Briefly noted: N Terrault et al. Gastroenterol 2018; 155: 705-18. In two trials, ADAPT-1 and ADAPT-2, the use of avatrombopag, a thrombopoietin receptor agonist, was superior to placebo in increasing platelet counts and reducing need for platelet transfusion for bleeding related to procedures.
Romaine Lettuce and E coli Outbreak
According to the CDC update (Nov 26, 2018) –Outbreak of E. coli Infections Linked to Romaine Lettuce: “43 people infected with the outbreak strain of E. coli O157:H7 have been reported from 12 states. A list of the states and the number of cases in each can be found on the Map of Reported Cases page.
Illnesses started on dates ranging from October 8, 2018 to October 31, 2018. Ill people range in age from 1 to 84 years, with a median age of 25. Sixty-nine percent of ill people are female. Of 38 people with information available, 16 (42%) have been hospitalized, including one person who developed hemolytic uremic syndrome, a type of kidney failure. No deaths have been reported.”
“CDC is advising that U.S. consumers not eat and retailers and restaurants not serve or sell any romaine lettuce harvested from the Central Coastal growing regions of northern and central California. If you do not know where the romaine is from, do not eat it.”
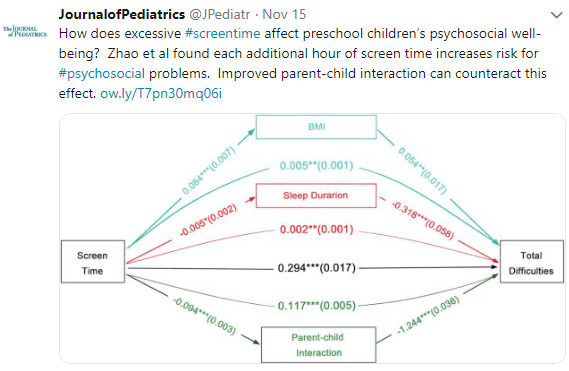
Interesting Study -Detrimental Dose-Response of Screen Time
In 1995, there was a Batman movie, “Batman Forever,” in which one of the central villains, the Riddler, places these brainwave devices over the TVs to gain control of Gotham. The sad part, according to a recent study (J Zhao et al. J Pediatr 2018; 202; 157-62) is there is no need to add a brainwave device to a TV set. Excessive screen time alone is quite detrimental.
In this cross-sectional survey in Shanghai with more than 20,000 children, the authors found the following:
- Mean screen time for preschool children was 2.8 hrs per day. 78.6% exceeded 1 hour per day and 53% exceeded 2 hrs per day.
- Every additional hour of screen time was associated with increased risk for poor psychosocial well-being; this effect on well-being had a number of mediators including reducing parent-child interaction as well as increased body mass index and reduced sleep duration.
My take: This study reinforces the consequences of excessive screen time –now, the hard part — how to translate these findings into reduction in screen time.
Related blog posts:
- Skinnier TVs and Heavier Children
- Sleep Duration and Subsequent Obesity | gutsandgrowth
- 7 Ways Parents Can Influence Risk of Obesity | gutsandgrowth
- Staggering cost of obesity | gutsandgrowth
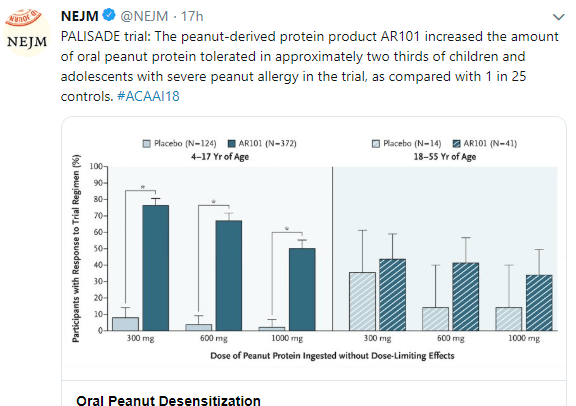
New Strategy to Overcome Severe Reactions to Peanuts
A recent study (N Engl J Med 2018; 379:1991-2001) showed that
Link to abstract: AR101 Oral Immunotherapy for Peanut Allergy
Methods: Participants with an allergic response were randomly assigned, in a 3:1 ratio, to receive AR101 (a peanut-derived investigational biologic oral immunotherapy drug) or placebo in an escalating-dose program.
