


This Georgia AAP (virtual) board meeting started with a brief review from Dr. Kathleen Tomey (Department of Health)
Some slides:


AAP Update from Dr. Scornik:



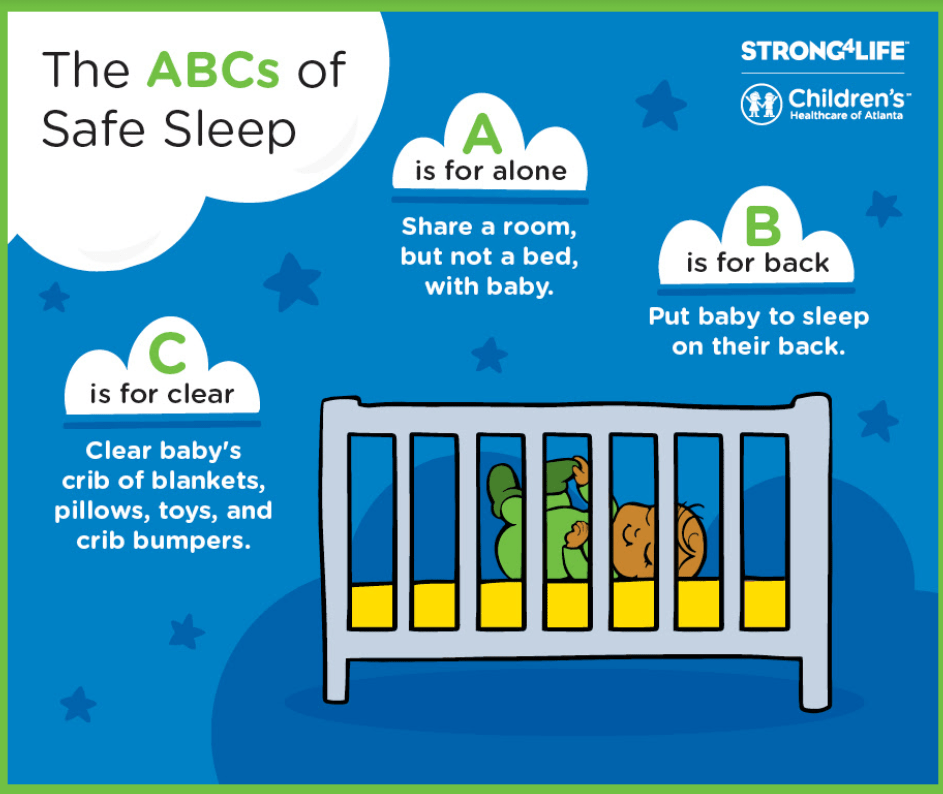
Safe sleep initiatives briefly discussed by Dr. Sarah Lazarus which aligns with Strong4Life campaign:




Other information:
Update on E-Cigarettes Webinar*+: Wednesday, October 28 at 12:30 pm
Please note new date! Here’s a chance to still register.
First in a series of three webinars offered to Georgia Pediatricians on the growing epidemic of youth e-cigarette use
Faculty: Alice Little Caldwell, MD, FAAP
https://register.gotowebinar.com/register/8457518617359610381
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition
F Poizeau et al. JAMA Dermatol. Published online September 9, 2020. doi:10.1001/jamadermatol.2020.2977. Association Between Early Severe Cardiovascular Events and the Initiation of Treatment With the Anti–Interleukin 12/23p40 Antibody Ustekinumab
Methods: “This case-time-control study used data from the French national health insurance database, covering 66 million individuals, on all patients exposed to ustekinumab between April 1, 2010, and December 31, 2016, classified according to their cardiovascular risk level (high- and low-risk strata). The risk period was the 6 months before the SCE, defined as acute coronary syndrome or stroke, and the reference period was the 6 months before the risk period. Statistical analysis was performed from September 20, 2017, to July 6, 2018.”
Key findings:

My take: This study suggests that the initiation of ustekinumab treatment may trigger SCEs among patients at high cardiovascular risk; however, the study conclusions are limited as this was an observational study (not a randomized trial).
I remember when I was first taught to dictate consultations. I was a resident doing a genetics rotation. My mentor, Peter Dignan, made several suggestions. One was to try to always include something nice about the patient. Many of my current colleagues are amused how many of my patients are ‘delightful.’ While there are a lot reasons for putting some kind information in the medical record, Dr. Dignan emphasized that patients and families can get hold of their records and undoubtedly they would appreciate a friendly word. Now with the 21st Century Cures Act Final Rule, access to records and notes will expand considerably and Dr. Dignan’s advice is probably even more important.
A good source of information on this new law, which is in effect Nov 2nd, 2020, is from the 33charts blog—Cures Act Final Rule – How It Will Change Medicine: “The ONC Cures Act Final Rule (Cures Rule) is the biggest health care law you’ve never heard of. But it’s a law that’s going to fundamentally shift the way we see patients and their information. It will change how physicians talk to patients about information. It will shift the way health professionals connect patients to their information.” This blog post details how this change is going to affect both healthcare providers and families. The two key changes are
The key point: “The Cures Rule will force health systems to be better stewards of information on behalf of our patients. I think this is going to force health professionals to help patients think about information and what they do with it. It will force patients to recognize the difference between information and knowledge and wisdom. I suspect that the most critical ultimate change will be transparent conversations and more timely physician follow-up on high stakes studies.”
Another take on the 21st Century Cures Act: C Blease et al. Annals of Internal Medicine; 2020: https://doi.org/10.7326/M20-5370. New U.S. Law Mandates Access to Clinical Notes: Implications for Patients and Clinicians

Some additional information (from EPIC training) — there are limited exceptions for note sharing:

Another reference:

