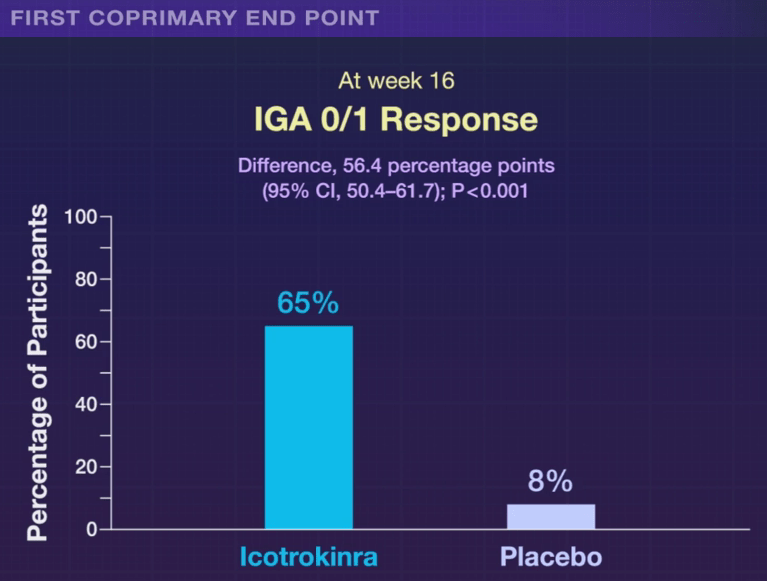
Friday’s blog post (Position Paper: Expediting Drug Approval for Pediatric IBD) has been updated to reflect the recent approval of Stelara (ustekinumab) for the treatment of pediatric patients 2 years and older with moderately to severely active Crohn’s disease. Here’s a link: Johnson & Johnson (JNJ) Gains FDA Approval for Pediatric Crohn’s Disease Treatment. Thanks to Matthew Kowalik, MD, the Director (Acting) of Division of Gastroenterology for FDA/CDER/OND/OII, for bringing this to my attention.
Congratulatons to Rachel Rosen who is the pediatric gastroenterology recipient of this year’s AGA Institute Council Section Research Mentor Award.

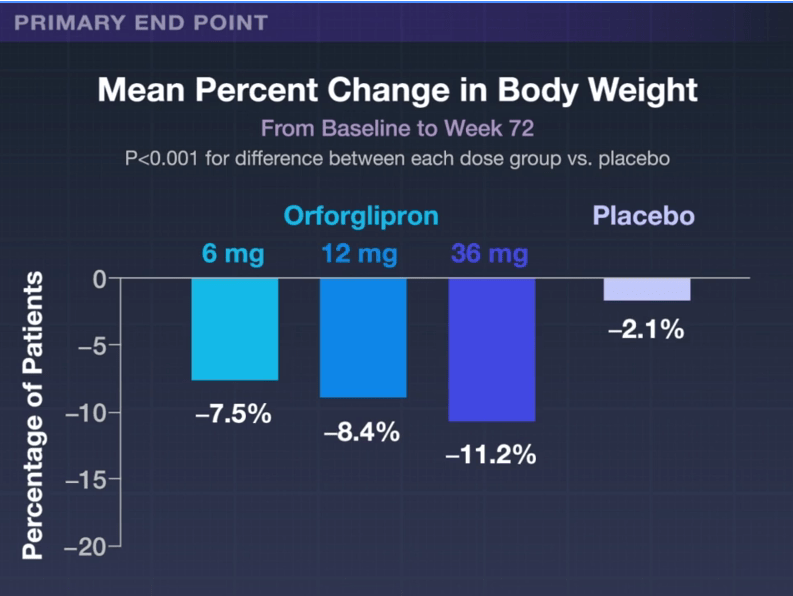
B Lovelace Jr, NBC News, 4/1/26: FDA approves weight loss pill from Eli Lilly
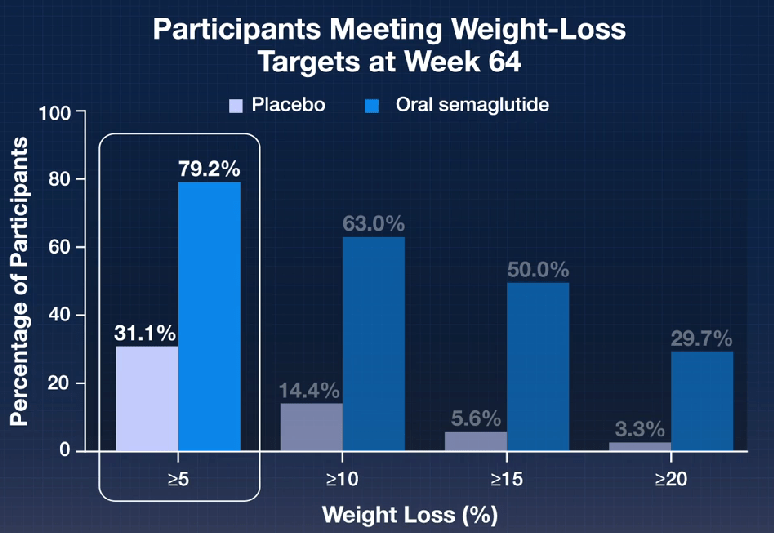
