My take: I-SEE provides a way to objectively assess and follow EoE severity at all ages.
Related blog posts:
- I-SEE for Eosinophilic Esophagitis
- Practical Tips for Eosinophilic Esophagitis
- AGA: High Quality Upper Endoscopy
- “Esophageal Hypervigilance” and Outcomes in Eosinophilic Esophagitis
- “Real-World” Dupilumab for Eosinophilic Esophagitis
- Dupixent dosage (per manufacturer): 15 kg-<30 kg: 200 mg every 2 weeks, 30 kg-<40 kg: 300 mg every 2 weeks, >40 kg: 300 mg every week
Link: I-SEE Tool Scoring Table
AGA has an I-SEE App available: AGA I-SEE App

