Advances in Hemostasis for Upper GI Bleeding —Brad Barth, MD, MPH (page 77)
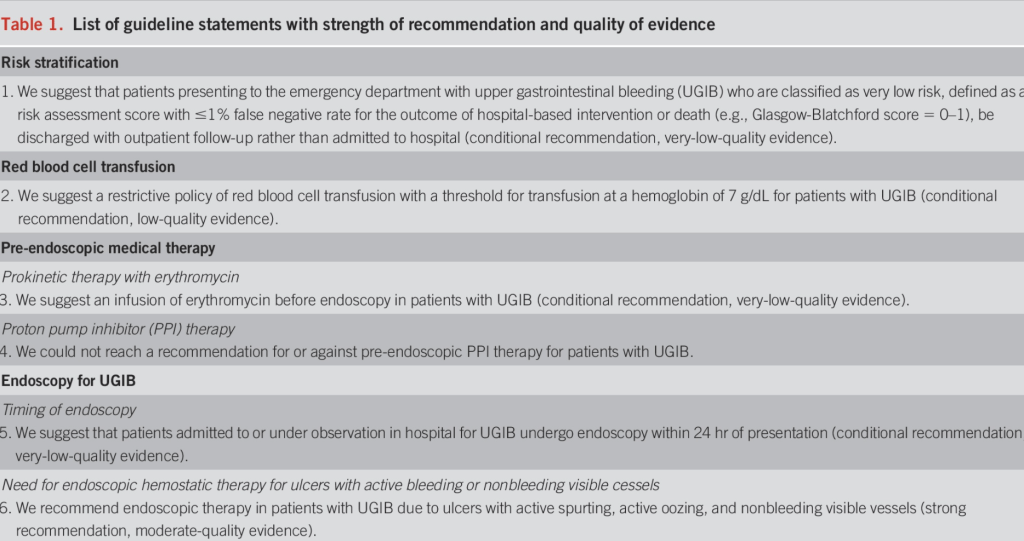
Upper GI Bleeding
Effect of IV PPI on patients with UGI bleeding PRIOR to EGD
- 6 trials including 2223 patients
- No significant difference in mortality, rebleeding or need for surgery compared to controls
- DID significantly reduce rates of high risk stigmata identified on EGD
- DID significantly decrease the need for endoscopic therapy
- Reference: Sreedharan A, Martin J, Leontiadis G, et al. Proton pump inhibitor treatment initiated prior to endoscopic diagnosis in upper gastrointestinal bleeding. Cochrane Database of Systematic Reviews 2010
Upper GI Bleeding – Proton Pump Inhibitors/Prokinetics
- Omeprazole 1 mg/kg q 12 hours (Solana, et al. J Pediatr 2013:162:776-82)
- Proposed PPI drip dose: 1 mg/kg bolus followed by 0.1 mg/kg/hour infusion
- IV erythromycin or metoclopramide; infuse 20-120 minutes prior to endoscopy in patients with acute UGIB; decreased need for repeat endoscopy to determine cause and site of bleeding. Prokinetic did NOT affect transfusion requirements, duration of stay, need for surgery. Reference: Barkun et al. Prokinetics in acute upper GI bleeding:a metaanalysis. GIE 2010;17:126-132
Upper GI Bleeding —Other points:
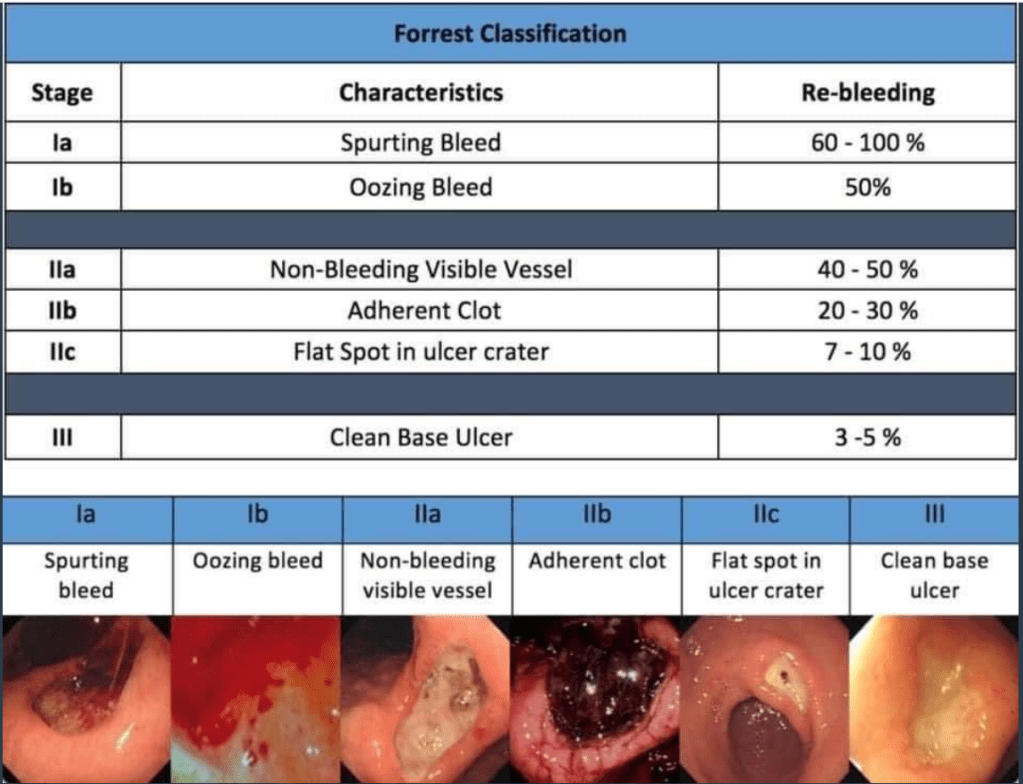
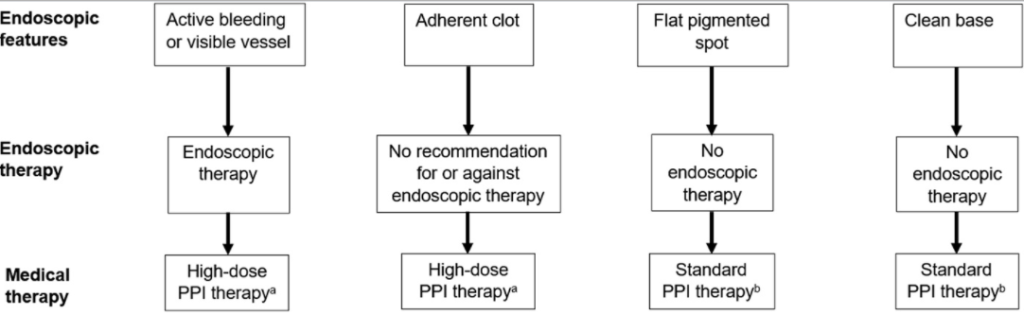
