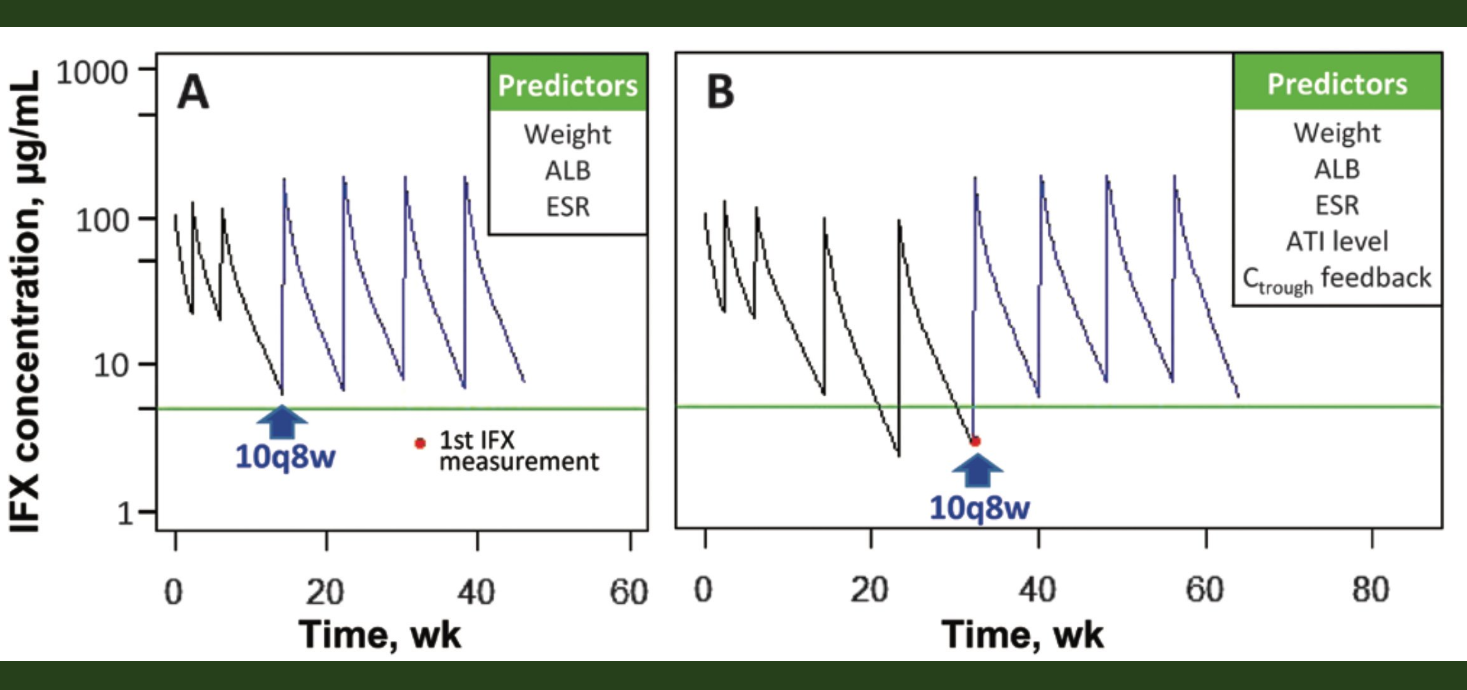
AS Cheifetz et al. Am J Gastroenterol 2021;116: 2014-2025. A Comprehensive Literature Review and Expert Consensus Statement on Therapeutic Drug Monitoring of Biologics in Inflammatory Bowel Disease (published online August 13, 2021)
Key recommendations:
- The panel agreed that reactive TDM should be used for all biologics for both primary nonresponse and secondary loss of response
- It was recommended that treatment discontinuation should not be considered for infliximab or adalimumab until a drug concentration of at least 10–15 mg/mL was achieved
- Consensus was also achieved regarding the utility of proactive TDM for anti–tumor necrosis factor therapy. It was recommended to perform proactive TDM after induction and at least once during maintenance.
- More data are needed with regard to proactive TDM for biologics other than anti-TNF agents
- There are no differences in interpreting TDM between originator biologics and biosimilars
- When considering switching within drug class in case of secondary loss of response to a first anti-TNF drug because of the development of antidrug antibodies, an immunomodulator should be added to a subsequent anti-TNF therapy
- Low-titer antidrug antibodies can be overcome by treatment optimization (dose escalation, dose interval shortening, and/or addition of an immunomodulator)

My take: This article should help support the practice of proactive TDM and discourage stopping anti-TNF agents until an adequate therapeutic level is achieved.
Related blog posts:
- Can Therapeutic Drug Monitoring with Monotherapy Achieve Similar Results as Combination Therapy?
- Real-World Experience with Proactive Therapeutic Drug Monitoring in Inflammatory Bowel Disease
- Here’s The Proof That Proactive Drug Monitoring Improves Outcomes in Children With Crohn’s Disease
- Combination Therapy Study Points to Central Role of Adequate Drug Levels | gutsandgrowth
- Proactive Therapeutic Drug Monitoring -Different Time Points | gutsandgrowth
- For the Next Insurance Appeal: Therapeutic Drug Monitoring in Adalimumab Treatment (Pediatrics) & Satire on Prior Authorizations | gutsandgrowth
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.