RJ Colman et al. AP&T 2022; https://doi.org/10.1111/apt.17277. Open access! Real world population pharmacokinetic study in children and young adults with inflammatory bowel disease discovers novel blood and stool microbial predictors of vedolizumab clearance
“The study included data from 463 observed vedolizumab concentrations (59 peaks and 404 troughs) from 74 patients with IBD (52 with Crohn’s disease and 22 with ulcerative colitis or unclassified IBD, median age 16 years)…This study was part of the multicentre REFINE study, which aimed to investigate paediatric PK factors among different biological therapies. Both induction and maintenance doses were between 6 and 10 mg/kg for patients less than 30 kg and 300 mg for patients above 30 kg.”
Key findings:
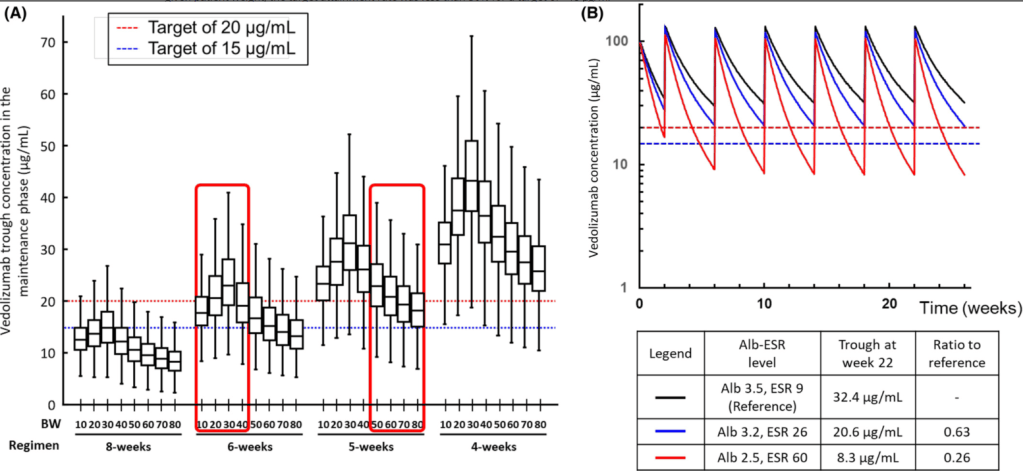
- “Using the new model in a simulation analysis of standard vedolizumab infusions (0, 2 and 6 weeks followed by every 8 weeks), we demonstrate that the expected cTrough at week 22 (infusion-5) in the majority of patients would result in drug exposure below current cTrough targets..The dosing simulations in our current study found that receiving standard dosing would lead to <20% of patients achieving a cTrough of 20 μg/ml at infusion-5.”
- “The severity of hypoalbuminemia resulted in higher drug CL (lower cTrough) than the inflammatory burden (elevated ESR).”
- Infusion-3 cTrough of at least 37 μg/ml and infusion-4 cTrough of at least 20 μg/ml best predicted SFCR (steroid-free clinical remission) at infusion-4. In contrast, we showed inadequate drug exposure during induction (AUCweek 14 of <134,580 μg h/ml) was associated with clinical non-response
My take: This study shows that therapeutic drug monitoring (TDM) is likely to be beneficial in improving outcomes in pediatric patients receiving vedolizumab. Low albumin in particular is associated with increased drug clearance. From this study, it looks like most pediatric patients will need dosing every 4 to 6 weeks to achieve good levels. The authors in their discussion reinforce the utility of TDM to “guide anti-TNF dose optimisations has been shown to improve durability and reduce both immunogenicity and loss of response.”
References:
13 Dubinsky MC, Mendiolaza ML, Phan BL, Moran HR, Tse SS, Mould DR. Dashboard-driven accelerated infliximab induction dosing increases infliximab durability and reduces immunogenicity. Inflamm Bowel Dis. 2022; 28: 1375– 85.
51 Strik AS, Löwenberg M, Mould DR, Berends SE, Ponsioen CI, van den Brande JMH, et al. Efficacy of dashboard driven dosing of infliximab in inflammatory bowel disease patients: a randomized controlled trial. Scand J Gastroenterol 2021; 56: 145– 154.
Related blog posts:
- Improving Outcomes with Proactive Therapeutic Drug Monitoring + Swiss COVID-19 Data
- Expert Consensus: New Recommendations for Therapeutic Drug Monitoring
- Combination Therapy Study Points to Central Role of Adequate Drug Levels
- Can Therapeutic Drug Monitoring with Monotherapy Achieve Similar Results as Combination Therapy in IBD?
- “Denials, Dilly-dallying and Despair”
- Low Anti-TNF Levels or Antibodies Are Associated with Antibodies to Subsequent Anti-TNF Agent
- For Next Insurance Appeal: TDM for Adalimumab & Satire on PA
- Here’s the Proof That Proactive Therapeutic Drug Monitoring Improves Outcomes in Children with Crohn’s Disease

Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.
