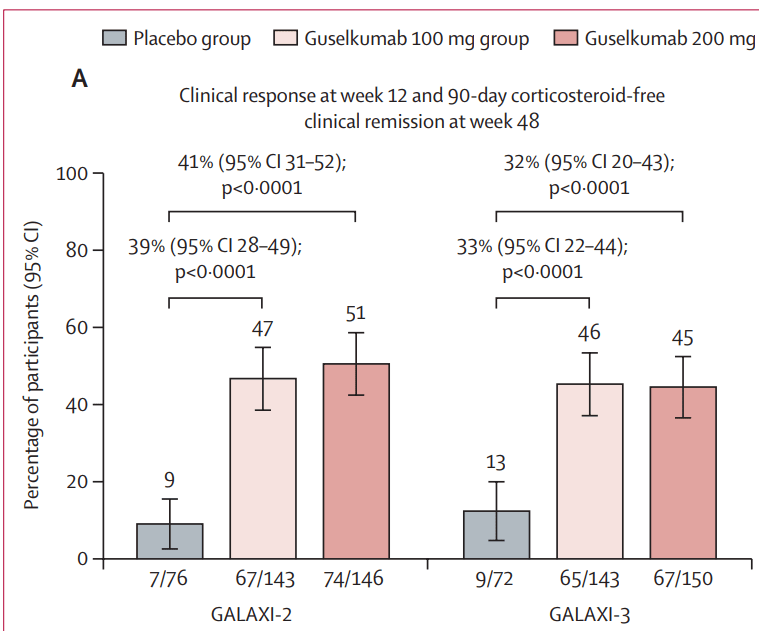
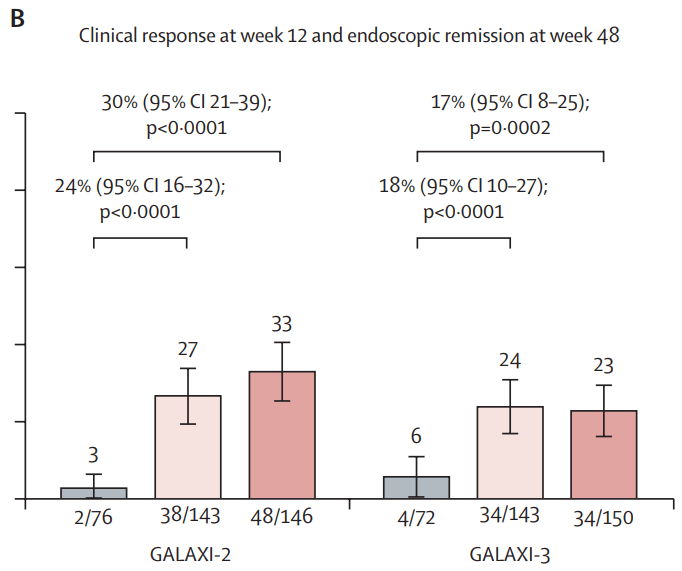
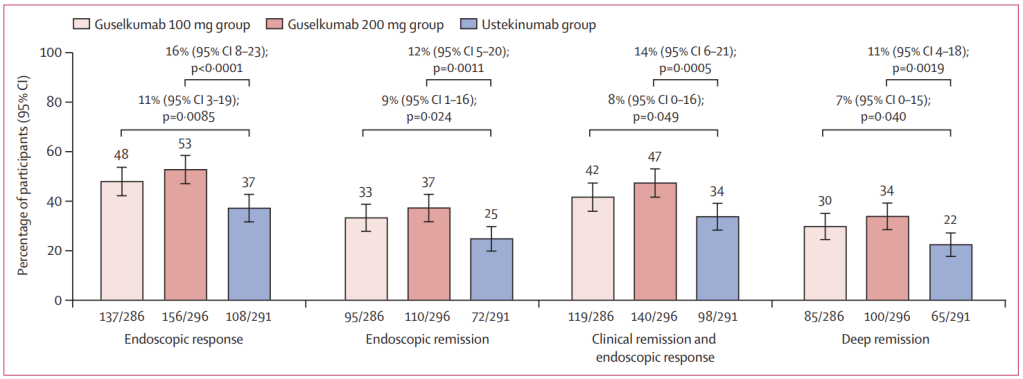
Q Aziz et al. Clinical Gastroenterology and Hepatology, Volume 23, Issue 8, 1291 – 1302. Open Access! AGA Clinical Practice Update on GI Manifestations and Autonomic or Immune Dysfunction in Hypermobile Ehlers-Danlos Syndrome: Expert Review
This review and practice update includes 16 “best practice advice” statements. Here are nine of them:
- #1: Clinicians should be aware of the observed associations between hEDS or HSDs and POTS and/or MCAS and their overlapping gastrointestinal (GI) manifestations; while theoretical explanations exist, experimental evidence of the biological mechanisms that explain relationships is limited and evolving.
