In June 2024 (special issue), Gastroenterology published an entire issue (193 pages) focused on celiac disease. There was a lot of useful information on almost every aspect of this disease. Below I have summarized some of the points.



F Zingone et al. Open Access: Celiac Disease–Related Conditions: Who to Test?
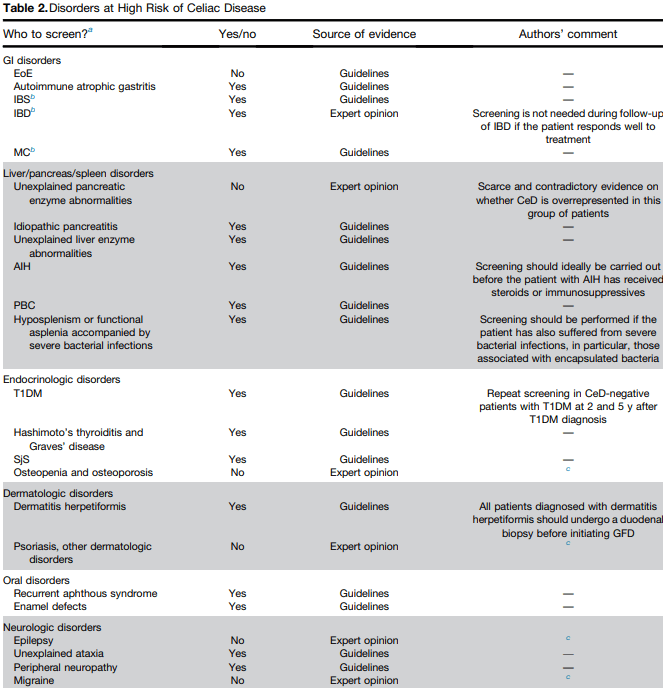
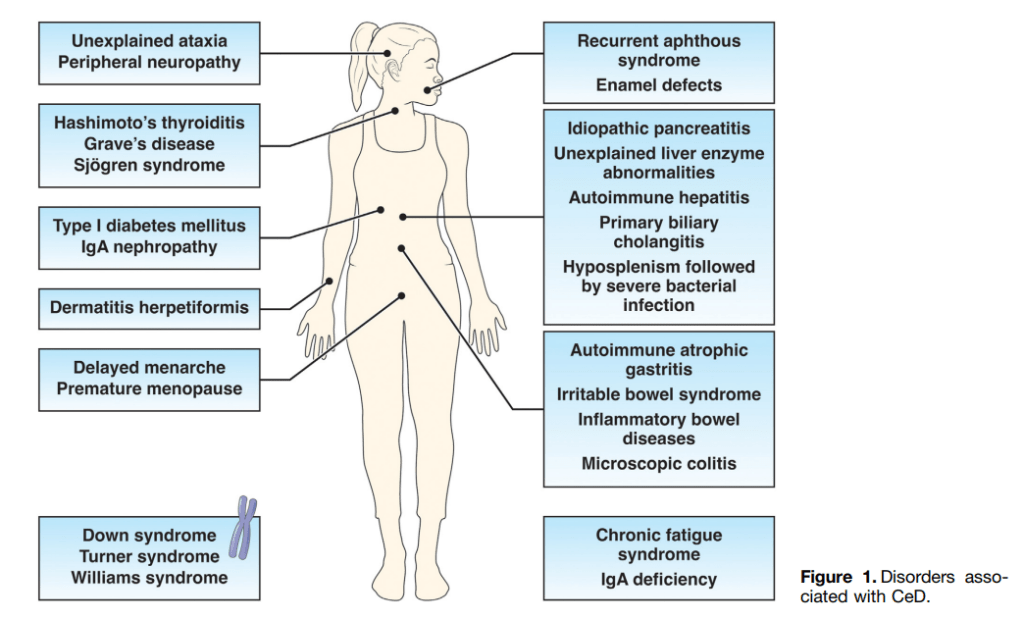
- The authors detail disorders with increased risk for CeD and which merit screening (see Table 2 below). Some disorders that merit screening that are more obscure include idiopathic pancreatitis, autoimmune hepatitis, delayed menarche, and chronic fatigue.
- They note that type 1 diabetes mellitus could require serial screening. “Because CeD can manifest at any time and with greater frequency during the initial 5 years, conducting additional screenings in CeD-negative T1DM patients 2 and 5 years after T1DM diagnosis and those who later develop gastrointestinal (GI) symptoms may be advisable.” In addition, it is important to recognize that CeD serology testing is less reliable in patients with T1DM.

S Gatti et al. Patient and Community Health Global Burden in a World With More Celiac Disease, describes the worldwide burden of celiac disease and how to improve detection.
- They note that the worldwide prevalence is between 0.7% and 2.9%. In this issue, most authors estimate the prevalence to be about 1% with more than 50% undetected.
- There are many places with higher rates. In U.S. “children in Colorado had a 2.5-fold higher risk compared to Washington State…similar regional differences were seen …in Sweden, Finland, and Germany.”
- They note the burden before and after diagnosis. Before diagnosis/undetected, there can be persistent symptoms, complications (eg. osteoporosis, decreased fertility) and impaired quality of life. Afterwards, there are increased costs of a GFD and psycho-social burden of GFD.
- In terms of generalized screening compared to case-finding, the authors note that given the number of at-risk groups, the case-finding approach could entail screening >50% of the population.
V Abadie et al. New Insights on Genes, Gluten, and Immunopathogenesis of Celiac Disease, reviews the intricate details of genetic, biochemical, and immunologic studies, which together have revealed mechanisms of gluten peptide modification and HLA binding, thereby enabling a maladapted anti-gluten immune response.
- What I was most interested in was the mechanisms behind ‘potential’ celiac disease (PCeD) in which patients have autoimmunity (+serology) but normal histology. In potential CeD, the anti-CD4+ T-cell response is present but decoupled from tissue cytotoxicity. However, notably, IL-21, a cytokine produced by gluten-specific CD4+ T cells in active CeD, is not up-regulated in potential CeD…In addition, patients with potential CeD lack the presence of an epithelial stress response associated with IL-15, HSP70, and HSP27 upregulation in epithelial cells.” “The presence of epithelial stress is a crucial prerequisite for the development of tissue damage.”



