Before reviewing today’s article, I wanted to make a comment about the blog post on 12/17/23 (Endoscopy of the Ileal Pouch Anal Anastomosis) which was a JPGN topic of the month. The editorial staff encourages author-driven communication and author-driven initiatives for these types of articles. If you have a topic for JPGN, please send an email to the Section Editor Darla Shores (dshores1@jhmi.edu) or to the editor Sandeep Gupta. (skgupta@uabmc.edu). This includes articles that you would like to write (fellow/interested faculty with senior faculty, up to 5 authors, 1500 words, 12 references), or if you have a topic that you would like to see in JPGN but do not wish to write yourself, please inform the editorial team as well.
———
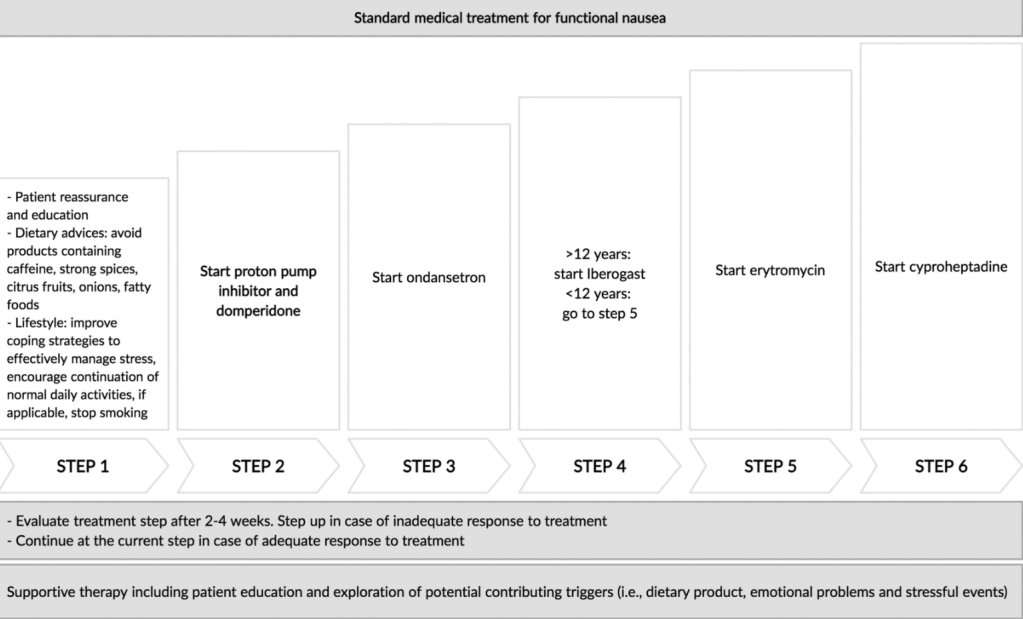
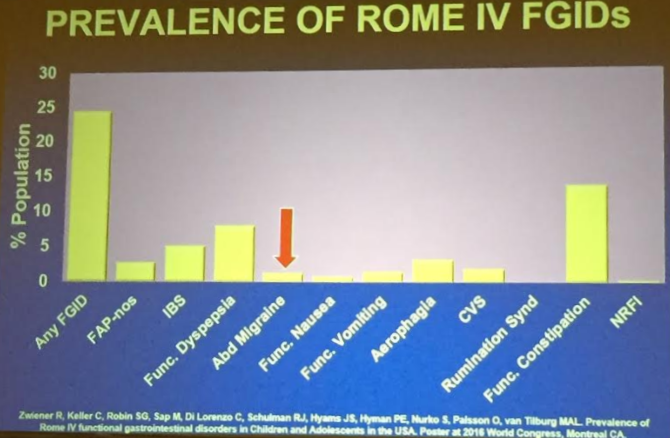
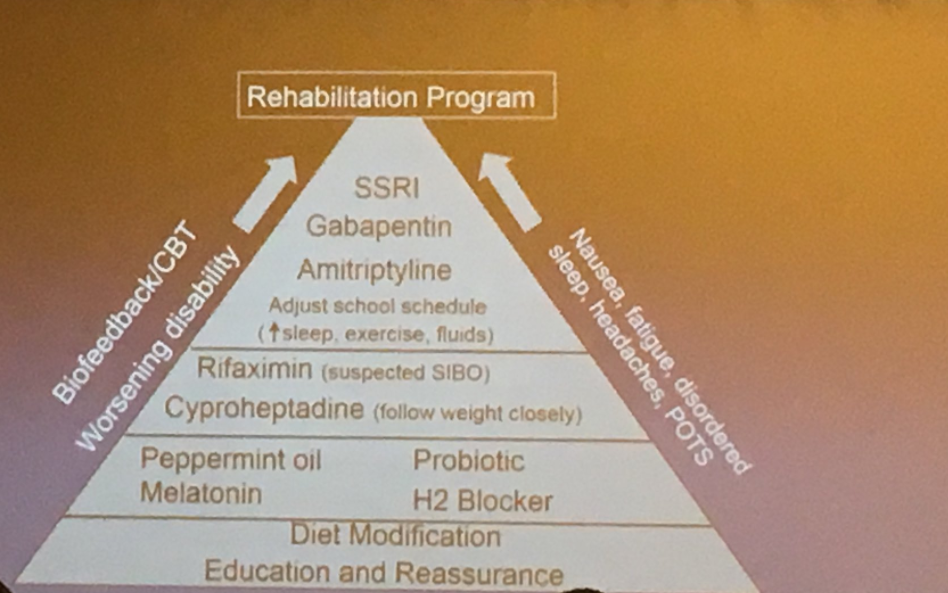
PT Osgood et al. JPGN 2023; 77: 726-733. Intrapyloric Botulinum Toxin Injection for Refractory Nausea and Vomiting in Pediatric Patients
