Briefly noted: RM Navari et al. NEJM 2016; 375: 134-42. Olanzapine (marketed as Zyprexa), compared with placebo, in combination with dexamethasone, aprepitant (or fosaprepitant) and a 5-hydroxytryptomaine type 3 antagonist (eg palonosetron, ondansetron, or granisetron) helped reduced nausea/vomiting. Among a total of 380 patients, 74% in the olanzapine group had no nausea/vomiting compared with 45% in the placebo group in the first 24 hours. In the 1st 120 hours, the rates of no nausea/vomiting were 37% vs. 22%. A “complete response,” defined as no emesis episodes and no rescue medications, occurred in 64% vs 41% in the 1st 120 hours. The most concerning side effect reported was severe sedation which was reported in 5%.
Category Archives: Gastroenterology
Image Only: Gastric Ascaris
Image
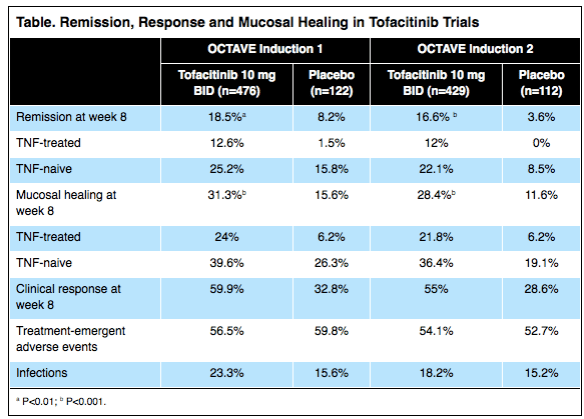
Latest on Tofacitinib for Refractory Ulcerative Colitis
From Gastroenterology & Endoscopy News July 2016: Tofacitinib Effective in Refractory and Severe UC
An excerpt:
Tofacitinib (Pfizer), an oral agent already approved for certain patients with rheumatoid arthritis, can induce clinical remission in up to 25% of individuals with moderate to severe, refractory ulcerative colitis (UC) and clinical response in as many as 60% of these patients.
The results, based on two placebo-controlled trials involving more than 1,100 patients, showed the drug also increased the risk for serum lipid elevations but was otherwise safe. Researchers presented the data at the 2016 annual meeting of the European Crohn’s and Colitis Organization (ECCO; oral presentation 019)…
The new data are from the OCTAVE Induction 1 and Induction 2 trials, identically designed, randomized, double-blind and placebo-controlled Phase III studies…In the OCTAVE 1 trial, 476 patients received 10 mg of tofacitinib orally twice daily for eight weeks and 122 received an oral placebo. In OCTAVE 2, 429 and 112 patients were randomized to receive the two regimens, respectively.
Also from Gastroenterology & Endoscopy News August 2016: Update on Diagnosis and Treatment for Ulcerative Colitis This article provides a succinct summary regarding diagnosis and treatments of ulcerative colitis; treatments discussed include emerging therapies like tofacitinib.
Cutting Edge for Endoscopic Control of Bleeding
A recent review elaborates on the newest methods for endoscopic control of bleeding. Topics included caplock clips, endoscopic suturing, and hemostatic sprays.
Full text: New Endoscopic Technologies and Procedureal Advances for Endoscopic Hemostasis (from Clinical Gastroenterology and Hepatology)
Related blog posts:
- All Bleeding Stops (part 2) | gutsandgrowth
- All bleeding stops | gutsandgrowth
- Watch the Bleeding Ulcer Stop! | gutsandgrowth
Hepatocellular Carcinoma Still Occurs in Patients who Clear HBsAg
Briefly noted:
Editorial: MH Nathanson, N Terrault Hepatology 2016; 64: 328-29. This very unusual editorial explains a published erratum of 2010 paper reversing a claim that patients with Hepatitis B who had achieved HBsAg clearance had markedly decreased rates of hepatocellular carcinoma (37 cases per 100,000 person-years). After correction of the arithmetic error, the rate of HCC in this population was actually 442 cases per 100,000 person-years. The editorial does reiterate that studies have shown lower HCC among those with low HBV DNA which is a prerequisite for HBsAg clearance. Exact risk is difficult in this population due to infrequent development of HBsAg loss and infrequent development of HCC. The message: While loss of HBsAg may lower HCC risk, there remains a need for HCC surveillance.
Related blog posts:
Fecal Calprotectin Monitoring Helpful at Identifying Relapse in IBD
Thanks to Ben Gold for this reference: Y. Zhulina et al. Aliment Pharm Ther 2016; 44: 495-504.
Methods:
- Patients aged 18 years or older, with a known diagnosis of IBD in clinical remission, were prospectively studied. Patients provided faecal samples every third month and were prospectively followed until the first clinical relapse or the end of the 2-year follow-up period.
- Relapse was defined as increasing symptoms necessitating intensified medical therapy or surgery.
Key finding:
- Among 104 patients, Crohn’s disease (n = 49) and ulcerative colitis (n = 55), 37 had a relapse. A doubling of faecal calprotectin level between two consecutively collected samples was associated with a 101% increased risk of relapse (HR: 2.01; 95% CI: 1.53–2.65; P < 0.001).
My take: Another study showing that stool calprotectin is quite useful. How long will it be until I will not need to write letters to insurance companies to get this test covered?
Also noted in the same issue:
“The safety of autologous and metabolically fit bone marrow mesenchymal stromal cells in medically refractory Crohn’s disease – a phase 1 trial with three doses” (pages 471–481) T. Dhere, I. Copland, M. Garcia, K. Y. Chiang, R. Chinnadurai, M. Prasad, J. Galipeau and S. Kugathasan. Aliment Pharm Ther 2016; 44: 471-81. This study examined the use of mesenchymal stromal cells in 12 patients.
