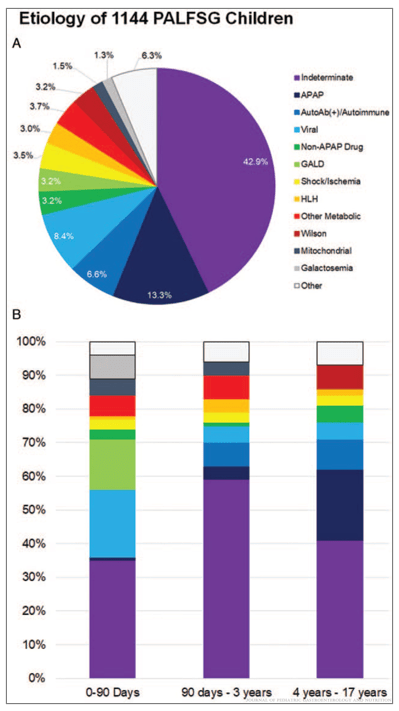
From GeneralSurgeryNews.com: Open Access: Novel Device Uses Mechanotransduction To Treat Small Bowel Syndrome
An excerpt:
“The basic concept is similar to distraction osteogenesis, which orthopedic surgeons have used for years, applying distraction force to broken bone that will grow up to a millimeter a day,” said Andre Bessette, the CEO and a co-founder of Eclipse Regenesis, Inc…
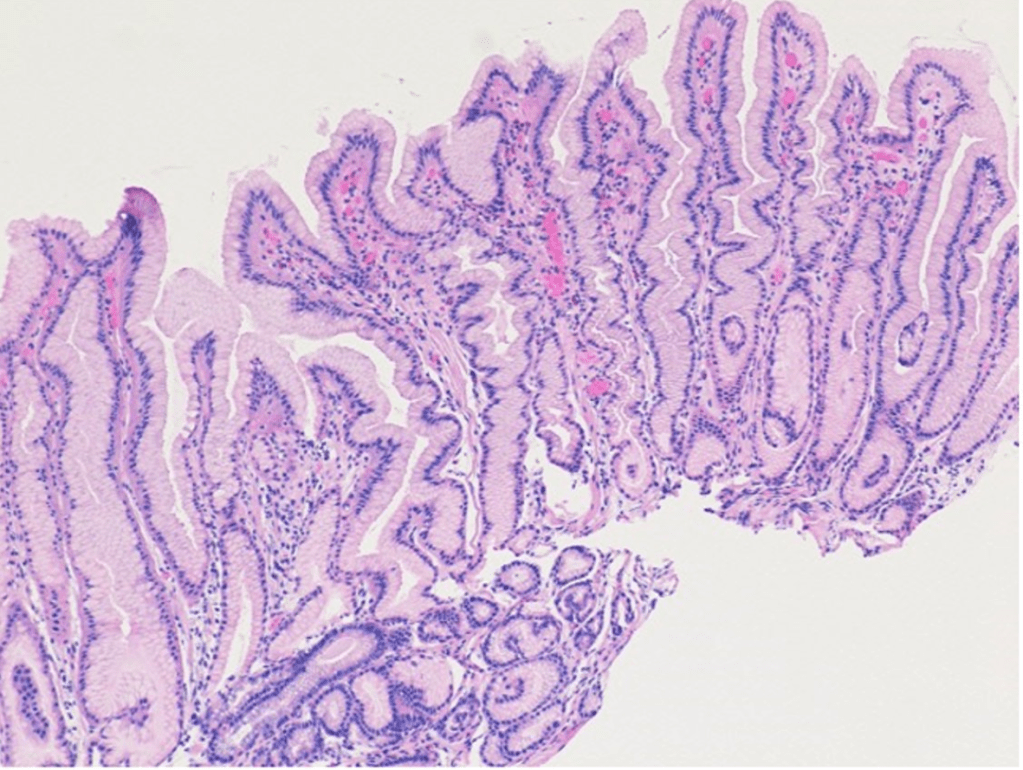
To regenerate small-bowel tissue, a surgeon inserts the device, which looks like a small, compressed coil, inside the small intestine and secures both ends with plication sutures applied to the outside of the intestine. Over two to three weeks, the device slowly expands to its uncompressed state, stimulating new tissue growth—ultimately two to three times the segment’s original length, about 4 cm...[thus] they’ll need more than one device applied or more than one procedure.
Once this process is complete, the chromic sutures dissolve over about a month, allowing the device to pass through the body to be excreted...
[The researchers] expect to start these [human] trials in the first half of 2022, and they have identified two primary investigator sites: Boston Children’s Hospital and Cincinnati Children’s Hospital
Preclinical studies: Eclipse’s website at www.eclipseregenesis.com/ publications.