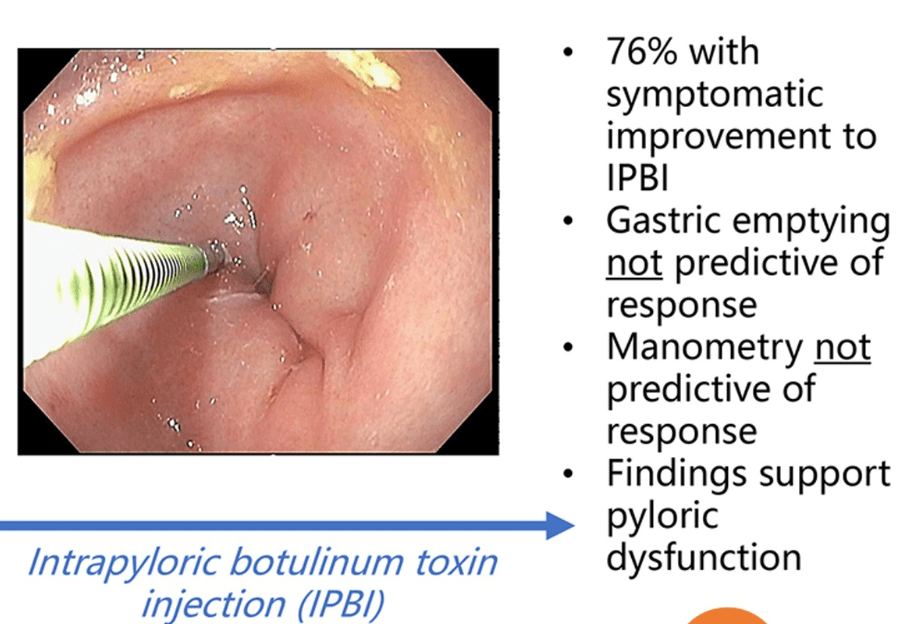
In this retrospective review, pediatric patients (n=25) received intrapyloric botox injections: (80-100 IU divided into 4 doses administered via sclerotherapy needle.
Key findings with botox injections:
- Of 22 patients completing a GE study, 14 had delayed GE with no significant difference between IPBI responders and nonresponders
- Improvement in vomiting in 80% (16/20), nausea 75% (15/20), abdominal pain 79% (15/19).
- In those with psychiatric diagnosis, improvement was seen 71%. In those with orthostatic intolerance, improvement was noted in 67%.
- In those with delayed GE, improvement was noted in 79% compared with 63% (5/8) with normal GE
My take: Botox was associated with improvement in this refractory pediatric group regardless of gastric emptying/manometry. This suggests that relaxation of pylorus is a useful therapeutic modality in a subset of patients.
Related blog posts: