RI Rusu et al. Clin Gastroenterol Hepatol 2026; 24: 365-374. Open Access! The Optimal Duration of pH Monitoring: Testing the Validity of Lyon 2.0 Recommendations for Wireless pH Measurement
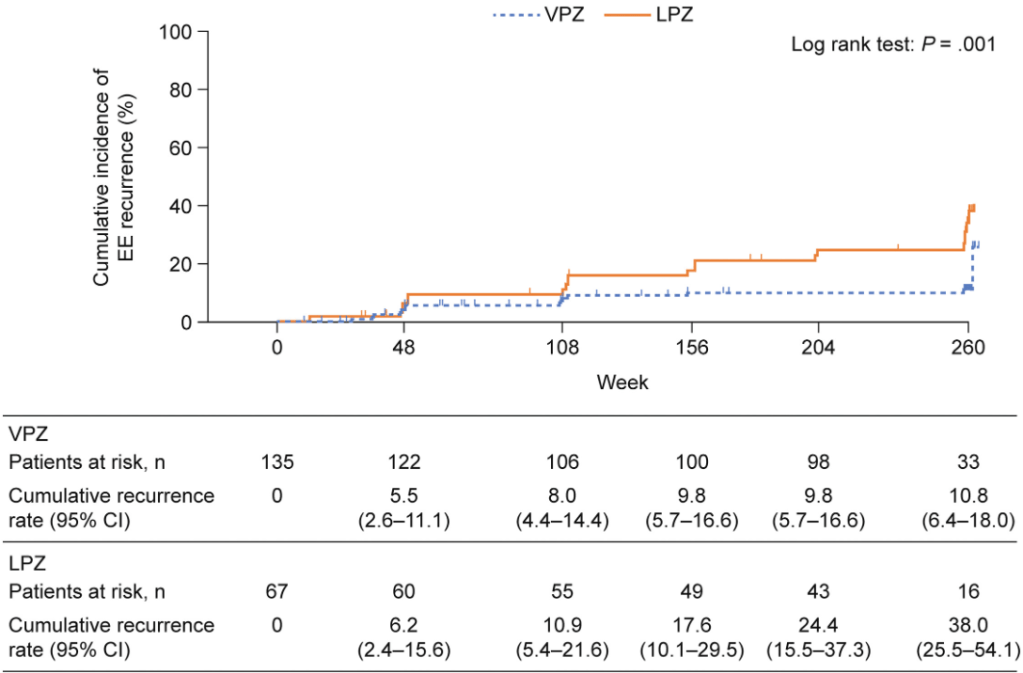
Background: “The Lyon 2.0 consensus recommends 96-hour wireless pH studies for gastroesophageal reflux disease (GERD) diagnosis…Ninety-six-hour monitoring is not always possible, either for technical reasons (eg, early detachment of the pH sensor) or for practical and financial reasons (48-hour studies are routine and cost-efficient in many centers).”
Methods: Data from 944 patients (16-85 years) with 4-day recordings (Bravo capsule) was reviewed. Patients were classified at 24, 48, and 72 hours against the 96-hour reference standard. Acid exposure time (AET) <4% was conclusively negative, and AET >6% was conclusively positive for GERD.
Key findings:
- With longer duration, more patients were able to be diagnosed with GERD and fewer patients were in the indeterminate group. The proportion of patients with inconclusive results (AET 4%–6%) reduced from 113 of 944 at 24 hours to 40 of 113 at 96 hours (35% of subgroup; P = .02)
- Only 3 of 60 patients with LA grade B esophagitis demonstrated physiologic reflux burden across all 96 hours of recording (which indicates that for significant reflux esophagitis, pH monitoring is not needed in most cases)
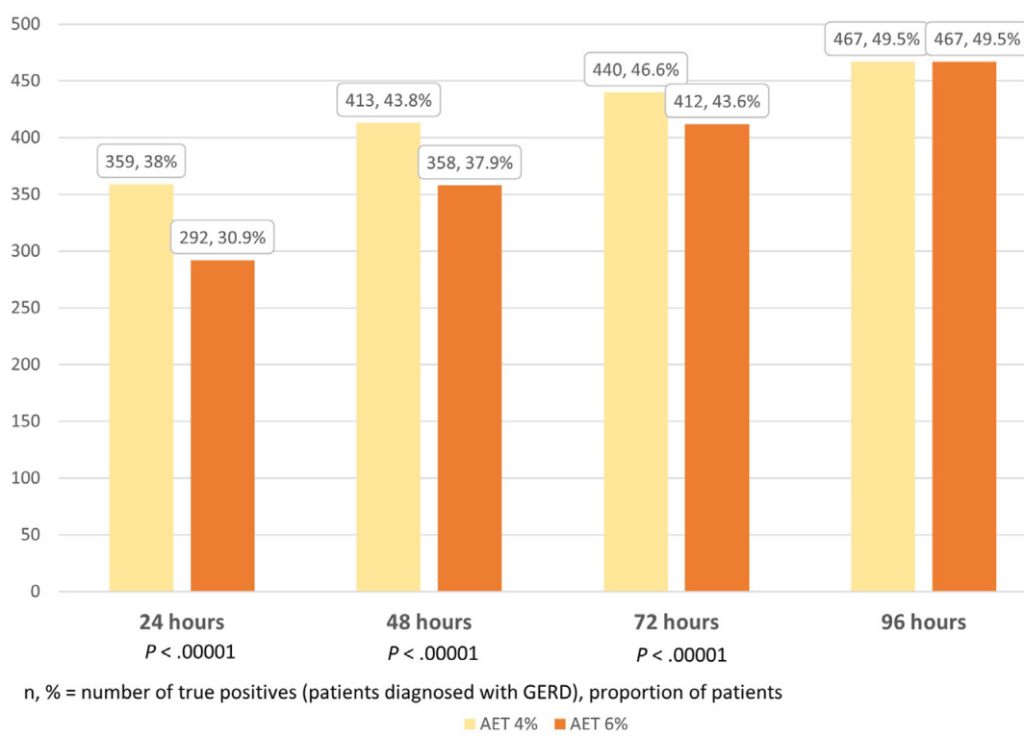
- In the associated editorial (pg 306-308), the authors note that “the minimum duration for an effective study seems to be 3 days, because ~90% of conclusively positive studies identified over 4 days would be detected with 72 hours of recording. However, to achieve this, a 4-day study would need to be planned because of potential data loss, reported in almost a third of the cases in this and prior cohorts.”
- “96-hour wireless monitoring is optimal for ruling out GERD when the pretest likelihood of reflux is low, especially when empiric PPI therapy is ineffective.”
My take: pH probe tests have many limitations. This study reinforces the need for longer studies in many patients when the findings would be equivocal with shorter duration studies.
Related blog post:
- #5000: Simple Rule for Deciding When pH Impedance Testing Should Be Done While on Therapy
- Gastroesophageal Reflux Phenotypes and “Where Rome, Lyon, and Montreal Meet”
- How helpful is a pH-Impedance Study in Identifying Reflux-Induced Symptoms?
- What’s Going On in Patients with Reflux Who Fail to Respond to PPIs?
- How Many Kids with Reflux Actually Have Reflux?
- How to Make a Study Look Favorable to Surgery for Reflux over Medical Therapy
- Why didn’t patient with documented reflux get better with PPI …
- Failure of PPI test | gutsandgrowth
- Guidelines on Functional Heartburn
- Better to do a coin toss than an ENT exam to determine reflux
- How Likely is Reflux in Infants with “Reflux-like … – gutsandgrowth