- Epinephrine alone is RARELY enough
- Non bleeding adherent clot has 8-35% chance of rebleeding in adults. Consider removing it CAREFULLY!
- A conservative transfusion strategy is usually appropriate
Useful References
Surveillance Endoscopies: The established, the debated, and the unknown –Mitchell Shub, M.D. (page 85)
“Beware of false knowledge; it is more dangerous than ignorance.” —George Bernard Shaw
Familial Adenomatous Polyposis
Surveillance protocol: Age of initial evaluation/Type of procedure/Frequency
- Colon 10 – 12 y of age (Sooner: family h/o aggressive disease) -Flex sig or Colonoscopy, 1 – 2 y
- Upper GI tract 20 – 25 y or at initial colonoscopy
- EGD and side viewing scope, 1 – 3 y
- Post-colectomy (pouch) 6 – 12 mo. after surgery, Flex sig 1 y (6 mo. If retained rectum)
- Small bowel: capsule or MRI, frequency unknown
Peutz-Jeghers Syndrome: begin screening at age 8 years or when symptomatic with colonoscopy, EGD, and small bowel imaging (?capsule vs alternatives); then every 2-3 years
Juvenile Polyposis Syndrome: begin screening at age 10-15 years or when symptomatic with colonoscopy, EGD, and possibly small bowel imaging (?capsule vs alternatives); then every 1-3 years
Discussed guidelines for IBD cancer surveillance and for Barrett’s esophagus
- For UC, start surveillance 8-10 years after diagnosis.
