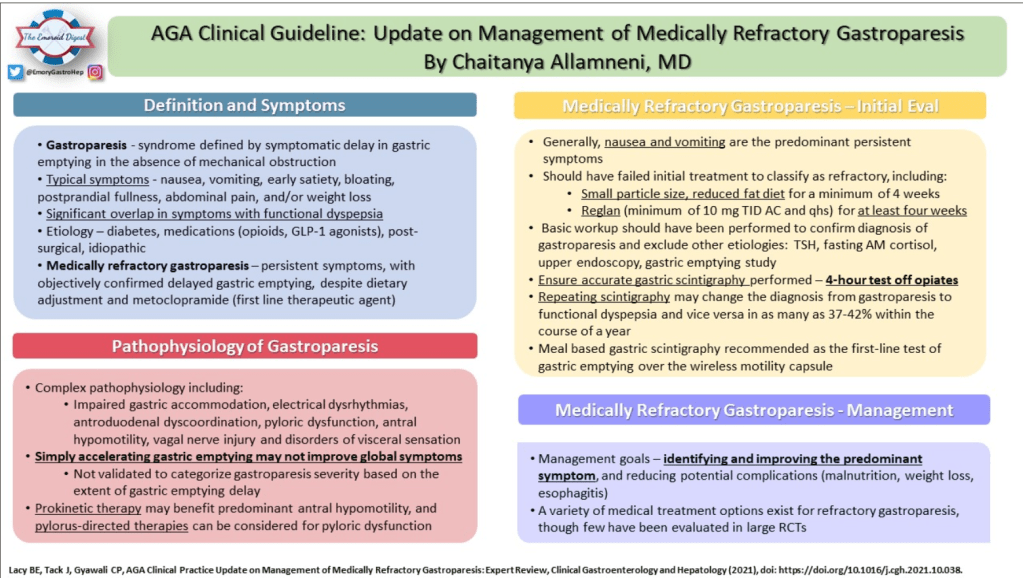
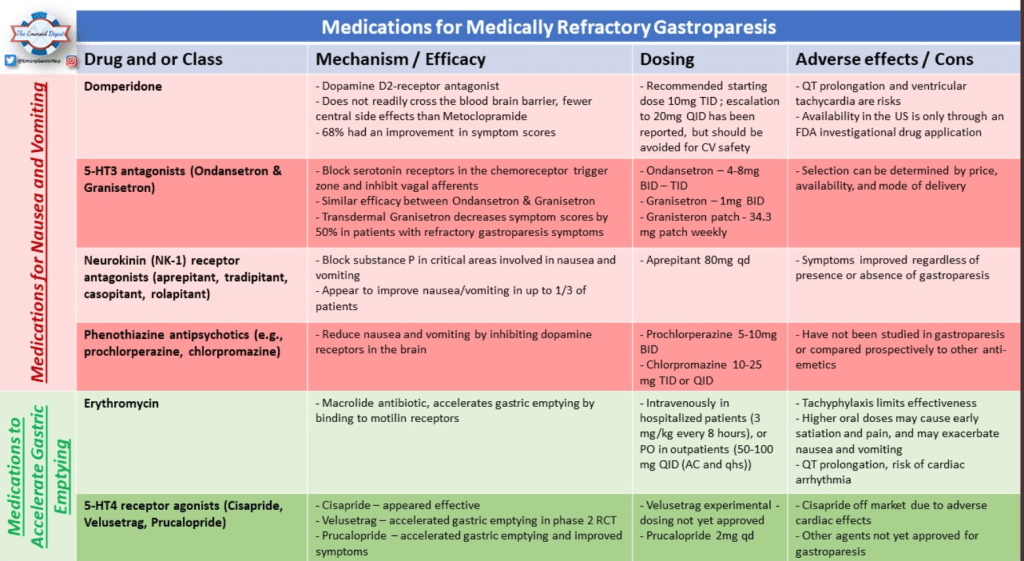
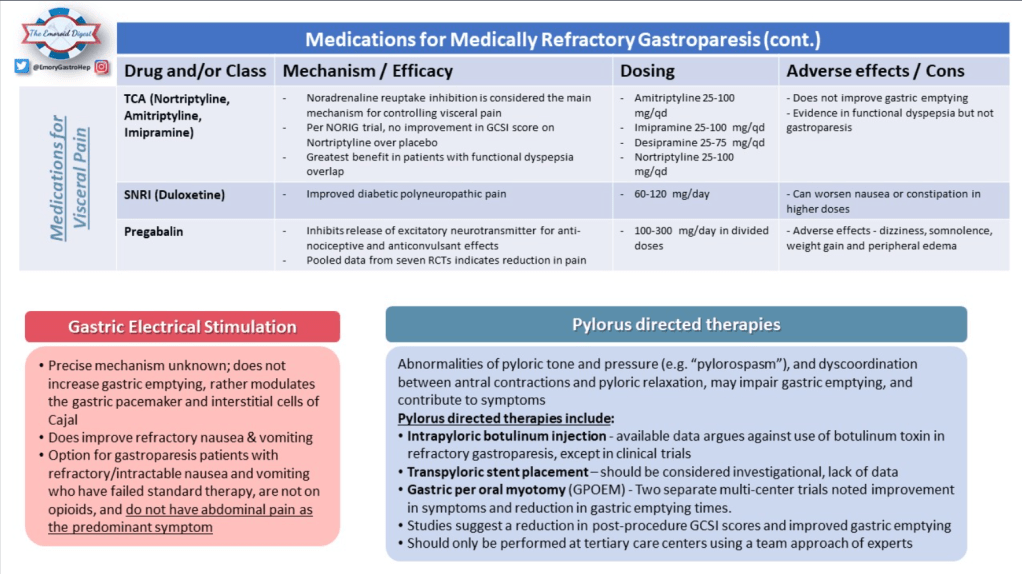
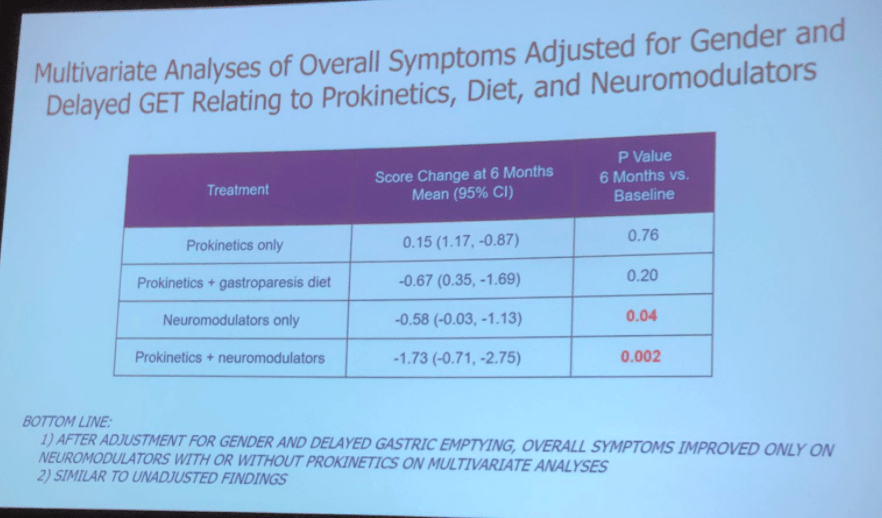
Recently, Dr. Katja Karrento gave our group a great update on chronic nausea. My notes below may contain errors in transcription and in omission. Along with my notes, I have included many of her slides.


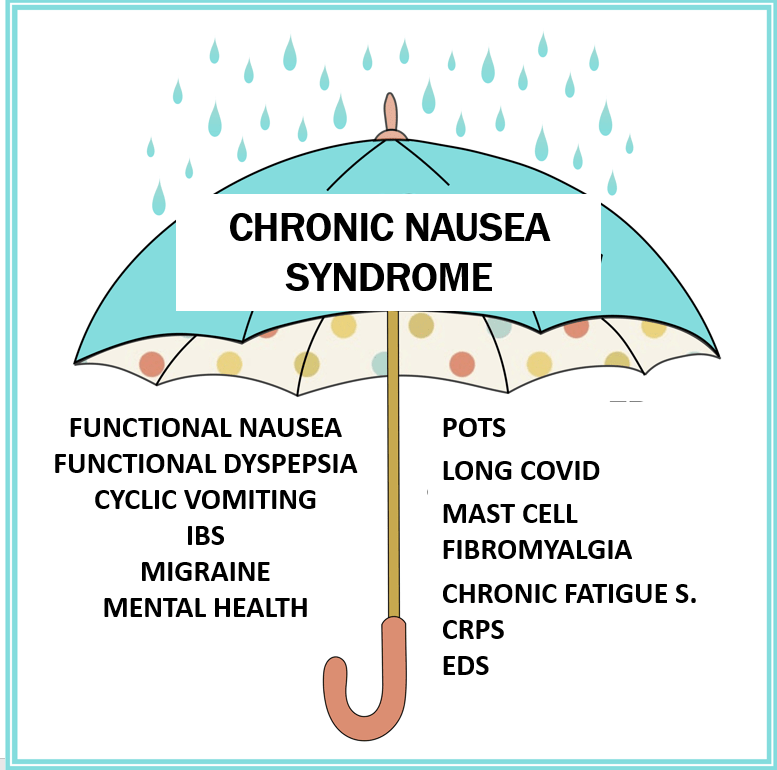
- Recent research suggests that functional nausea overlaps with other DGBIs including functional dyspepsia, IBS and even gastroparesis
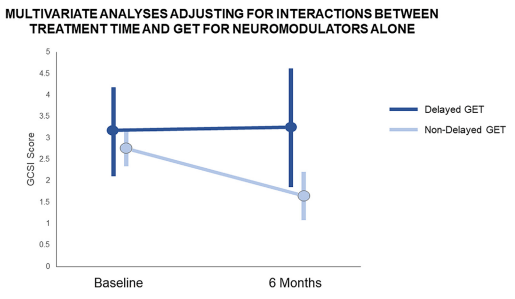
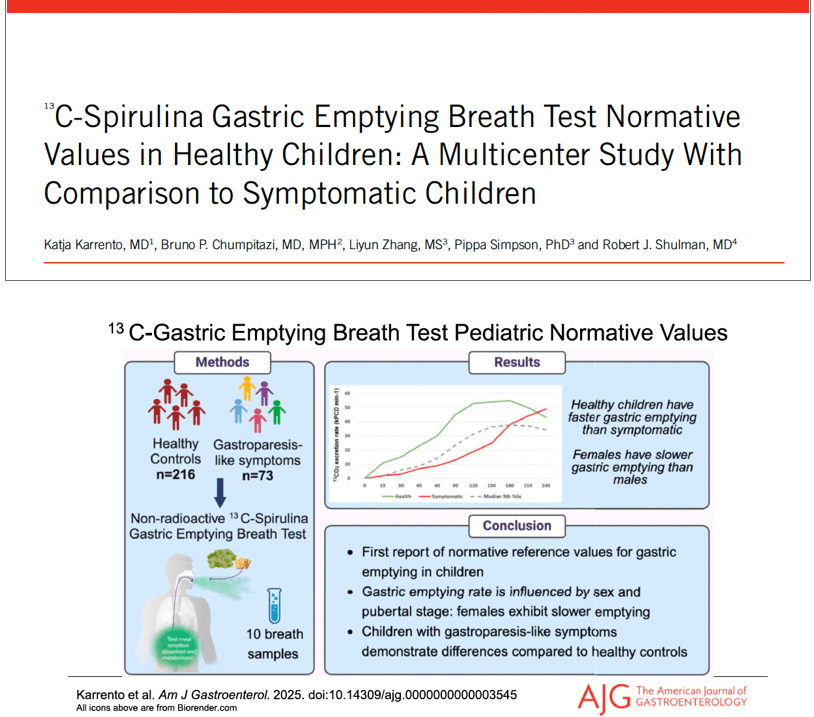
- Delayed gastric emptying is found in a subset of patients who meet criteria for functional nausea (though there are limitations of GE studies)
