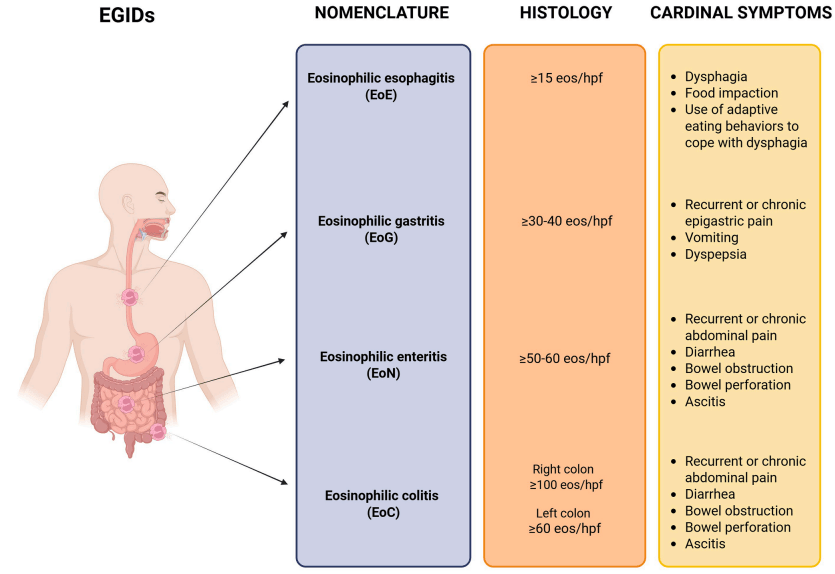
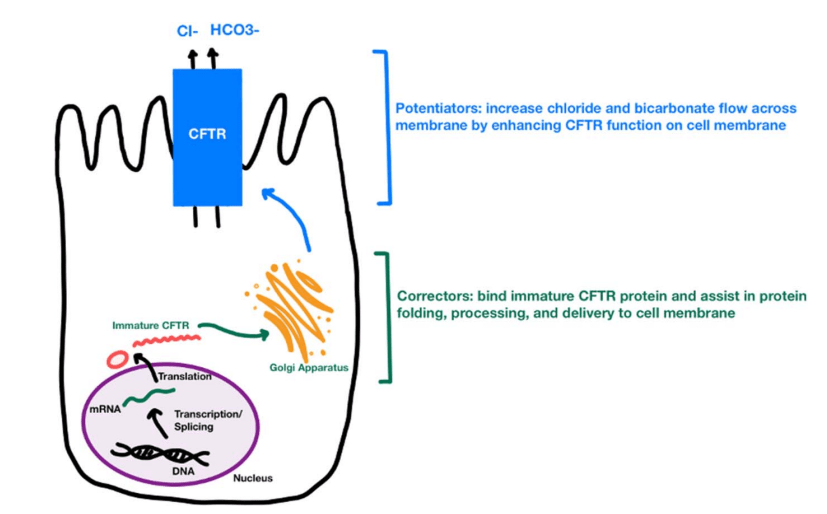
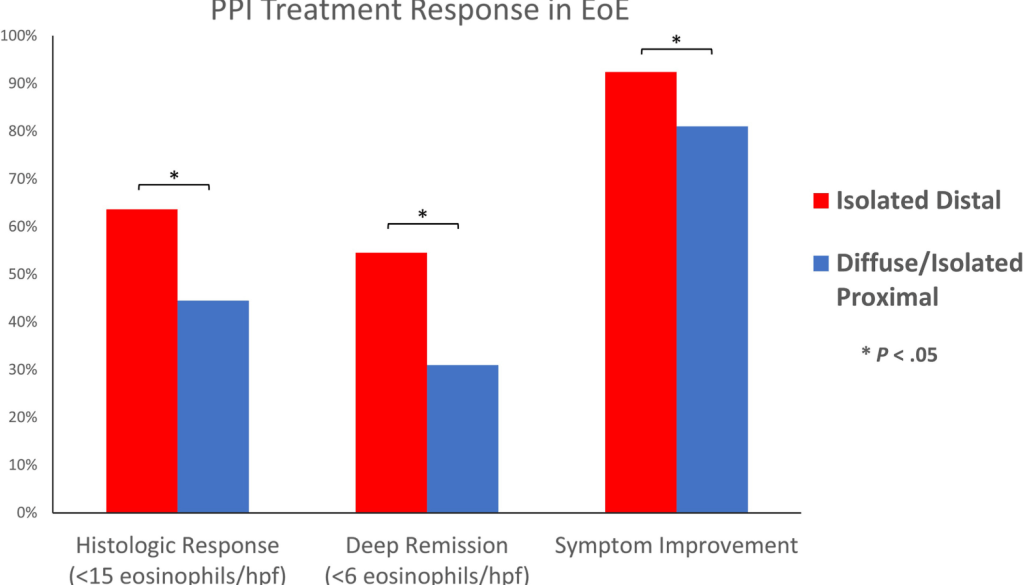
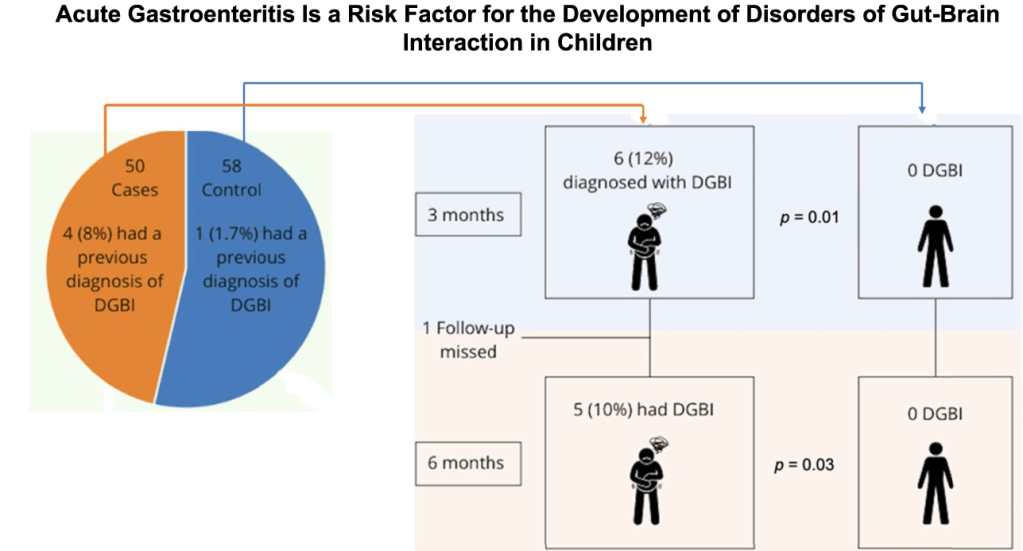
MD Stutvoet et al. J Pediatr Gastroenterol Nutr. 2026;82:699–707. Open Access! Fatigue in pediatric inflammatory bowel disease: Explained by transdiagnostic and disease-focused factors
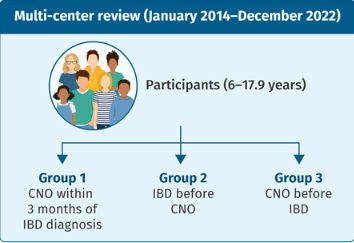
Methods: “The PROactive cohort is a longitudinal study that collects transdiagnostic patient-reported outcomes on fatigue, daily life participation, and psychosocial well-being in children with chronic conditions. It is based at the Wilhelmina Children’s Hospital in the Netherlands.” this study enrolled 127 patients with IBD.
Key findings:
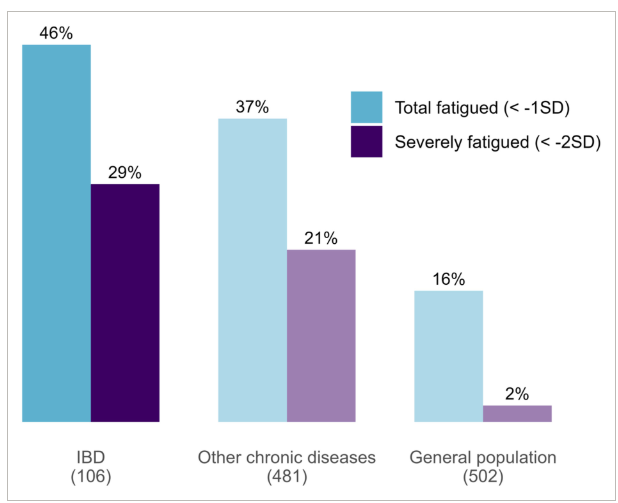
- Fatigue is highly prevalent in pediatric IBD, with 29% of patients reporting severe fatigue—despite most being in clinical remission
- 78% of variance in fatigues explained by transdiagnostic lifestyle, psychological and social factors
Discussion Points:
- “Clinical disease activity was significantly associated with fatigue in univariate analysis (p < 0.01). This is consistent with earlier studies showing modest associations between clinical disease activity and fatigue2–4, 33–35 and no association with biochemical markers such as FC, CRP, and anemia.2–4, 11, 35 The discrepancy between clinical and biological markers may reflect the inclusion of functional limitations in the clinical disease activity scores, and their weak correlation with measures of biological disease activity as FC.36… Taken together, the association with clinical disease activity shows that fatigue may be closely linked to perceived disease-related limitations rather than inflammatory activity alone.”
- “Transdiagnostic factors—including depressive symptoms, poor sleep, and social functioning—were strongly associated with fatigue, consistent with previous findings in pediatric IBD cohorts,3, 4, 33 and other chronic diseases.14, 16…Our results emphasize the complex interactions among psychosocial factors and support a more integrative approach to fatigue assessment. “
My take (borrowed in part from the authors): “While disease activity may initially precipitate fatigue, other factors—such as poor sleep or school pressure—can perpetuate it, even after inflammation resolves.”
Related blog posts:
- A Treatment for Severe Fatigue Associated with Inflammatory Bowel Disease?
- #MondayNightIBD and Fatigue
- IBD Updates: Extending Mirkizumab Induction, Best Biologic, Fatigue in Pediatric IBD, Adalimumab Success in Patients with Abdominal Abscess
- IBD Updates: Fatigue Trajectory, Risk of IBD with Derm Findings