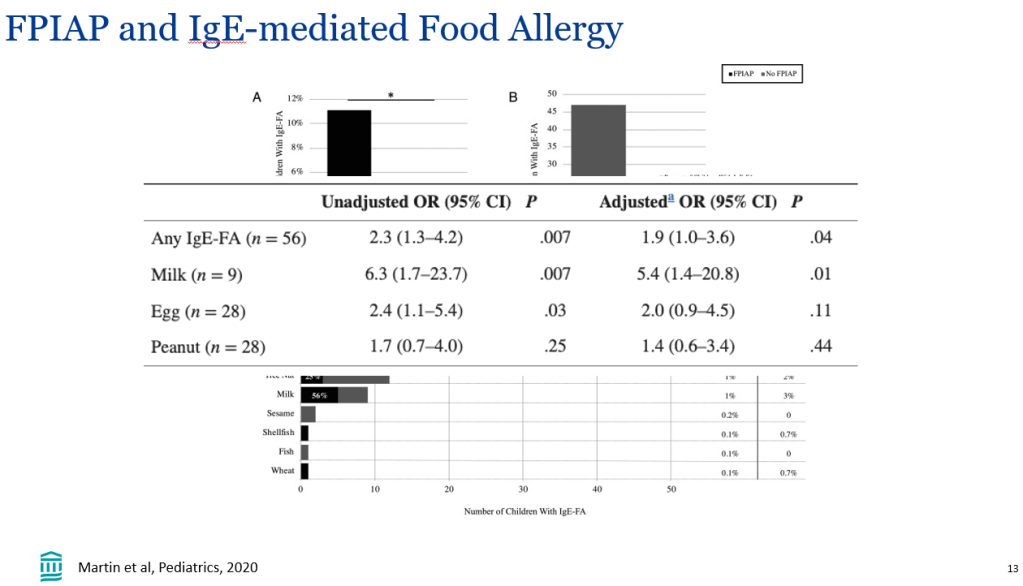
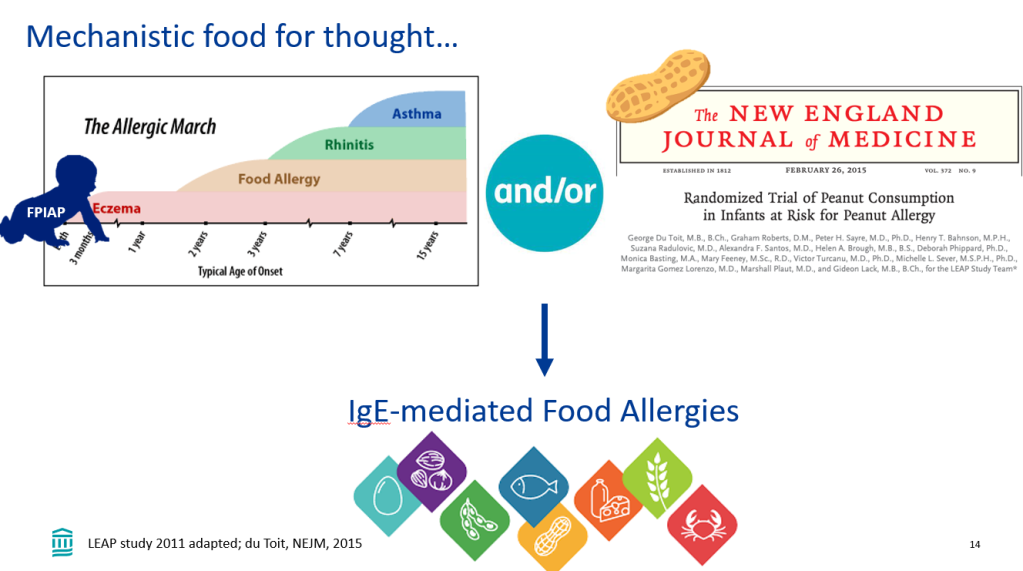
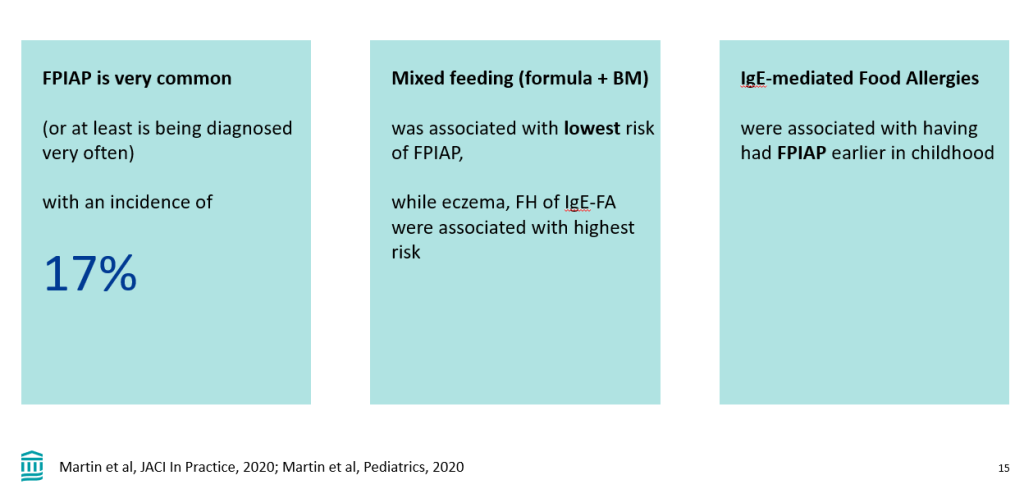
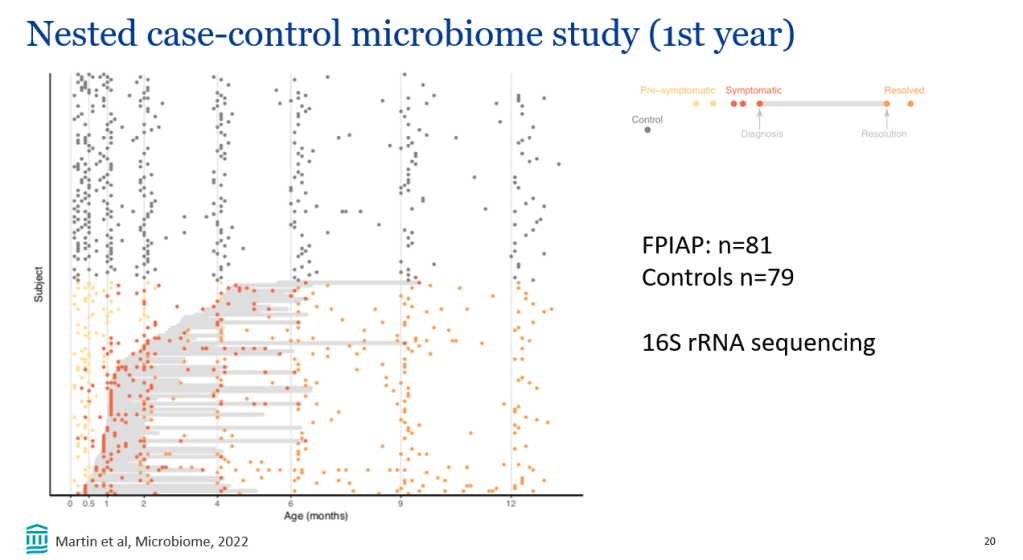
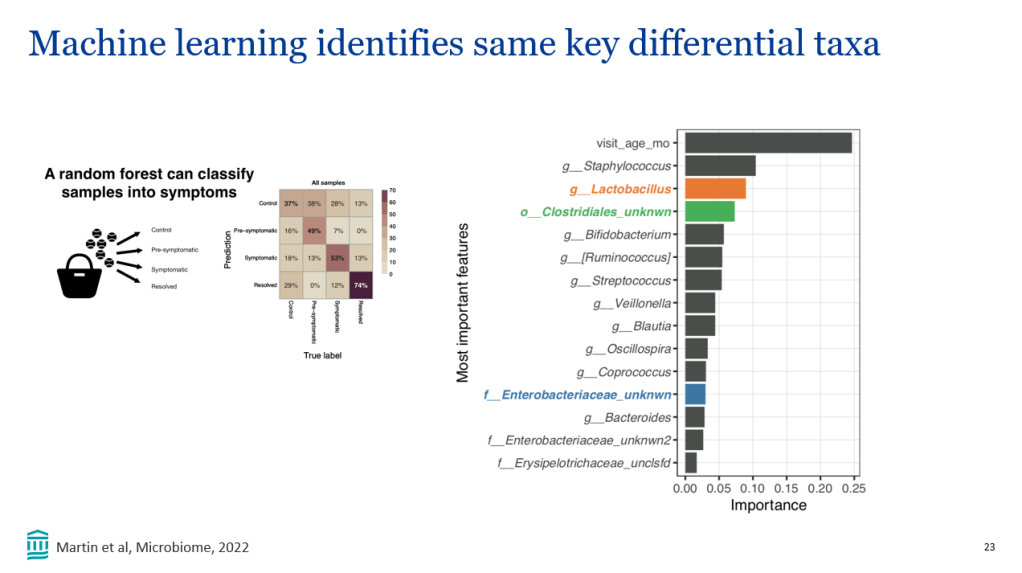
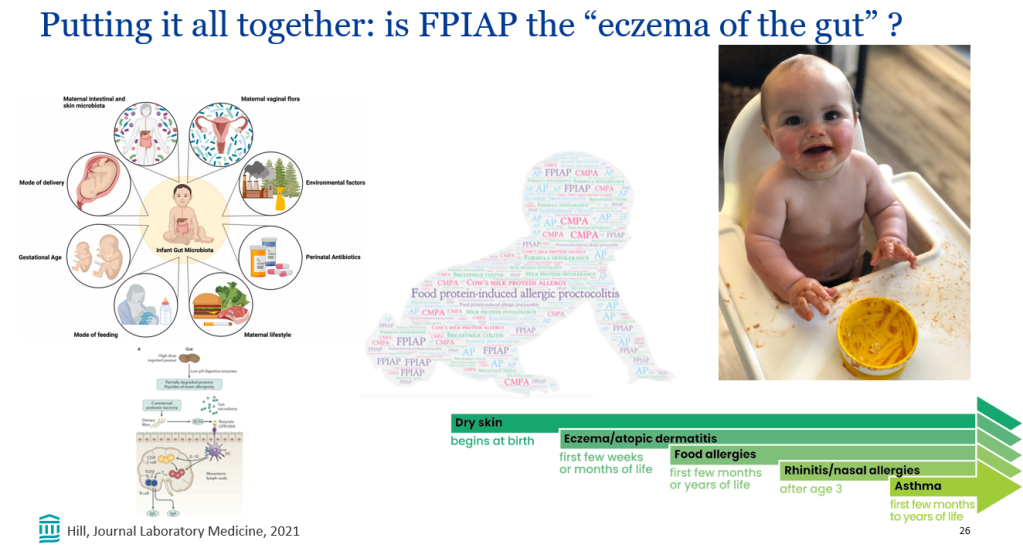
Recently, Dr. Victoria Martin gave our group an excellent update on cow’s milk protein allergy/food protein-induced allergic proctocolitis (FPIAP). My notes below may contain errors in transcription and in omission. Along with my notes, I have included many of her slides.


