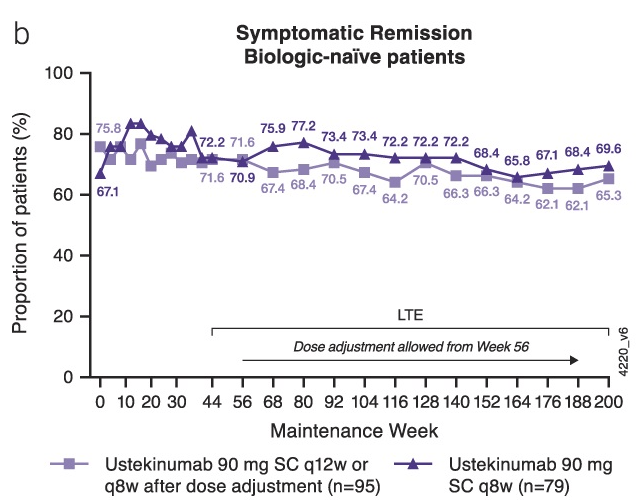
- SR Lieber et al. Liver Transplantation 2024; 30: 932-944. The financial burden after liver transplantation is significant among commercially insured adults: A large US National Cohort
- NN Ufere et al. Liver Transplantation 2024;30:918–31. Financial burden following adult liver transplantation is common and associated with adverse recipient outcomes.
- G Cholankeril, S Fasiha. Liver Transplantation 2024; 30: 873-874 (editorial). Open Access! Unseen foe: The financial toxicity of liver transplantation
- D Ladner, C Manski. Liver Transplantation 2024; 30:875-876 (editorial) Financial burden of cirrhosis is significantly reduced after liver transplantation
Using a nationally representative database spanning the years 2006-2021 with 1412 recipients, Lieber et al describe the patient financial burden after LT. Key findings:
- 21% had extreme liability > $10K for 1-year post-LT care
- 69% paid between $1 and 10K, with 48% having liability >$5K in the initial year following LT
- Medication costs comprised ~30% of outpatient financial liability
- Potential indirect costs from wages lost were $2,201–$6,073 per person
Ufere et al surveyed 207 adult recipients of liver transplant across 5 US transplant centers. Key finding: Nearly 1 in 4 experienced high financial burden (>/= 10% annual income spent on out-of-pocket costs)
The editorial by Ladner et al. notes that “LT is the only curative treatment for cirrhosis, with a 5-year survival of over 80% and, in most cases, returning patients from a chronic disease state to full physical and mental health.6 However, LT is resource-intensive, associated with an average cost of >$700,000, and is only performed in ~10,000 patients every year due to many barriers, including limited organ supply.7 Hence, this lifesaving therapy is currently provided to less than 1% of the patients affected by cirrhosis.8“
Ladner et al note that the financial burden of cirrhosis, though, is reduced after LT. “Without LT, the best that a patient can anticipate is to continue living with chronic liver disease/cirrhosis, with a baseline out-of-pocket cost of $19,390 per year. In this context, receiving an LT appears to be cost-saving for most (>80%) recipients of LT rather than a financial burden—with only 21% of recipients of LT having out-of-pocket costs >$10,000 during the first year after LT. In fact, the average out-of-pocket costs following LT appear quite similar to the $5,567 costs reported by patients without liver disease.5“
My take: LT is expensive. The financial burden needs to addressed with patients. However, for patients with cirrhosis, LT is usually a good value with lower out-of-pocket costs for 80% of recipients along with better quality of life, and longevity.
Related blog posts:
- Costs and Opportunity Costs in Pediatric Liver Transplantation
- Unfortunate or Unfair Disparities in Liver Transplantation
- Liver Shorts: Relationship of Hepatic Steatosis to Cardiovascular Disease and the Cost of Liver Transplantation
- What Can Go Wrong with Living Liver Transplantation for the Donor
- How to Lower Pediatric Liver Transplantation Waitlist Mortality