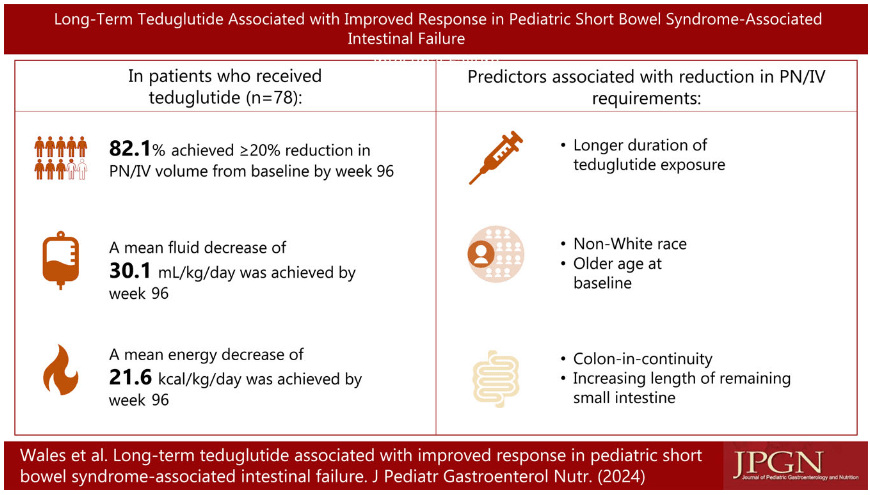
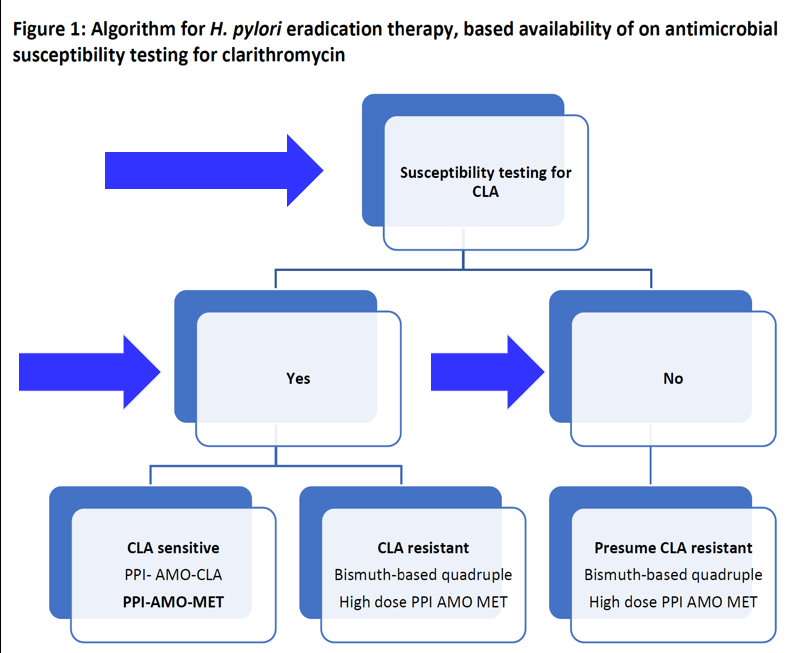
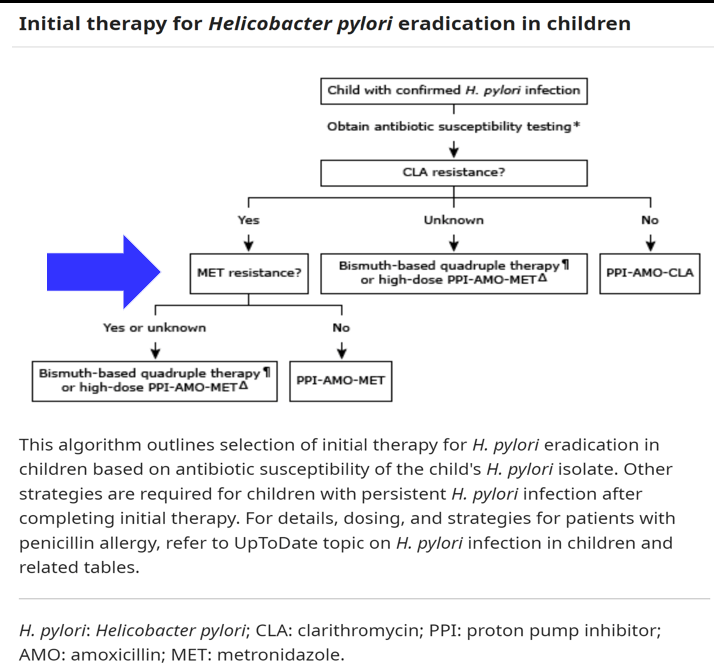
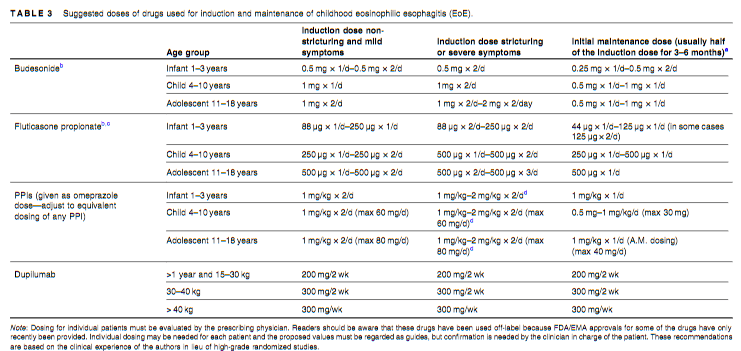
Link: OpenBiome Voluntarily Suspends FMT Shipments
An excerpt:
On September 29, 2024, OpenBiome will voluntarily suspend the distribution of investigational Fecal Microbiota Transplants (FMT) for patients with recurrent Clostridioides difficile infection (C. diff)...
Recent interaction with the FDA has informed our decision to voluntarily suspend the distribution of all investigational FMT as we continue to seek clear direction on aligning our operations with the final Enforcement Policy published in 2022. Our commitment has always been to adhere to FDA regulations and guidelines for the manufacture and distribution of investigational FMT as a therapeutic option for patients with C. diff. Thus, this is not a safety or quality matter. Investigational FMT preparations provided by OpenBiome are manufactured and distributed in compliance with current good manufacturing practices (cGMP).
We continue to hear from clinicians, our frontline partners, that despite the availability of FDA-approved therapeutics, there remain patients who do not respond to these treatments and, according to clinical guidelines, should have access to traditional FMT. ..