- Emerging research on use of C13-Spirulina breath test to assess gastric emptying
- Nausea is linked to disability more than stomach pain



- Functional dyspepsia and gastroparesis overlap and may be part of same disorder


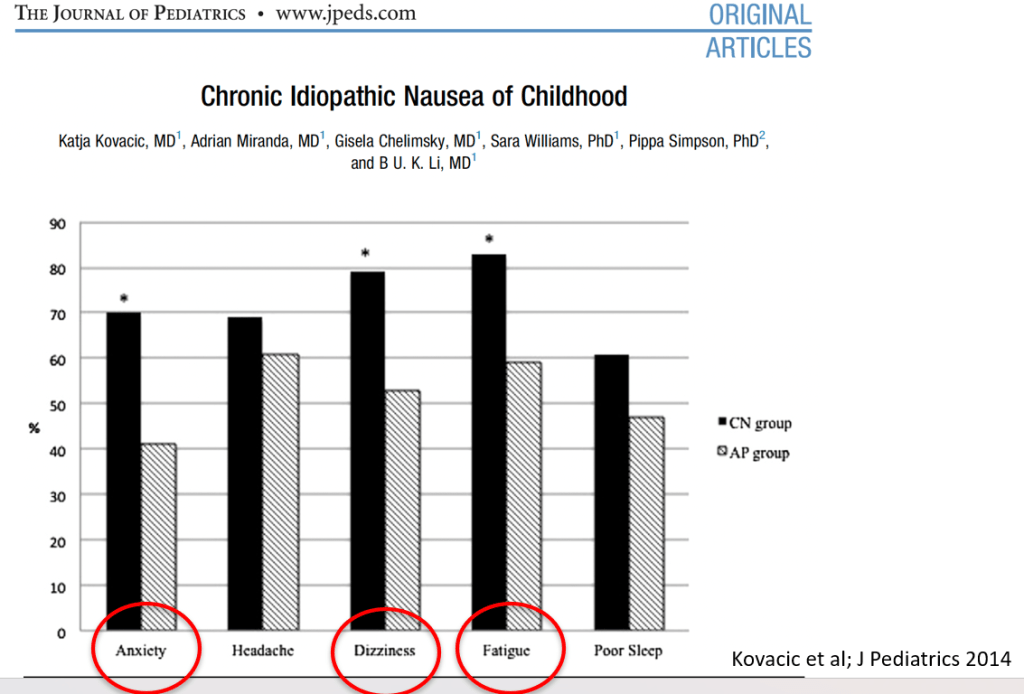
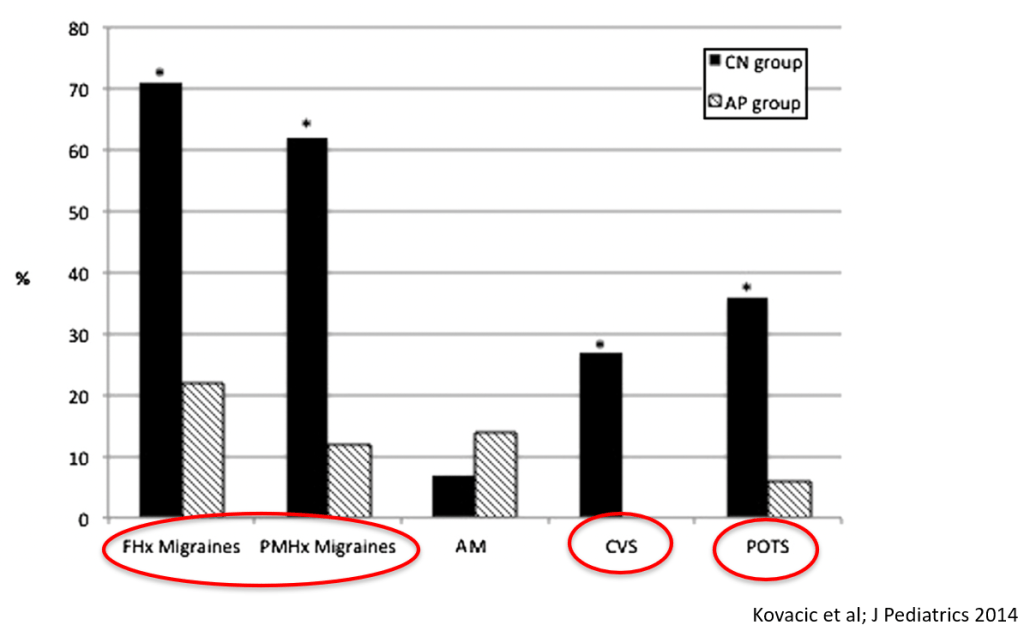
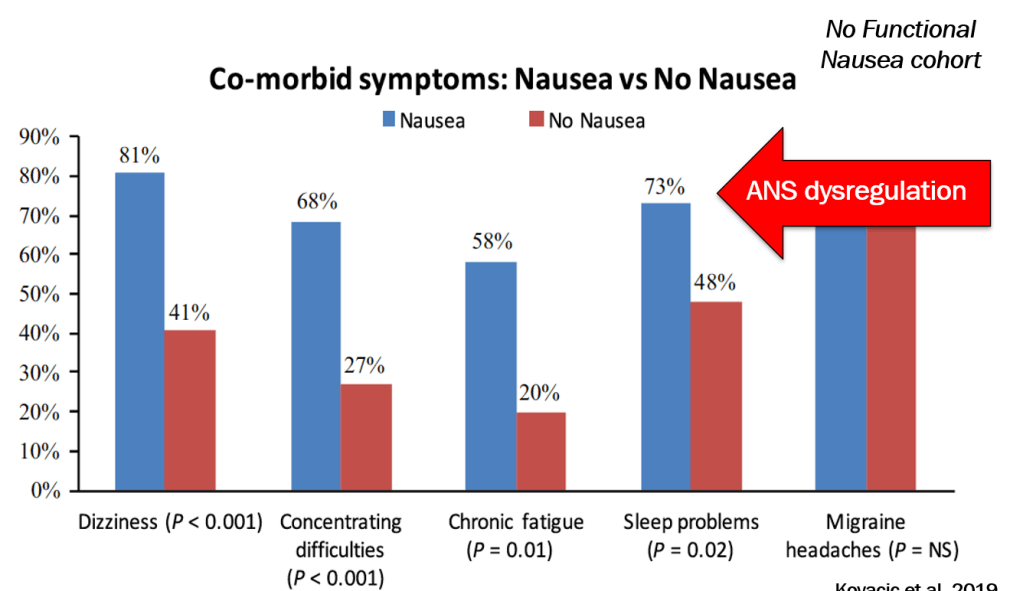
- Nausea is associated with numerous extraintestinal comorbidities symptoms

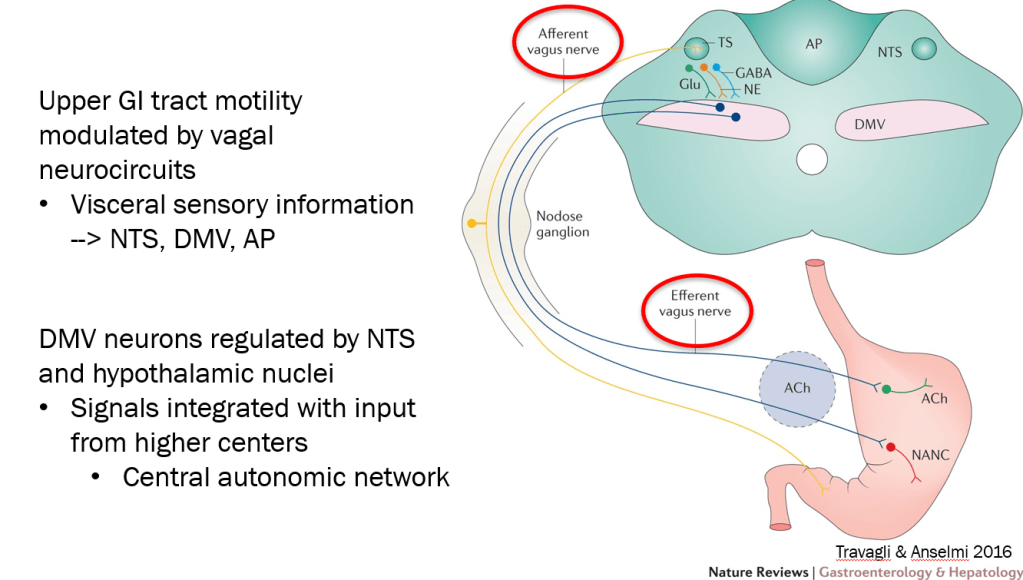