- #2: Testing for POTS/MCAS should be targeted to patients presenting with clinical manifestations of POTS/MCAS, but universal testing for POTS/MCAS in all patients with hEDS/HSDs is not supported by the current evidence.
- #3: Gastroenterologists seeing patients with DGBI should inquire about joint hypermobility and strongly consider incorporating the Beighton score for assessing joint hypermobility into their practice as a screening tool; if the screen is positive, gastroenterologists may consider applying 2017 diagnostic criteria to diagnose hEDS (https://www.ehlers-danlos.com/wp-content/uploads/2017/05/hEDS-Dx-Criteria-checklist-1.pdf) or offer appropriate referral to a specialist where resources are available.
- #4: Testing for POTS through postural vital signs (eg, symptomatic increase in heart rate of 30 beats/min [40 beats/min for 12-19 yo] or more with 10 minutes of standing during an active stand or head-up tilt table test in the absence of orthostasis) and referral to specialty practices (eg, cardiology or neurology) for autonomic testing should be considered in patients with hEDS/HSDs and refractory GI symptoms who also report orthostatic intolerance after exclusion of medication side effects and appropriate lifestyle or behavioral modifications (eg, adequate hydration and physical exercise) have been attempted but is not required for all patients with hEDS/HSDs who report GI symptoms alone.
- #5: In patients presenting to gastroenterology providers, testing for mast cell disorders including MCAS should be considered in patients with hEDS/HSDs and DGBI who also present with episodic symptoms that suggest a more generalized mast cell disorder (eg, visceral and somatic pain, pruritus, flushing, sweating, urticaria, angioedema, wheezing, tachycardia, abdominal cramping, vomiting, nausea, diarrhea, urogynecological and neurological complaints) involving 2 or more physiological systems (eg, cutaneous, GI, cardiac, respiratory, and neuropsychiatric), but current data do not support the use of these tests for routine evaluation of GI symptoms in all patients with hEDS/HSDs without clinical or laboratory evidence of a primary or secondary mast cell disorder.
- #6: If MCAS is suspected, diagnostic testing with serum tryptase levels collected at baseline and 1–4 hours following symptom flares may be considered by the gastroenterologist; increases of 20% above baseline plus 2 ng/mL are necessary to demonstrate evidence of mast cell activation.
- #12: Medical management of GI symptoms in hEDS/HSDs and POTS/MCAS should focus on treating the most prominent GI symptoms and abnormal GI function test results. In addition to general DGBIs and GI motility disorder treatment, management should also include treating any symptoms attributable to POTS and/or MCAS.
- #13: Treatment of POTS may include increasing fluid and salt intake, exercise training, and use of compression garments. Special pharmacological treatments for volume expansion, heart rate control, and vasoconstriction with integrated care from multiple specialties (eg, cardiology, neurology) should be considered in patients who do not respond to conservative lifestyle measures.
- #14: When MCAS is suspected, patients can benefit from treatment with histamine receptor antagonists and/or mast cell stabilizers, in addition to avoiding triggers such as certain foods, alcohol, strong smells, temperature changes, mechanical stimuli (eg, friction), emotional distress (eg, pollen, mold), or specific medications (eg, opioids, nonsteroidal anti-inflammatory agents, iodinated contrast).
Background: “Clinical gastroenterologists are encountering an increasing number of patients with chronic GI symptoms who also appear to experience comorbid hEDS/HSDs, POTS, and/or MCAS.15,16 Recognizing and treating GI symptoms in patients with hEDS/HSDs and comorbid POTS or MCAS present major challenges for clinicians, who often feel under equipped to address their needs.”
The article provides guidance on measuring hypermobility (Beighton Scoring System), Diagnosis/classification of mast cell activation (Table 1) and treatments for these disorders (Table 2)







My take: This is a useful reference for the overlap of DGBIs with hypermobile Ehlers-Danlos, POTS and Mast cell Activation. Nevertheless, the relationship between these disorders is unclear. In fact, there have been some studies indicating that joint mobility is not associated with an increase in functional GI disorders. Some of the association may be related to a surveillance bias.
Related blog posts:
- Joint Mobility –Not Associated with Increased Functional GI Disorders
- Antroduodenal Dysmotility in Hypermobility Disorders and Ehlers-Danlos Syndrome
- Dr. Carlo DiLorenzo: Advice for Managing DGBIs (Part 1)
- Dr. Carlo DiLorenzo: Advice for Managing DGBIs (Part 2)
- Dr. Neha Santucci: Management of DGBIs in the Post-Pandemic Era (Part 1)
- Craig Friesen: Understanding Food Allergies and Food Intolerance in DGBIs
- Myth or Fact: Joint Hypermobility is Related to Pediatric Functional Abdominal Pain & Dr. Roy Link
- “An Allergic Basis for Abdominal Pain”
- Outcome with POTS –Better than Expected?
- Mechanisms of Postinfectious Irritable Bowel Syndrome & Functional Disorders
- Dreaded Nausea (2017)