- iMAP guidelines for FPIAP were published in 2019. 2023-2024 guidelines have now been published
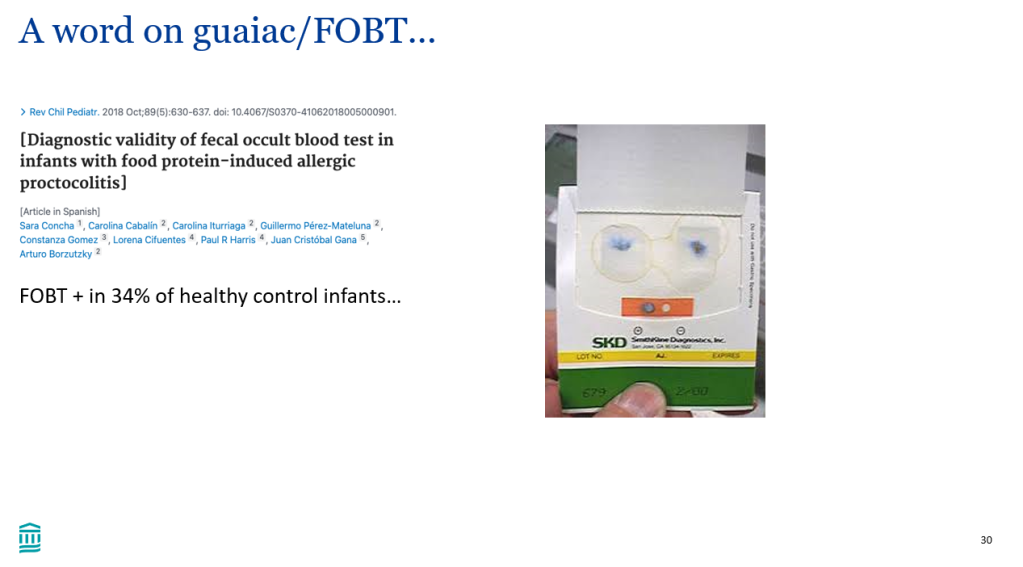
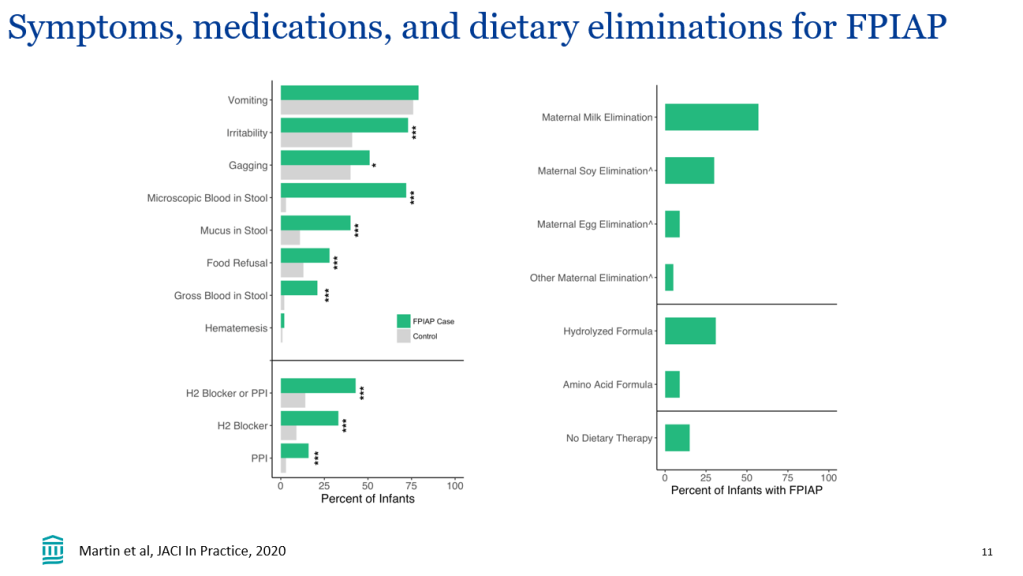
- Guaiac testing in infants is usually not helpful (in the absence of visible blood). In one study, 34% of healthy infant controls tested positive
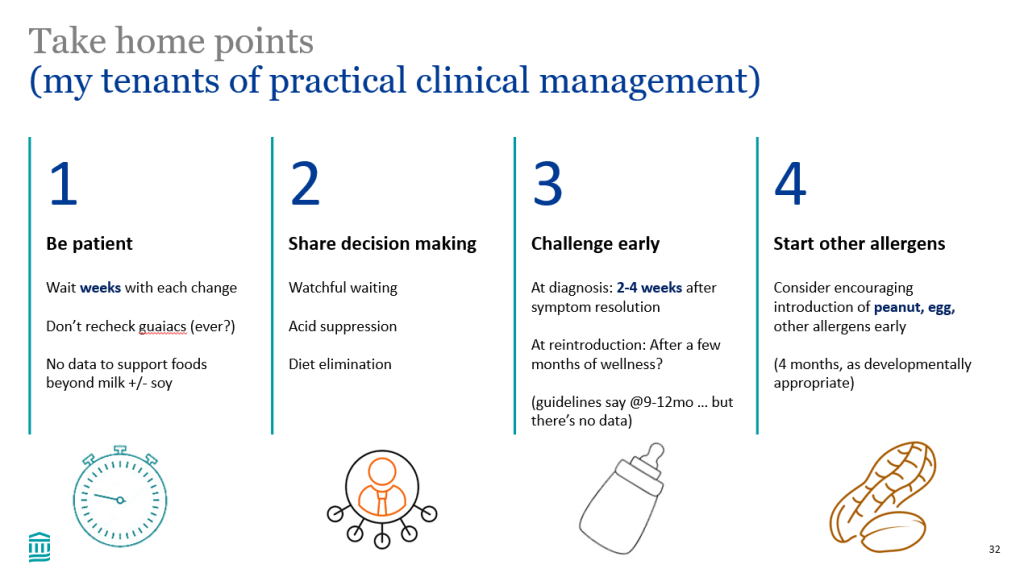
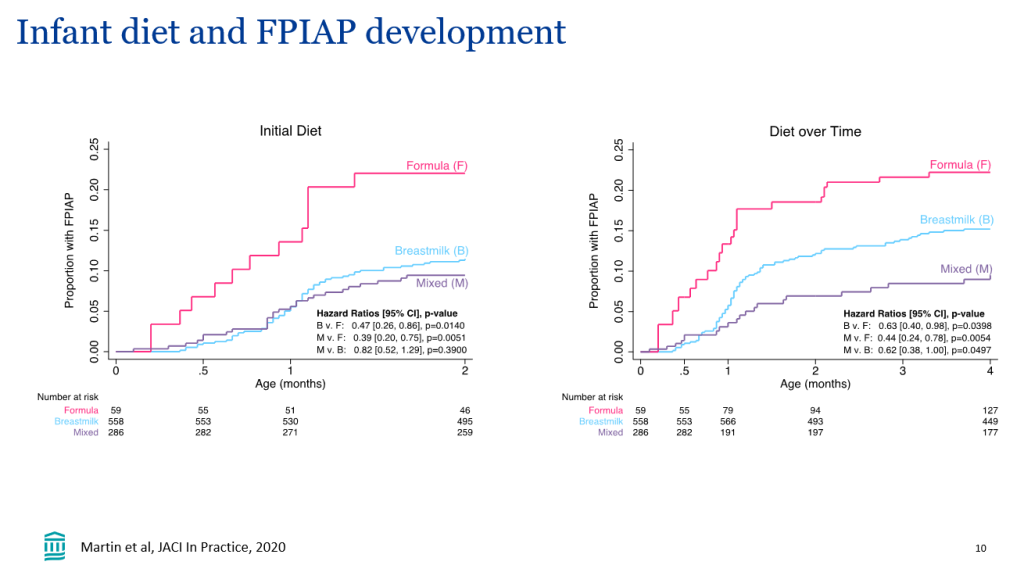
- Clinical management: when restricting a food, it may take 2-4 weeks to determine if it is helping. In breastfed infants, there is not data supporting