- DGBIs are associated with abnormal autonomic nervous system signaling
- Morning nausea, associated with being upright, is characteristic of dysautonomia. Other symptoms include ; palpitations, anxiety, sweating



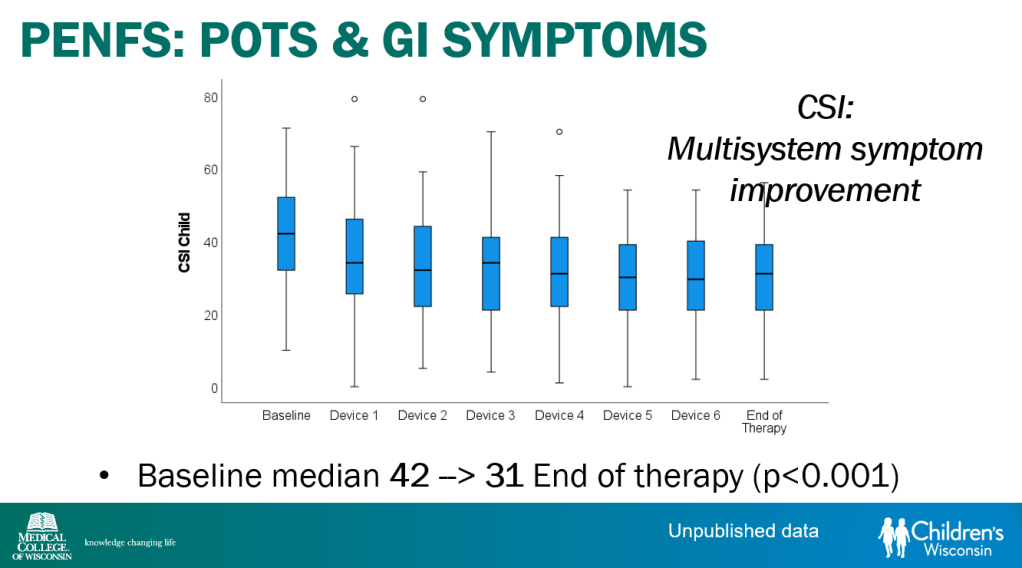
- POTS defined by increased HR >40 within 10 minutes with tilt test along with symptoms. In adults, increased HR>30 with symptoms.