- For Crohn’s with ~1/2 colon (or more) involvement, follow same guidelines
- For coexisting PSC, annual surveillance
- Barrett’s esophagus in children: adenocarcinoma very rare, evidence lacking to develop surveillance schedule
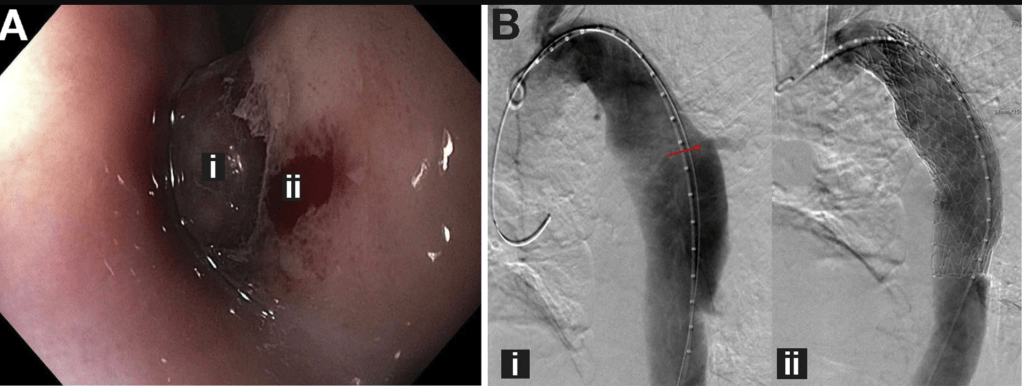
Expanding the view: Update on Upper GI Strictures —Mark A. Gilger, M.D. (page 95)
Why balloons for kids (for dilatation)?
You can see what you’re doing
- Blind pouches
- Abnormal mucosa
- Caustic injury
- Epidermolysis bullosa
- Already requires general anesthesia
- Ability to wire through narrow strictures
- Ability to use radiographic assistance
Tip: Can use vegetable spray (eg. Pam) to make advancement of balloon catheter easy
How to do balloon dilation
- Inflate balloon to ½ desired initial atmospheres & re‐check placement
- Begin dilation at to 1‐2 mm more than initial estimated stricture diameter
- Hold for 1 minute/dilation
- •ove balloon catheter in and out during dilation; if balloon moves freely, increase diameter by 1mm. If stricture moves with the balloon, hold x 1 minute, then done
- Oh, oh, there’s blood! Good! No blood, no dilation.
- After dilation, carefully advance endoscope through the stricture; if resistance stop, can try cork‐screw maneuver
- Document everything; especially stricture location (CM from incisors), dilation diameters (to help you next time)
Adjunct therapy for recalcitrant strictures –adjunct therapy to sustain dilation needs further study
- Oral & intravenous corticosteriods
- Injectable corticosteroids – Thins the mucosa, OK 1‐2 times, but not repeated
- Mitomycin C
- Acid reduction
- Stents
Endoscopy in the high‐risk patient: Keeping your patient safe —Jenifer R. Lightdale, MD, MPH (page 63)
Safety of Pediatric GI Procedures
- Peds‐CORI data from >10,000 procedures
- Overall rate of complications 2.3%: risk of hypoxia 1.5%; risk of bleeding 0.3%
Examples of pediatric populations at increased risk for perforation
- History of caustic ingestion
- Esophageal atresia/tracheo‐esophageal fistula
- Severe duodenitis
- Severe ulcerative colitis
- Patients with multiple co‐morbidities (i.e. Type I diabetes, cerbrovascular disease, peripheral vascular disease, renal insufficiency, liver disease)
- Ehlers‐Danlos Syndrome (Vascular Type)
Pre‐procedure Assessment –lends itself to a checklist
Thrombocytopenia -Current recommendations
- EGD ok if platelets >20,000/mL
- Biopsies ok if platelets >50,000/mL
Bleeding –discussed high risk conditions
Decreasing Risk of Perforation:
- Avoiding excessive pressure
- Avoiding premature cutting of a polyp – Coagulate before cutting
- Avoiding blind intubation of the lumen
Decreasing Risks of Infection
- SBE Prophylaxis– generally NOT indicated in diagnostic procedures. Congenital heart disease is complex & may be needed on a case‐by‐case basis
- Single‐dose cephalexin has been shown to decrease peristomal infection during PEG placement
- Prophylactic antibiotics recommended for cirrhotic patients admitted with GI hemorrhage
Postgraduate Course Syllabus (posted with permission) with complete slides of above lectures: PG Syllabus
Related blog references:
Disclaimer: These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) and specific medical management interventions should be confirmed by prescribing physician. Application of the information in a particular situation remains the professional responsibility of the practitioner.