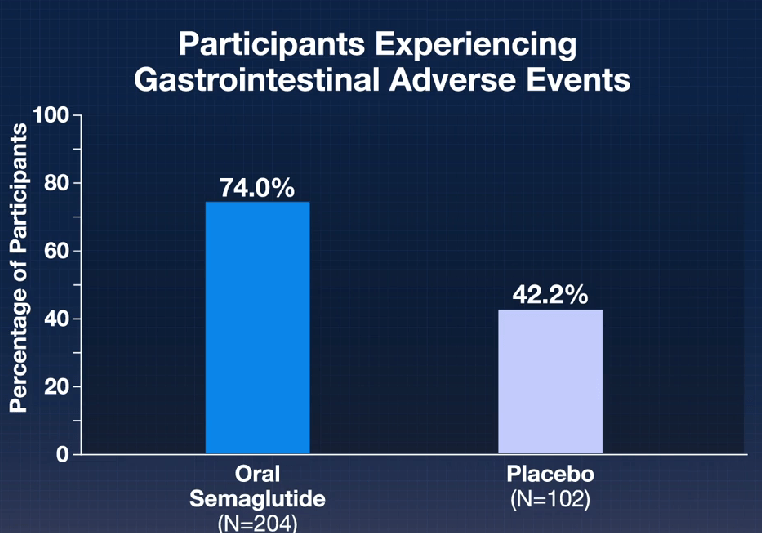
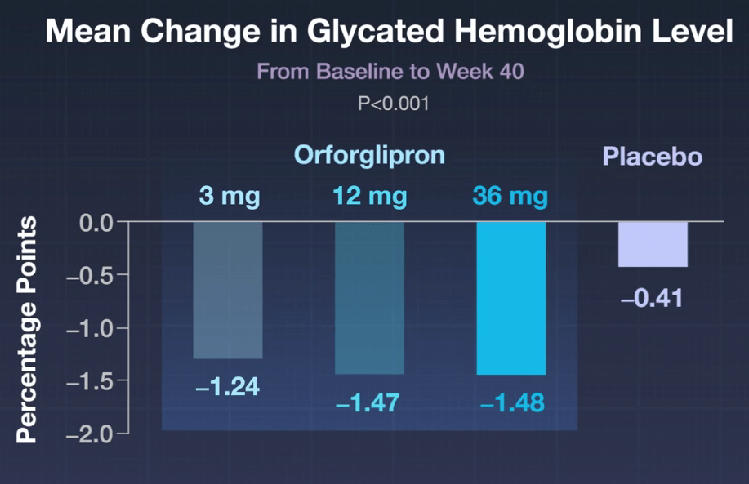
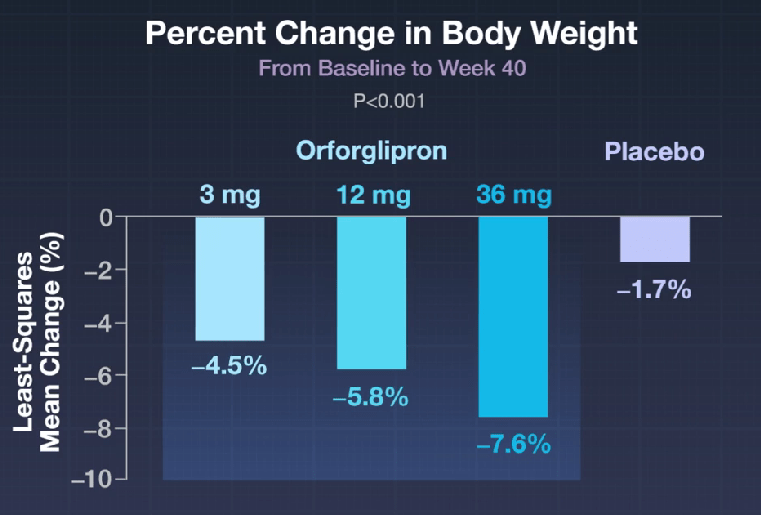
“The daily pill, called Foundayo [orforglipron], follows the approval of Novo Nordisk’s Wegovy pill in recent months. The lowest dose is expected to cost $149 a month for people paying out of pocket…Foundayo will be offered in six doses, with patients typically starting on the lowest dose and working their way up to reduce side effects. It can be taken at any time of day without meal restrictions — unlike the Wegovy pill, which must be taken on an empty stomach each morning…Phase 3 clinical trial data found that Foundayo helped people lose 12.4% of their weight, on average, at its highest dose after 72 weeks — similar to the Wegovy pill but less than injectable versions of Wegovy and Zepbound.”