In conclusion, a single infusion of fresh autologous bone marrow-derived mesenchymal stromal cells propagated ex vivo using a non xenogeneic human platelet lysate growth supplement at doses ranging 2–10 million cell/kg BW was well tolerated in patients with medically refractory moderate to severe Crohn’s disease in this preliminary study. Our data neither addressed long-term safety nor sustained efficacy. However, this study informs that a future phase 2 study
A previous study of mesenchymal stromal cells was briefly discussed in a previous blog: Sanjay Gupta is Wrong…about Stem Cell Therapy
Related blog posts:
Bowel Wall Vasculitis
All Bleeding Stops (part 2)
Several years ago, this blog provided a summary of the guidelines for stopping upper gastrointestinal bleeding (All bleeding stops | gutsandgrowth). Most of the recommendations are unchanged. A nice review of this topic (L Laine. NEJM 2016; 374: 2367-76), specifically focused on peptic ulcers, provides a few new pointers.
Two areas with more data:
- Transfusion. “A randomized trial showed lower rates of death (the primary outcome), rebleeding, and adverse events with a transfusion threshold of 7 g per deciliter than with a transfusion threshold of 9 g per deciliter.”
- Proton pump inhibitors. “A recent meta-analysis showed that intermittent oral or intravenous proton-pump inhibitor therapy results in outcomes that were non inferior to those after continuous infusion.” (JAMA Intern Med 2014; 174: 1755-62) For adults, the suggested dosing was 80 mg followed by 40 to 80 mg twice daily for 72 hours.
As before, the author recommends preoperative (30 min prior) erythromycin (250 mg) and states that a nasogastric tube in not needed. The author also recommends that in patients with idiopathic ulcers (not due to NSAIDs or H pylori) that ongoing (indefinite) use of once-daily maintenance therapy with a proton-pump inhibitor is recommended to prevent rebleeding.
Related blog posts:
Gluten-Free for IBS-D?
A recent study (I Aziz et al. Clin Gastroenterol Hepatol 2016; 14: 696-703) shows that a 6 week gluten-free diet reduced IBS-D symptoms in 29 of 41 (71%) patients.
- The authors performed a prospective study with all patients receiving a gluten-free diet. At 6 weeks, 21 of 29 who had responded to GFD continued GFD through 18 months followup.
- One difference with this study compared to prior studies –these patients were irritable bowel syndrome with diarrhea and fulfilled Rome III criteria. Celiac disease had been excluded with serology and histology; thus, these patients did not have “potential” celiac disease.
- In addition to GI symptoms like abdominal pain, distention, and stooling problems, patients experienced improvement in mood, fatigue and quality of life.
- The authors note that the response rate of 71% is much higher than they would have expected if the response was related solely to a placebo effect.
My take: This small study shows that a gluten free diet may be effective in improving the symptoms in many patients with IBS-D. Other studies have shown that several other diets are effective as well.
Related blog posts:
- Which Diet is Best for Irritable Bowel Syndrome? | gutsandgrowth
- Mechanism for FODMAPs diet | gutsandgrowth
- Low FODMAPs Diet in Pediatric Irritable Bowel | gutsandgrowth
Five Year Data on Magnetic Device for GERD
Here’s an abstract regarding the efficacy of a magnetic device for gastroesophageal reflux in adults:
Background & Aims
Based on results from year 2 of a 5-year trial, in 2012 the US Food and Drug Administration approved the use of a magnetic device to augment lower esophageal sphincter function in patients with gastroesophageal reflux disease (GERD). We report the final results of 5 years of follow-up evaluation of patients who received this device.
Methods
We performed a prospective study of the safety and efficacy of a magnetic device in 100 adults with GERD for 6 months or more, who were partially responsive to daily proton pump inhibitors (PPIs) and had evidence of pathologic esophageal acid exposure, at 14 centers in the United States and The Netherlands. The magnetic device was placed using standard laparoscopic tools and techniques. Eighty-five subjects were followed up for 5 years to evaluate quality of life, reflux control, use of PPIs, and side effects. The GERD–health-related quality of life (GERD-HRQL) questionnaire was administered at baseline to patients on and off PPIs, and after placement of the device; patients served as their own controls. A partial response to PPIs was defined as a GERD-HRQL score of 10 or less on PPIs and a score of 15 or higher off PPIs, or a 6-point or more improvement when scores on vs off PPI were compared.
Results
Over the follow-up period, no device erosions, migrations, or malfunctions occurred. At baseline, the median GERD-HRQL scores were 27 in patients not taking PPIs and 11 in patients on PPIs; 5 years after device placement this score decreased to 4. All patients used PPIs at baseline; this value decreased to 15.3% at 5 years. Moderate or severe regurgitation occurred in 57% of subjects at baseline, but only 1.2% at 5 years. All patients reported the ability to belch and vomit if needed. Bothersome dysphagia was present in 5% at baseline and in 6% at 5 years. Bothersome gas-bloat was present in 52% at baseline and decreased to 8.3% at 5 years.
Conclusions
Augmentation of the lower esophageal sphincter with a magnetic device provides significant and sustained control of reflux, with minimal side effects or complications. No new safety risks emerged over a 5-year follow-up period. These findings validate the long-term safety and efficacy of the magnetic sphincter augmentation device for patients with GERD. ClinicalTrials.gov no: NCT00776997.
It is important to note the following:
- Symptom control was not different between the magnetic sphincter and surgery
- The target population met the inclusion criteria: typical GERD symptoms, abnormal esophageal acid exposure by pH monitoring, partial response to daily PPI, and absence of large hiatus hernia or severe esophagitis
Related blog post: Stopping reflux with magnets | gutsandgrowth