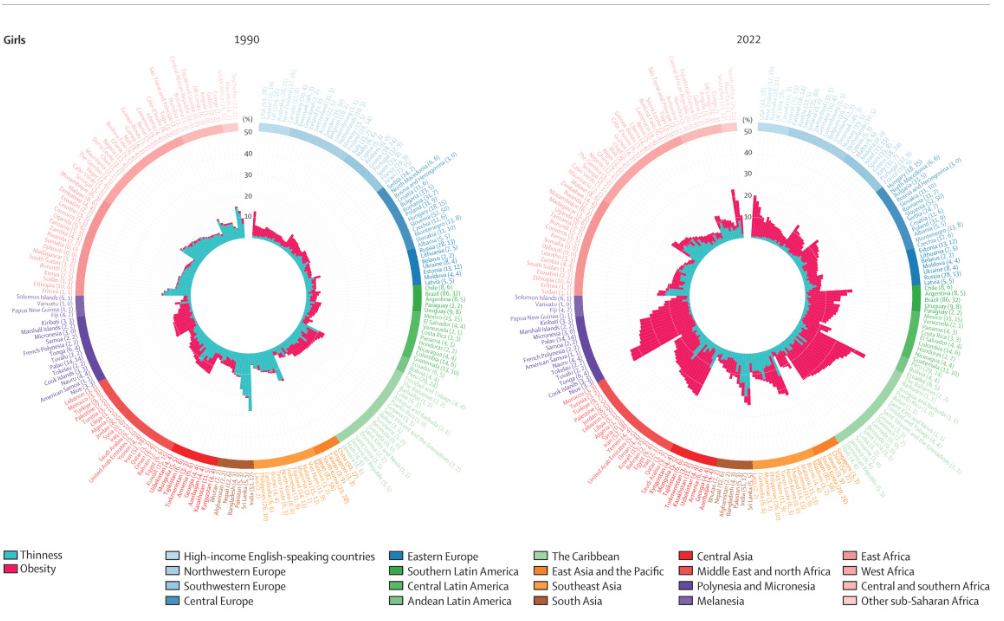
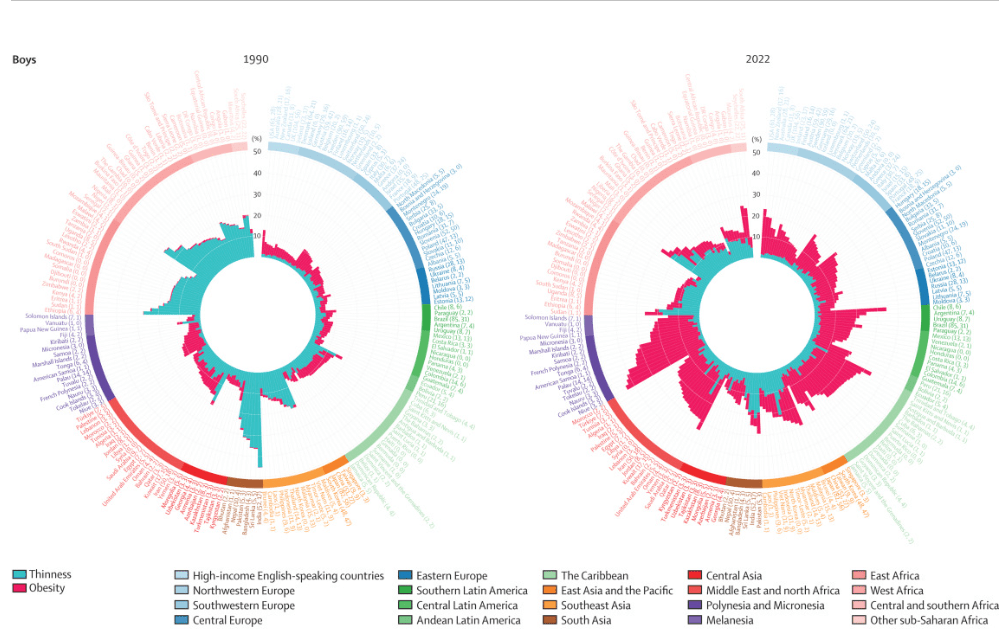
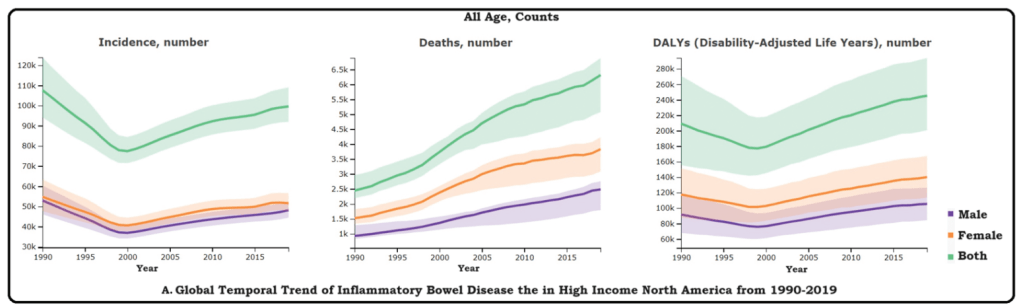
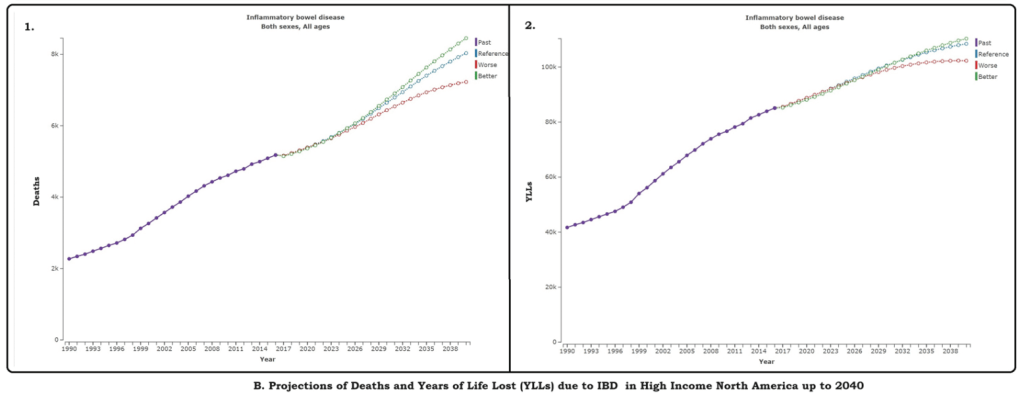
A recent issue of Clinical Gastroenterology and Hepatology focused solely on the clinical features and management of inflammatory bowel disease. Even for those with expertise in IBD, there is a lot of useful information and concise reviews of what is known.
Here are some of my notes from this issue:
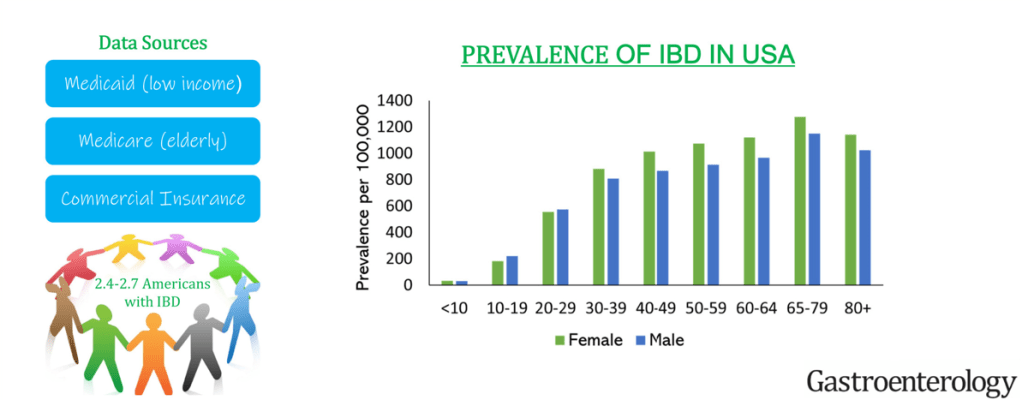
AN Ananthakrishnan et al. Clin Gastroenterol Hepatol 2020; 18: 1252-60. Changing Global Epidemiology of Inflammatory Bowel Diseases: Sustaining Health Care Delivery Into the 21st Century
Reviews risk factors and recommends the following as ways to lower risk of developing IBD for at-risk individuals:
- Breastfeeding in infancy
- Do not start smoking
- Avoid vitamin D deficiency
- Minimize non-steroidal anti-inflammatory drug use
- Minimize antibiotic use especially for young children and during pregnancy
- Encourage moderate physical activity, healthy weight, low stress and regular sleep
- Diet high in fruit, vegetables, fiber, and fish
Reviews the epidemiology and notes that there has been a evidence of a decline in incidence in IBD in (at least) the Western world; however, because of compounding prevalence, it is expected that the number of individuals with IBD will continue to rise. In Canada, for example, it is expected that the prevalence will rise from 0.7% in 2018 to 1% by 2030.