- A small (n=10) functional MRI study showed abnormal brain network organization in patients with nausea and orthostatic intolerance

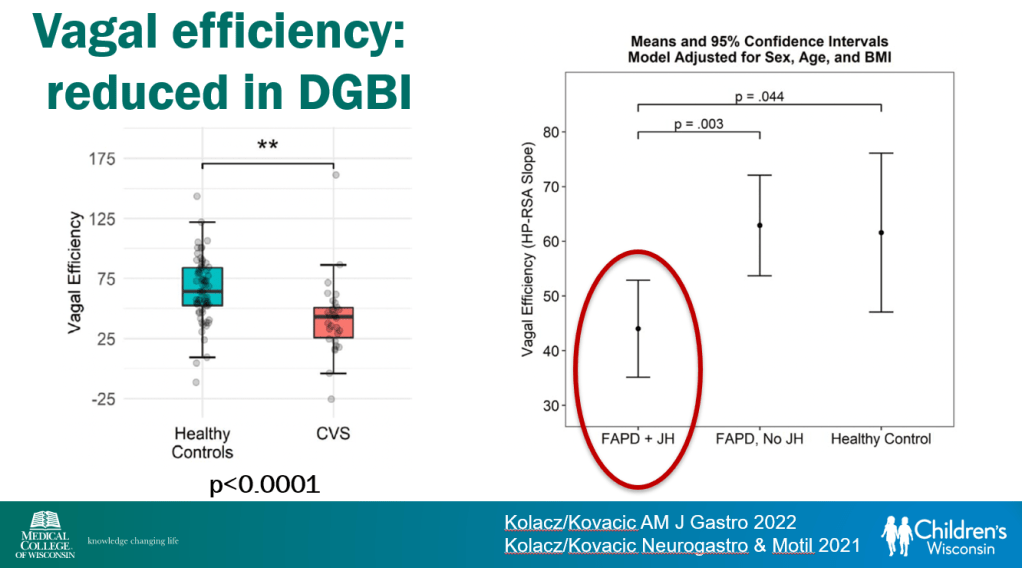
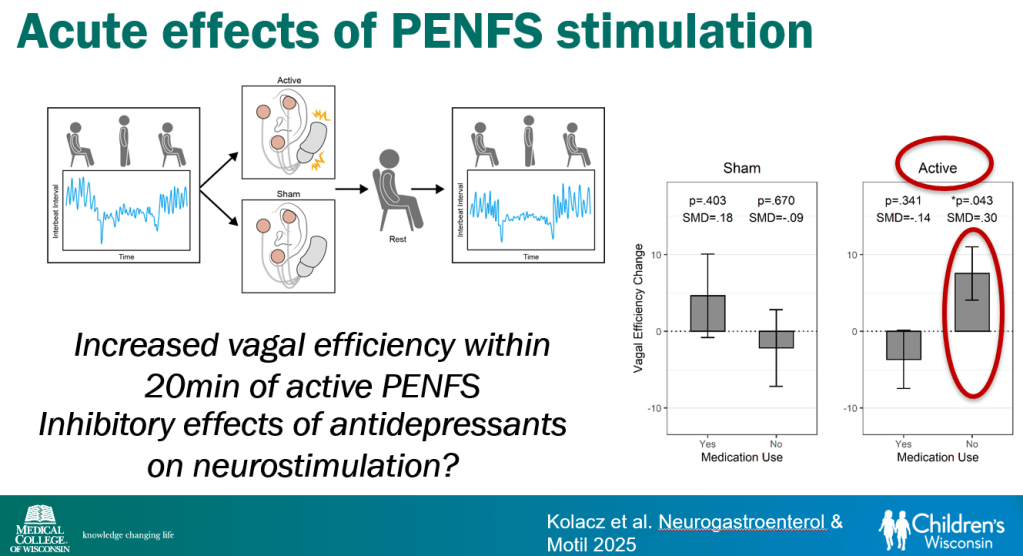
- Vagal efficiency, which is decreased in subsets of DGBI, is a measure of the heart’s regulation of sympathetically-elicited tachycardia (or ‘..heart’s regulation of postural tachycardia’)
- Cyclic vomiting is associated with autonomic nervous system alterations. In adolescence, the disorder often changes to chronic symptoms
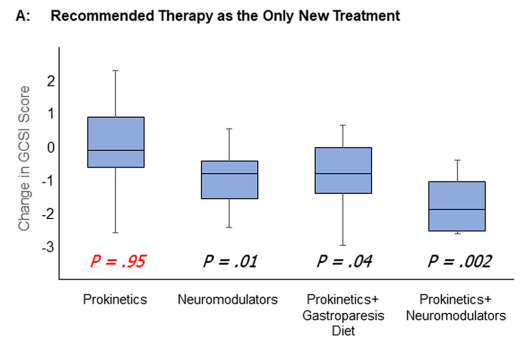