My take: When this rolls out, a lot of physicians (myself included) will need to make some adjustments; since it is the law, don’t expect to avoid these changes. I expect early on this will generate a lot of additional questions and phone calls. In the long run, this is likely to improve communication, transparency, and availability of patient information. For example, it is more likely that needed lab results from referring physicians will be more available after this law is in effect.
A recent editorial (JG Hashash et al. Inflamm Bowel Dis 2020; 26: 1315-1318. Approach to Latent Tuberculosis Infection Screening Before Biologic Therapy in IBD Patients: PPD or IGRA?) provides some guidance on screening for tuberculosis prior to biologic therapy as well as background on how these tests work.
Key points:
My take: TB blood testing is more convenient but more costly. The authors indicate that for patients from low endemic areas, yearly TB testing is mainly to check boxes mandated by insurance companies rather than improving care.
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition
A recent study (DE Freedberg et al. Gastroenterol 2020; DOI:https://doi.org/10.1053/j.gastro.2020.05.053. Famotidine Use Is Associated With Improved Clinical Outcomes in Hospitalized COVID-19 Patients: A Propensity Score Matched Retrospective Cohort Study, highlighted on AGA blog, indicates that famotidine may improve outcomes in those with COVID-19.
Methods: Freedberg et al collected data from 1620 patients who tested positive for SARS-CoV-2 no more than 72 hours following admission; 84 of the patients (5.1%) had received famotidine (any dose, form of administration, or duration; median dose of 136 mg) within 24 hours of hospital admission.
Key finding: After the authors adjusted for baseline patient characteristics, use of famotidine was independently associated with risk for death or intubation (adjusted hazard ratio 0.42, 95% CI, 0.21–0.85). This did not change after propensity score matching to balance covariables (hazard ratio 0.43, 95% CI 0.21–0.88).
My take: While these results indicate that famotidine may improve outcomes with COVID-19, a randomized controlled trial is needed to confirm these findings (currently one is underway to determine whether famotidine can improve clinical outcomes in hospitalized patients with COVID-19 (NCT04370262)).
AGA Blog Summary: Use of Famotidine Associated With Improved Outcomes of Hospitalized COVID-19 Patients
Related blog posts:
In some patients with celiac disease, institution of a gluten-free diet may be detrimental without good dietary counseling as a highly-processed diet can increase the risk of adverse cardiovascular events.
A recent study (J Runde et al. JPGN 2020; 71: 533-535. A Narrow Window: Booming Gluten-free Market and Fostering Healthy Dietary Habits in Children With Celiac Disease) assessed dietary patterns of children with celiac disease and indicates that early counseling is crucial.
In total dietary surveys were completed for 100 children with celiac disease. Key findings:
The authors speculate that highly-processed foods are leading to obesity which is increasingly reported in pediatric celiac disease.
My take:
Related blog posts:

A recent case series (A Assa et al. JPGN 2020; 71: 516-520. Therapeutic Drug Monitoring-guided High-dose Infliximab for Infantile-onset Inflammatory Bowel Disease: A Case Series) describes four infants (2 mo-12 mo) with infantile-onset IBD who received high doses of infliximab.
Treatments regimens utilized infliximab dosing of 10-22 mg/kg/dose with initial three doses over 2-4 weeks. Other prior treatments in these patients included antibiotics (eg. vancomycin/gentamicin) and corticosteroids. Sulfasalazine was administered in two of the patients.
Other Key Points:
The authors note that higher doses may increase adverse events, including infections
My take: This study shows that highly-selected patients may need both accelerated and higher doses of infliximab to enable response. It adds to the literature that children, in general, are at high risk of under-dosing with ‘standard’ infliximab dosing.
Related blog posts:
A recent commentary (M Fiedler. NEJM 2020; 383: 1197-99. Competing Visions for the Future of Health Policy) describes two competing approaches to U.S. healthcare policy.
My take: Overall, I favor more expansive health care coverage.
RD Santos et al. NEJM 2020; 383: 1317-1327. Evolocumab in Pediatric Heterozygous Familial Hypercholesterolemia
Methods: In this 24-week, randomized, double-blind, placebo-controlled trial with pediatric patients (n=157) with heterozygous familial hypercholesterolemia, patients 10 to 17 years of age were treated with evolocumab. All had been receiving lipid-lowering treatment before screening and had LDL cholesterol level of 130 mg/dL.
Key finding: At week 24, the mean percent change from baseline in LDL cholesterol level was −44.5% in the evolocumab group and −6.2% in the placebo group.
My take: Long-term data are needed. However, in high risk patients who have not responded to other intensive treatment, evolocumab may be worthwhile.
J Lei et al. NEJM 2020; 383: 1340-1348. HPV Vaccination and the Risk of Invasive Cervical Cancer
Methods: We used nationwide Swedish demographic and health registers to follow an open population of 1,672,983 girls and women who were 10 to 30 years of age from 2006 through 2017.
Key findings:
My take: HPV vaccine (aka ‘Cervical Cancer Vaccine’) may lower the risk of cancer by 88% in those vaccinated before the age of 17 years.
Short answer -you should always be worried about words that are so difficult to spell.
Two years ago, this blog highlighted recommendations from the AAP regarding food additives (Food Additives and Child Health).
More information of phthalates is available from the Cincinnati Children’s website: Phthalates and Your Health. The backdrop to this article is in relation to a recently published study Pubertal Growth, IGF-1, and Windows of Susceptibility: Puberty and Future Breast Cancer Risk (F Biro et al. Journal of Adolescent Health 2020).
Key points from Cincinnati Children’s website:
My take: Environmental research is quite difficult. Yet, we know that changes in our environmental exposures are directly related to many adverse health outcomes since changes in disease frequencies related to genetics is a much slower process.