restrictions beyond cow’s milk and soy. If other foods are eliminated, foods that were previously eliminated could be reintroduced
- In families who selected watchful waiting rather than dietary elimination, symptom resolution was similar in GMAP cohort. This was an observational study and findings could be influenced by selection bias
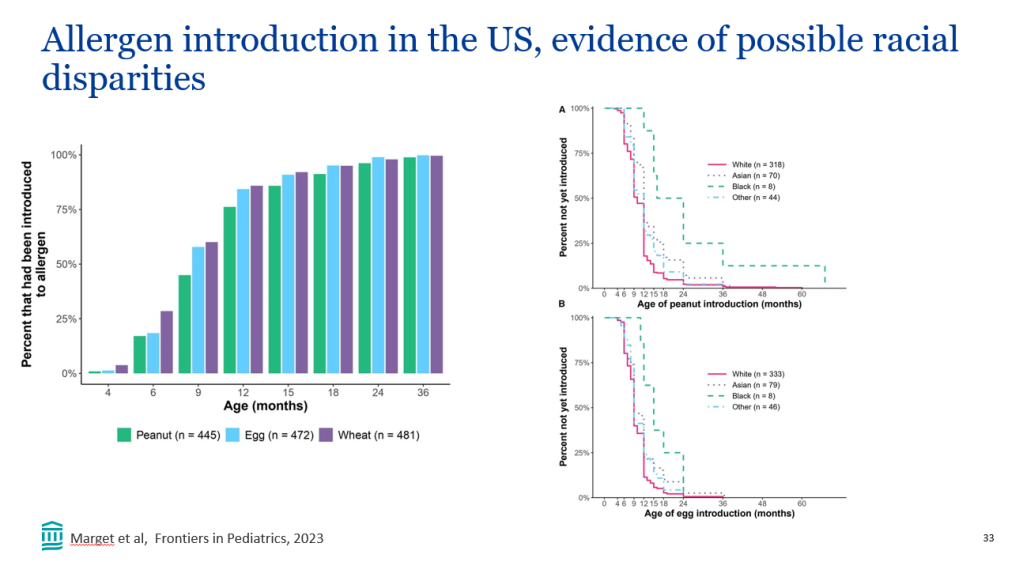
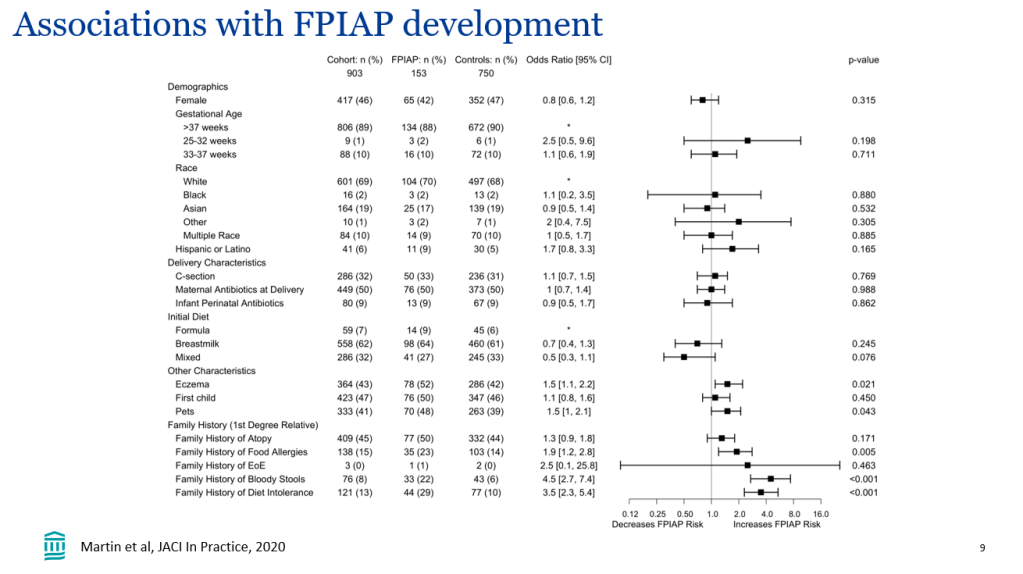
- In the GMAP cohort, there was a disparity in allergen introduction among different ethnicities (Ref: M Marget et al. Frontiers in Pediatrics; 2023: https://doi.org/10.3389/fped.2023.1207680. Open Access! Factors influencing age of common allergen introduction in early childhood). Compared to White children, Black children were less likely to have been introduced to peanut and egg, and Asian children were less likely to have been introduced to peanut as early (P < 0.05).
- In families concerned about food challenges, the families could challenge near a medical setting (eg. ER); however, anaphylactic reactions could occur after the first dose
- Challenging patients with FPIAP to establish diagnosis is generally recommended 2-4 weeks after resolution. In some patients, the FPIAP may have resolved and in some the diagnosis of FPIAP may be inaccurate. In those with more severe symptoms (eg required hospitalization), challenging at a later timeframe should be considered
- Probiotics: no clear role in their use for FPIAP at this time

Related blog posts:
- Good Bowel Sound Podcasts: Cow’s Milk Intolerance and Hirschspurng Disease
- ESPGHAN Guidance for Suspected Cow’s Milk Allergy in Infants
- Current Approach for FPIES (2023)
- Allergy Prevention Strategy: Early Exposure Beneficial (Long-term Data for Peanuts)
- Overdiagnosis of Milk Allergy in Infancy and New Consensus Recommendations
- Overdiagnosis of Cow’s Milk Protein Allergy in Infants and Formula Industry Influence
- Best of Allergy Articles 2021 -Cow’s Milk Allergy/Allergic proctocolitis (Part 4)
- Best Allergy Articles 2021 -Cow’s Milk Allergy (Part 3)
- Best Allergy Articles 2021 -Cow’s Milk Allergy (Part 2)
- Best Allergy Articles 2021-Cow’s Milk Allergy