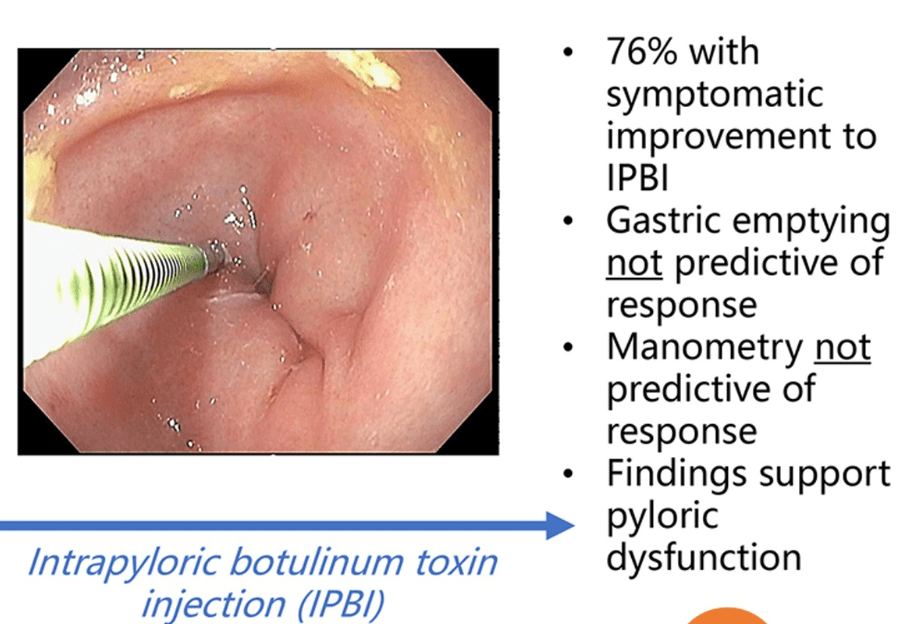
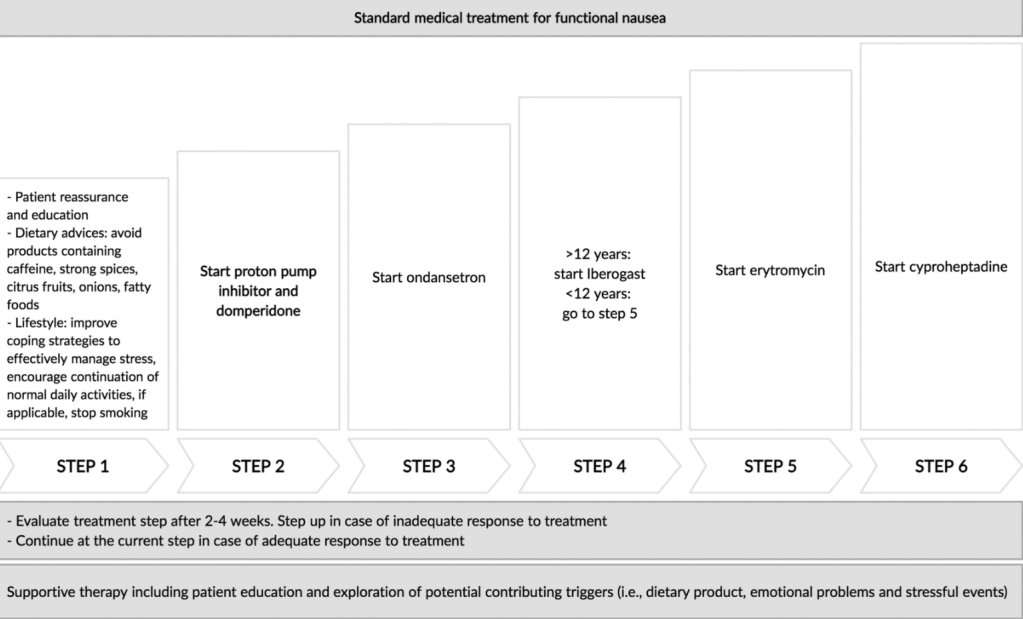
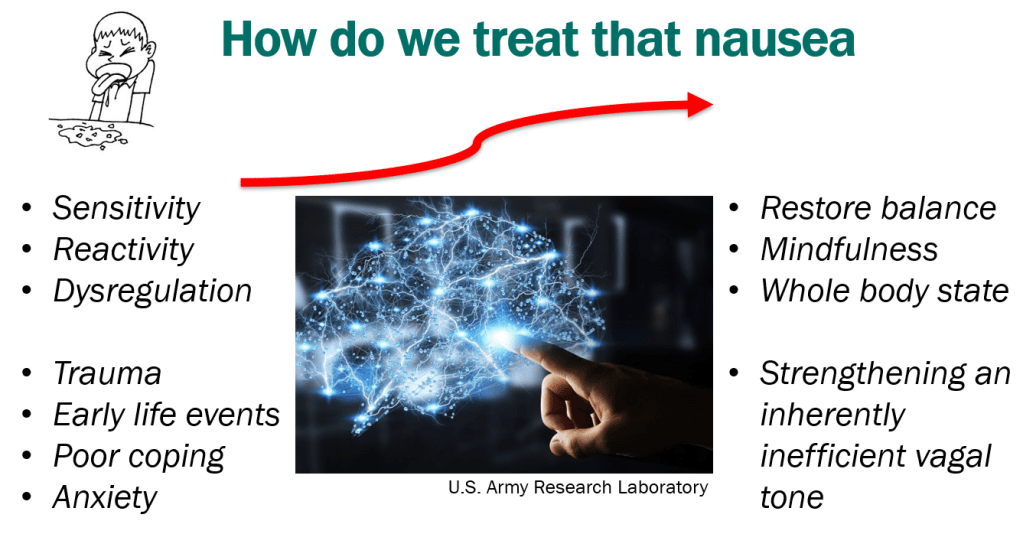
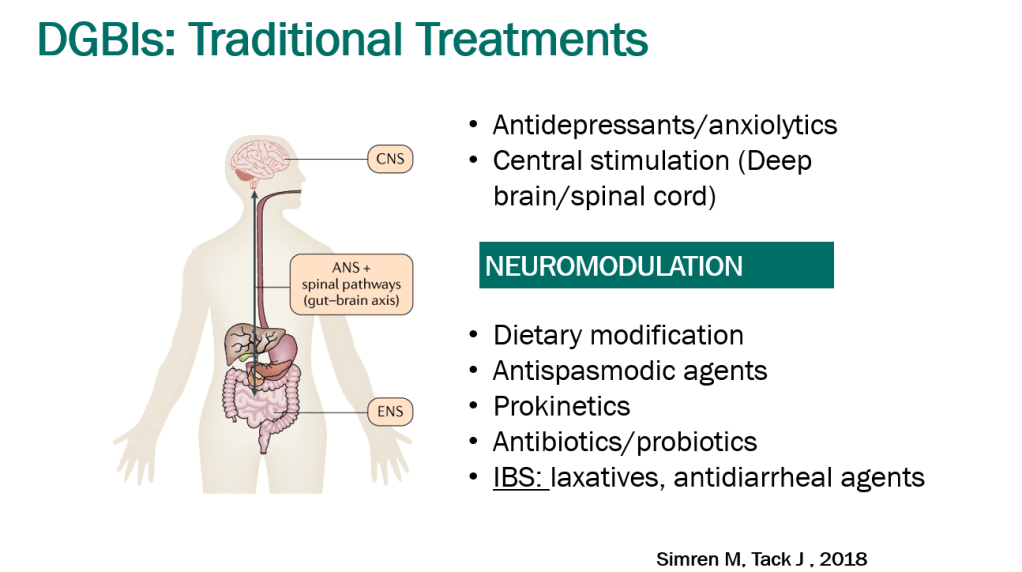
- Treatment Advice: Explain early on the difficulty in treating these symptoms. Usually there is not a quick fix medicine. Lifestyle changes and coping are integral parts. Neuromodulation is likely more effective than other approaches
- Dr. Karrento recommends The Dysautonomia Project to doctors and patients

- Exercise is helpful for DGBIs
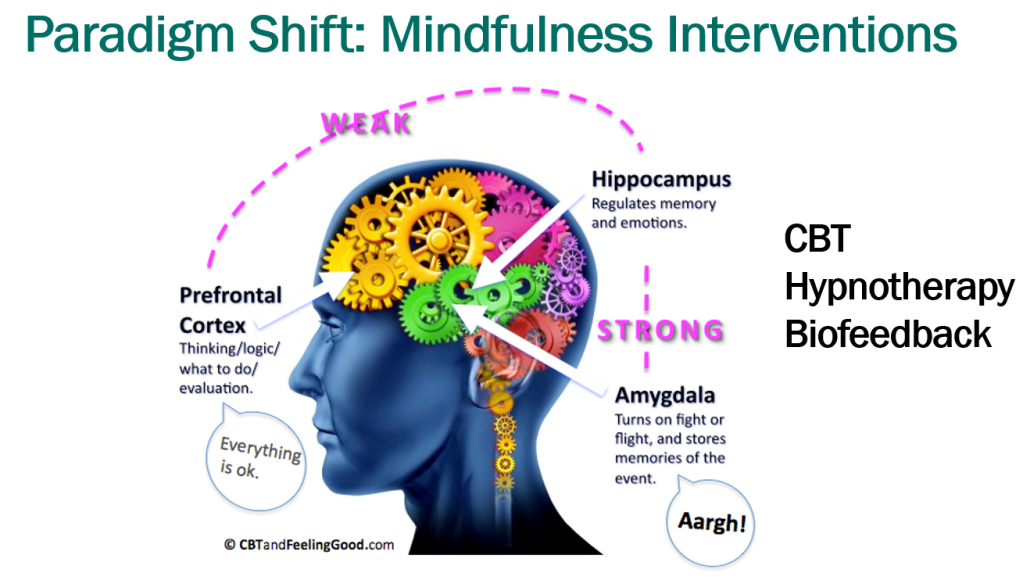