The FDA has asked Lilly for more safety data. NBC News 4/14/26: FDA asks Lilly for more safety data on weight loss pill Foundayo
Related blog posts:
- Oral GLP-1 Receptor Agonist for Obesity: Orforglipron
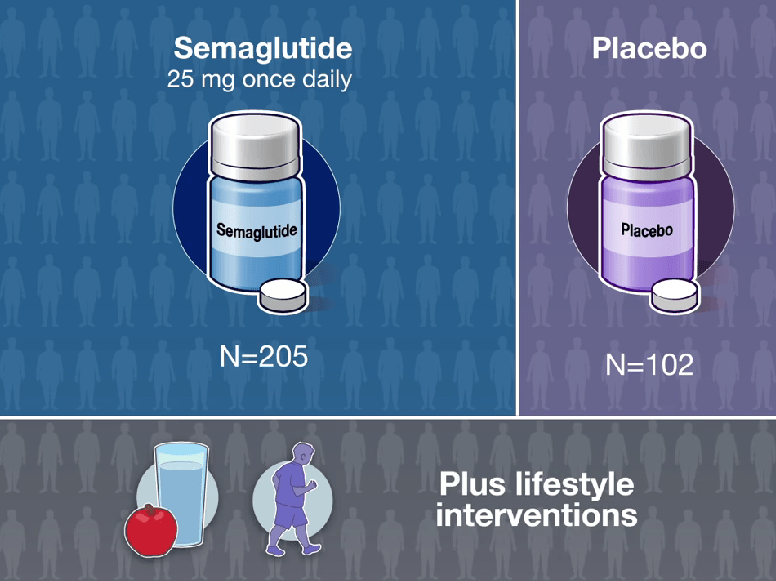
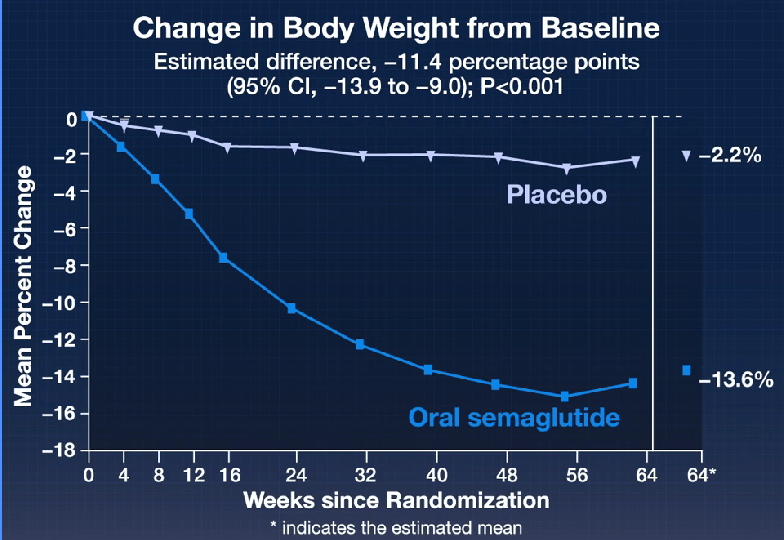
- The Rise of Oral Obesity Therapies: Semaglutide and Orforglipron
- Reduced Dosing of GLP-1 RAs May Work for Maintenance Treatment of Obesity
- 75% of U.S. Adults Have Obesity When Using New Definition With Anthropometrics (2025)

Also, there was a recent article in Nature (Genetic predictors of GLP1 receptor agonist weight loss and side effects) showing that a genetic variant can predict whether GLP-1 response is more robust. Explainer: Why obesity drugs work better for some people: these genes hold clues
AS Bukhbinder et al. Neurology 2026; 106 (8) e214782. Open Access! Risk of Alzheimer Dementia After High-Dose vs Standard-Dose Influenza Vaccination
This was a retrospective study comparing the high dose influenza vaccine (H-IIV) compared to the standard dose (S-IIV) in 65+ patients. The H-IIV group included 120,775 unique participants (185,183 person-trials; mean age 74.4 years, SD 5.5; 57.3% female), and the S-IIV group included 44,022 participants (53,918 person-trials; mean age 73.0, SD 6.1; 56.4% female).
Key findings:
- H-IIV was associated with significantly lower AD risk during months 1–25 postvaccination (minimum NNT = 185.2 at 25 months), with a stronger effect among women.
- Related blog posts:
- Shingles Vaccine Linked to Lower Dementia Risk
- Flu Shots & Other Vaccines Linked to Lower Rates of Dementia

Also, infectious disease outbreaks are expensive. M Baker, American Action Forum 4/3/26. Vaccines Protect More Than Health – They Protect Economic Capacity
“Routine childhood immunizations in the United States from 1994 through 2023 are estimated to have prevented roughly 508 million illnesses, 32 million hospitalizations, and more than 1 million deaths. Those gains translated into approximately $540 billion in direct medical cost savings and nearly $2.7 trillion in total societal savings. Put simply, every $1 spent on childhood immunizations generated about $11 in savings…Adult vaccination also produces meaningful economic gains, particularly through reduced absenteeism, less presenteeism, and better labor-market continuity. Evidence suggests that adult immunization programs can return up to 19 times their initial investment.”
“In South Carolina, a 2025–2026 outbreak with 993 cases generated an estimated $35.5 million in costs, compared with $66,193 to vaccinate the same number of children through VFC. Those costs are only growing – the outbreak in South Carolina has not yet abated.”