AJ Ullman et al. NEJM 2025; 392: 161-172. A Comparison of Peripherally Inserted Central Catheter Materials
Background: Advances in material technology have introduced coatings and altered surfaces with purported antithrombotic and antiinfective properties to be used with polyurethane PICCs…A Cochrane review11 that included data from 42 randomized, controlled trials and 10,405 patients concluded that there was high-quality evidence showing that antimicrobial central venous catheters reduced the risk of bloodstream infections (risk ratio, 0.62)…On the basis of this indication, chlorhexidine-coated PICCs have been introduced to clinical practice… We conducted the Peripherally Inserted Central Catheter Innovation to Reduce Infections and Clots (PICNIC) trial to test the hypothesis that the risk of device failure due to complications would be lower with two technological innovations (hydrophobic and chlorhexidine PICCs) than with standard polyurethane PICCs.”
Methods: The authors conducted a randomized, controlled, superiority trial in three Australian tertiary hospitals. Adults and children (n=1098) who were referred for PICC placement were assigned in a 1:1:1 ratio to receive a hydrophobic or chlorhexidine PICC or a standard polyurethane PICC and were followed for 8 weeks
Key findings:
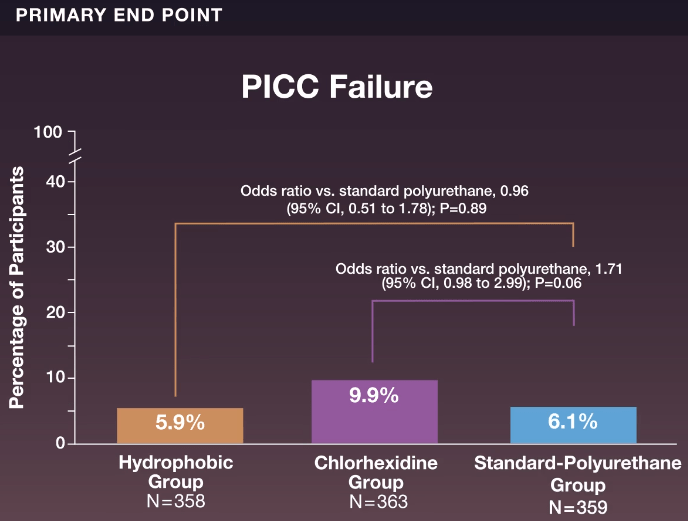
- Device failure occurred in 21 of 358 participants (5.9%) in the hydrophobic group, in 36 of 363 (9.9%) in the chlorhexidine group, and in 22 of 359 (6.1%) in the standard-polyurethane group
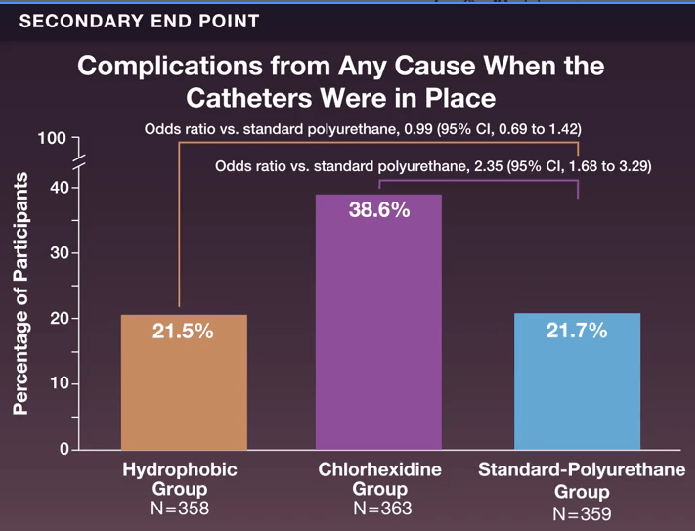
- Complications from any cause during the period of PICC placement occurred in 77 participants (21.5%) in the hydrophobic group, in 140 (38.6%) in the chlorhexidine group, and in 78 (21.7%) in the standard-polyurethane group (odds ratio, hydrophobic vs. standard polyurethane, 0.99)
Discussion point: This trial was conducted during the COVID epidemic which may have altered the results due to a focus of mitigating infectious exposures.
My take (borrowed from authos): “The risk of device failure due to noninfectious or infectious complications was not lower with hydrophobic or chlorhexidine PICCs than with standard polyurethane PICCs.”
Related blog posts:
- Practical Intestinal Rehabilitation (Part 1)
- Practical Intestinal Rehabilitation (Part 2)
- Antibiotic Selection for Suspected Central Line Infections (2020)
- When Is It Safe To Replace a Central Line in the Setting of Candida Infection?
- Prevalence of Bloodstream Infections in Children with SBS and Fever
- Short Bowel Syndrome is a Full Time Job