- Mindfulness training may help: CBT, Hypnotherapy, Biofeedback
- Potential treatments for autonomic dysfunction: Lifestyle changes, Neuromodulation, pharmacology: fludrocortisone, propranolol, pyridostigmine, midodrine
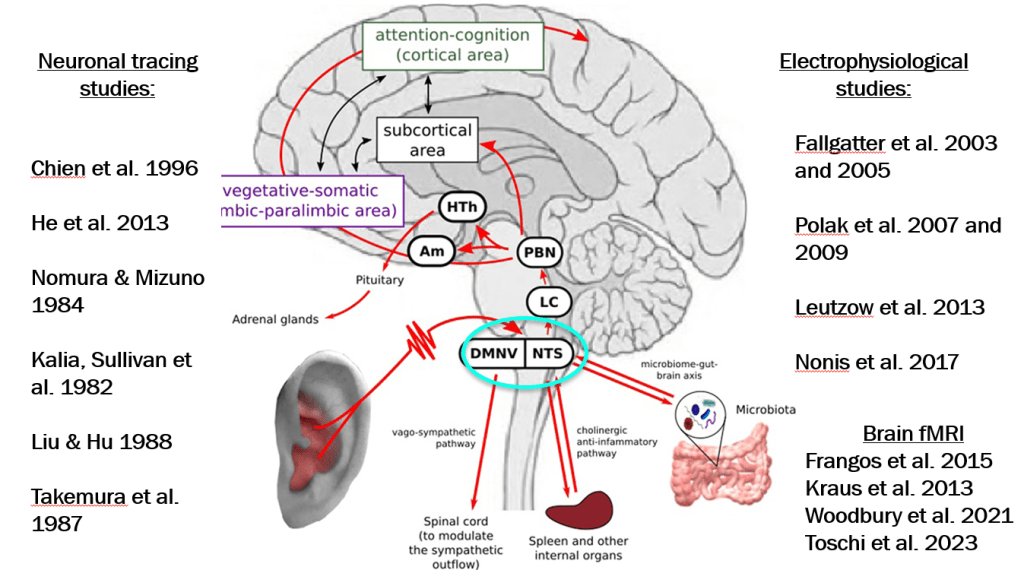
autonomic network
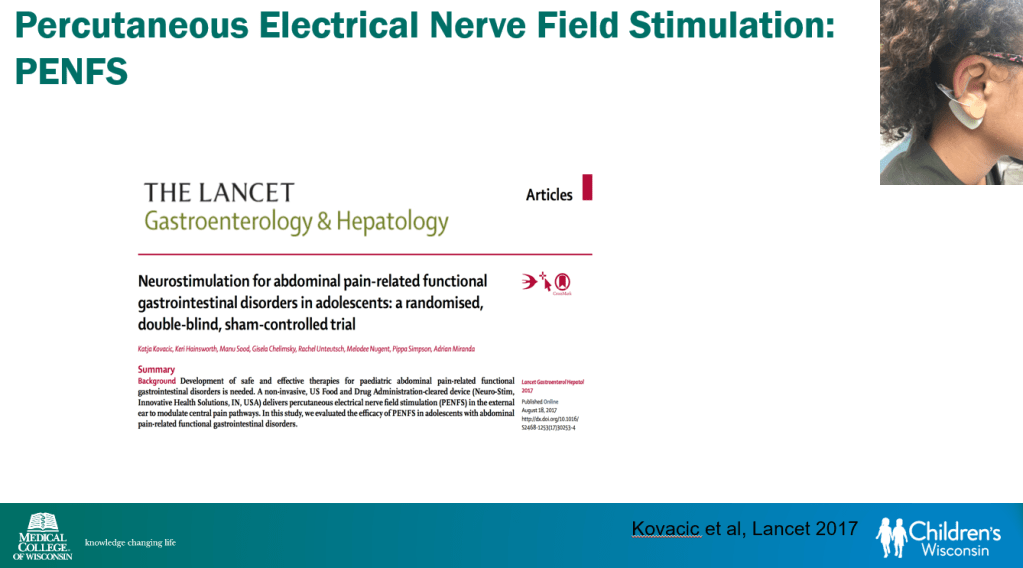
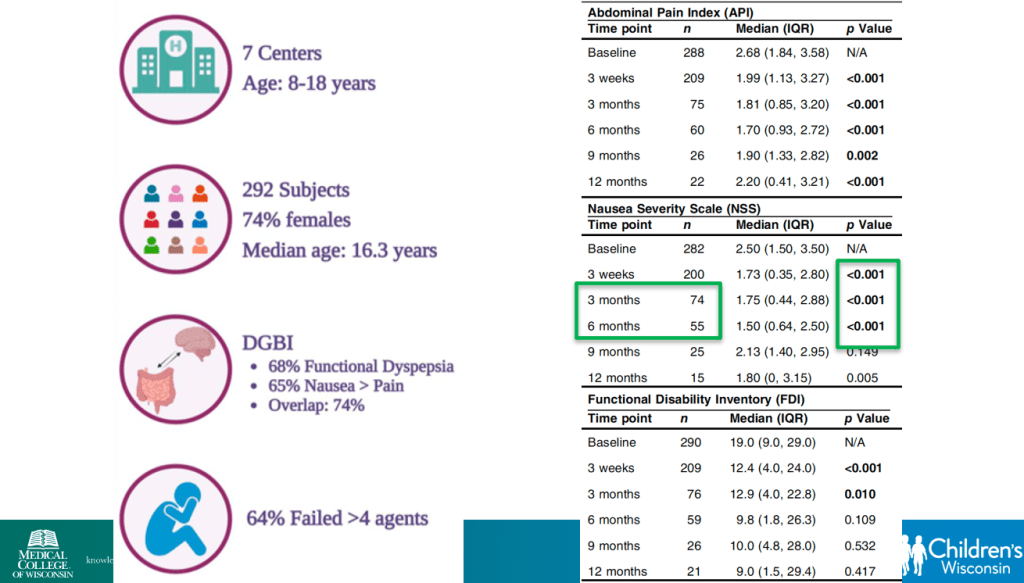
- Percutaneous electrical nerve field stimulation (PENFS) can be useful in functional nausea and many DGBIs



Related blog posts:
- Neuro-Stim for Refractory Cyclic Vomiting?
- Auricular Stimulation Associated with Less Pain, Less Disability, and Better Sleep
- Dr. B Li: Cyclic Vomiting Syndrome 2025
- Jump in Knowledge Regarding Gut-Brain Axis
- Dr. Carlo DiLorenzo: Advice for Managing DGBIs (Part 1)
- Dr. Carlo DiLorenzo: Advice for Managing DGBIs (Part 2)
- Dreaded Nausea (2022) Plus Skills or Pills
- Faulty Narrative with Functional Nausea Study
- Dr. Neha Santucci: Management of DGBIs in the Post-Pandemic Era (Part 1)
- Dr. Neha Santucci: Management of DGBIs in the Post-Pandemic Era (Part 2)
- 2025 Pediatric Cyclic Vomiting Syndrome Guidelines
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.