At a recent dinner, we had the opportunity to hear a review of some of the recent data on ustekinumab. These notes may include some errors in transcription and errors of omission. Most of this data is derived from a recent publication that has been summarized in this blog: Landmark Publication for Ustekinumab (Stelara) | gutsandgrowth
Some key points:
- In numerous studies of biologic agents, longer duration of disease is associated with a much lower response to therapy. For the UNITI-1/UNTI-2 studies, the patients enrolled had long duration of disease (~10 years in UNIT1-1, ~8 years in UNITI-2)
- Recent data indicate that vedolizumab is effective for Crohn’s disease, but works slowly. It likely takes ~6 months to determine if it is working.
- With ustekinumab, most patients respond within 3 weeks of the induction dose; however, among nonresponders, many responded after the first maintenance dose. Thus, probably need to give at least the induction dose and the first maintenance dose for determining whether ustekinumab is effective.
- Overall safety profile looks very good for ustekinumab. More than 4000 patients in a psoriasis registry showed no serious safety signals (though psoriasis patients receive a lower dose).
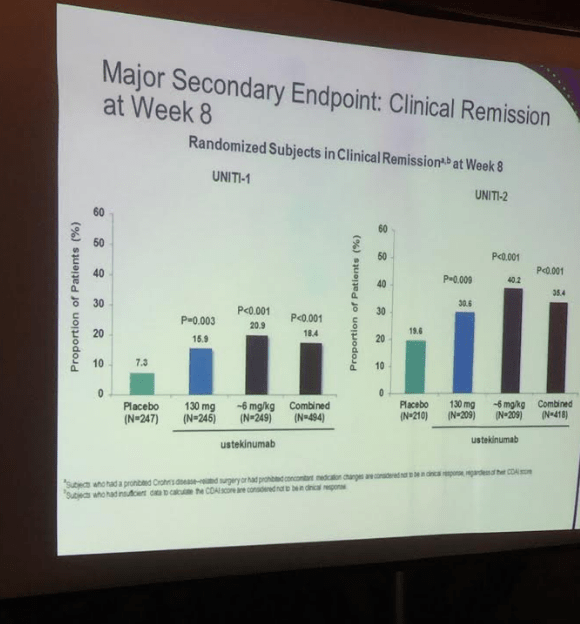
- Overall, the 6 mg/kg induction dose outperformed the 130 mg dose with regard to objective measures.
- High placebo rate in the IM-UNITI study likely is due to some residual response to the initial induction dose.
- Every 8 week dosing for ustekinumab was more effective than every 12 week dosing in these studies, though, there are likely patients who need more frequent and some who could benefit from less frequent dosing.
- 2.3% of patients in IBD studies had detectable antibody to ustekinumab but this did not preclude efficacy.
- ~20% of IBD patients do not develop increased CRP
- Pediatric studies of ustekinumab are ongoing testing different dosing regimens.
- One other anecdote in regards to magical thinking about which premedications are most effective: A man with a ridiculous hat was approached as he walked in an Atlanta.suburb. A lady asked him why he wore such an unusual hat. He replied that “the hat keeps elephants far away.” The lady said, “there are no elephants around here.” He said, “See it is working.”