- Contact us. If you have a patient suffering from severe or fulminant C. diff, please contact us at 617-575-2201 or info@openbiome.org to discuss options.
- Share your thoughts. We believe the FDA would benefit greatly from hearing directly from survivors and their advocates about the urgent need for continued access to rigorously screened and tested FMT. If you or your C. diff patients are willing, please submit comments to ocod@fda.hhs.gov with a copy to Dr. David Kaslow, director Office of Vaccines Research and Review, at david.kaslow@fda.hhs.gov, and Dr. Peter Marks, director of the Center for Biologics Research and Evaluation at peter.marks@fda.hhs.gov. Or you may share your experiences with us directly using this FORM.
My take: As FDA-approved therapeutics have not received a pediatric indication, NASPGHAN involvement to try to keep FMT available for children would be a worthwhile endeavor. In the absence of having FMT available from OpenBiome, NASPGHAN experts could provide guidance on best practices for refractory C diff.
Related blog posts:
- Summary of ACG Clostridioides Difficile Adult Guidelines
- C difficile three-fer: Overdiagnosis with Multiplex Testing, Fidaxomicin Pediatric Approval, & Changing Incidence
- New Target For Clostridioides Difficile Treatment
- How Effective and Safe is Fecal Microbiota Transplantation in Immunocompromised Pediatric Patients with Clostridioides difficile?