In newly industrialized countries, it is expected that rising incidence is going to substantially increase the global disease burden. The authors note the following as areas needed in research and clinical care to meet global IBD care burden:
- tools for early diagnosis
- early effective intervention to prevent irreversible bowel damage
- precision medicine to select the right treatment for the right patient
- need for less costly and more safe therapies
- simple tools to monitor disease activity
- primary disease prevention strategies, especially for those at high risk
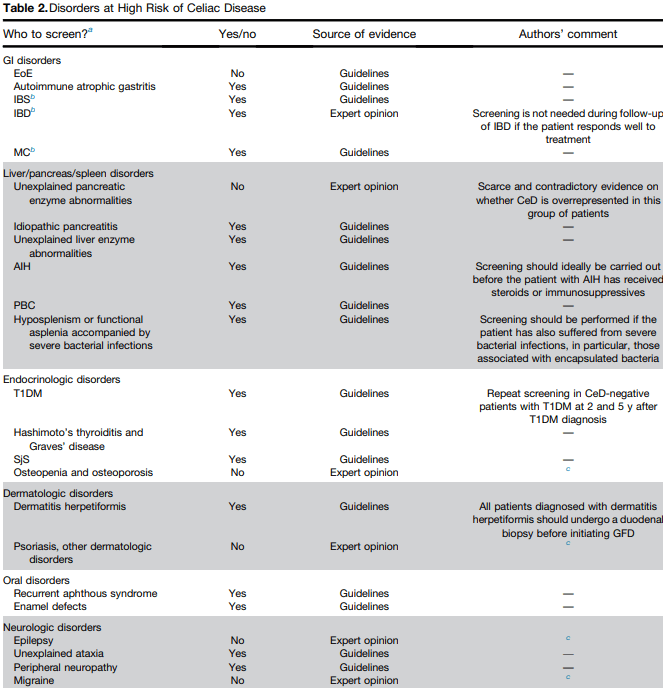
CA Siegel, CN Bernstein. Clin Gastroenterol Hepatol 2020; 18: 1261-7. Identifying Patients With Inflammatory Bowel Diseases at High vs Low Risk of Complications
This article’s disease-stratification information overlaps with subsequent articles which detail the positioning of therapies for Crohn’s disease (CD) and ulcerative colitis (UC) respectively.
NH Nguyen, S Singh, WJ Sandborn. Clin Gastroenterol Hepatol 2020; 18: 1267-79. Positioning Therapies in the Management of Crohn’s Disease.
Some of the information summarized in this article:
Table 2 -Comparative Efficacy of Biologics for Moderate to Severe Active Crohn’s Disease (CD):
- Infliximab: For induction: OR compared to placebo for remission: 5.90 (2.78-12.51); probability of remission 60%. For maintenance in those with clinical response: probability of remission SUCRA ranking: 48%; 0.68
- Adalimumab: For induction: OR compared to placebo for remission: 3.80 (1.76-8.18); probability of remission 49%. For maintenance in those with clinical response: probability of remission SUCRA ranking: 58%; 0.97
- Ustekinumab: For induction: OR compared to placebo for remission: 2.75 (1.76-4.32); probability of remission 41%. For maintenance in those with clinical response: probability of remission SUCRA ranking: 39%; 0.36
- Vedolizumab: For induction: OR compared to placebo for remission: 2.69 (1.36-5.32); probability of remission 40%. For maintenance in those with clinical response: probability of remission SUCRA ranking: 42%; 0.52
- Certolizumab pegol: For induction: OR compared to placebo for remission: 1.36 (0.89-2.08); probability of remission 25%. For maintenance in those with clinical response: probability of remission SUCRA ranking: 42%; 0.48
In deciding therapy, the authors specify factors that help classify as high-risk CD Table1):
- Structural damage: large or deep mucosal lesions, fistula or perianal abscess, prior resections (especially if >40 cm)
- Inflammatory burden: extensive disease involvement (ileal disease >40 cm or pancolitis), increased C-reactive protein, low albumin
- Impact on quality of life: presence of stoma, >10 loose stools/week, lack of symptomatic improvement with prior biologics and/or immunomodulators, presence of anorectal symptoms, anemia, daily abdominal pain
- Emerging predictors: antimicrobial antibody pattern, antimicrobial genetic peptide signature
Though the authors note a lack of adequate head-to-head comparative studies, they make some recommendations for treatment:
- For severe disease, they suggest first-line therapy for CD would be infliximab or adalimumab in combination therapy regimen (with infliximab favored for higher disease severity)
- For second-line therapy, they suggest ustekinumab for most patients in combination therapy or 2nd anti-TNF in those with loss or response due to immunogenicity or intolerance
- For those with higher risk factors for adverse events (or preference) and moderate disease severity, the authors recommend vedolizumab as 1st line and ustekinumab as 2nd line. For this same group with higher disease severity, they suggest ustekinumab as 1st line treatment.
Other key points:
- In terms of risk of malignancy, the authors note that in a comprehensive systematic review of 23 RCTs of TNF-alpha antagonists in IBD, there was NO significant increase in the risk of malignancy with TNF-alpha antagonists.
- In terms of combination therapy, the authors note that their has been an observed benefit which is “at least partly attributed to achieving a higher biologic trough concentration….no differences in efficacy of combination therapy vs infliximab were observed when evaluating patients by quartiles of infliximab trough concentration; however, currently this represents association rather than causation, and it is possible that superior remission rates drove higher trough concentrations, rather than vice versa.”

Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.