Conclusions: In this phase 3 trial of oral immunotherapy in children and adolescents who were highly allergic to peanut, treatment with AR101 resulted in higher doses of peanut protein that could be ingested without dose-limiting symptoms and in lower symptom severity during peanut exposure at the exit food challenge than placebo.
Related blog posts:
Probiotics -Lack of Efficacy for Acute Gastroenteritis (Part 2)
This blog post highlights a second study showing a lack of efficacy of probiotics for acute gastroenteritis. Link to 2 minute Summary: Quick Take on Probiotics for AGE
My take: While some probiotic strains have been shown to be helpful in some conditions (eg. antibiotic associated diarrhea), this study indicates that probiotics are likely ineffective in altering the course of acute gastroenteritis.
SB Freedman et al. N Engl J Med 2018; 379:2015-2026 Link to abstract: Multicenter Trial of a Combination Probiotic for Children with Gastroenteritis
Related blog posts:
- The Truth about Probiotics: Constipation Version
- Are Probiotics Effective in Changing the Microbiome?
- Lactobacillus rhamnosus GG Associated with Increased Necrotizing Enterocolitis in Observational Study
- How helpful are probiotics?
- Balanced summary of probiotics & Microbiome effects on brain
- Probiotics for Crohn’s Disease –No Beneficial Effects Noted | gutsandgrowth
- Probiotics For Fatty Liver Disease | gutsandgrowth
- “Low quality of evidence; strong recommendation” for Probiotics in Gastroenteritis
- Probiotics for Colic
- Do Probiotics Really Help Crying Infants? | gutsandgrowth
- Pushback on Probiotics
- One More Day Syndrome & Necrotizing Enterocolitis | gutsandgrowth
- Probiotics, Atopy, and Asthma | gutsandgrowth
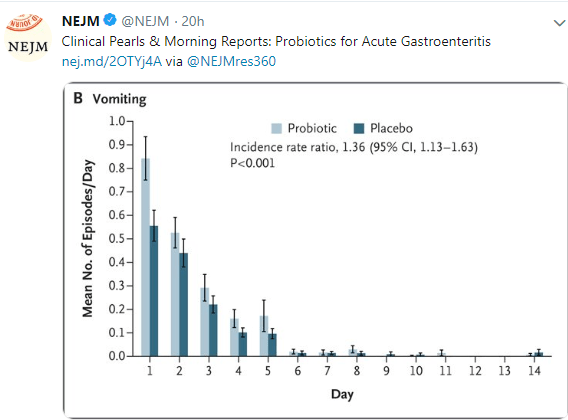
One for the Probiotic Skeptics: Lack of Efficacy in Acute Gastroenteritis (Part 1)
My opinion has been that probiotics are generally over-hyped and are likely ineffective for many conditions in which they are commonly used (see related blog posts below).
A recent study (D Schnadower et al.N Engl J Med 2018; 379:2002-2014) provided more data to support this skeptical view when probiotics are utilized for acute gastroenteritis. Another study in the same issue will be highlighted tomorrow and reaches a similar conclusion.
Link to Abstract: Lactobacillus rhamnosus GG versus Placebo for Acute Gastroenteritis in Children
METHODS: We conducted a prospective, randomized, double-blind trial involving children 3 months to 4 years of age with acute gastroenteritis who presented to one of 10 U.S. pediatric emergency departments. Participants received a 5-day course of Lactobacillus rhamnosus GG … twice daily or matching placebo…
RESULTS Among the 971 participants, 943 (97.1%) completed the trial…There were no significant differences between the L. rhamnosus GG group and the placebo group in the duration of diarrhea (median, 49.7 hours in the L. rhamnosus GG group and 50.9 hours in the placebo group; P=0.26), duration of vomiting (median, 0 hours in both groups; P=0.17), or day-care absenteeism (median, 2 days in both groups; P=0.67) or in the rate of household transmission (10.6% and 14.1% in the two groups, respectively; P=0.16).
CONCLUSIONS Among preschool children with acute gastroenteritis, those who received a 5-day course of L. rhamnosus GG did not have better outcomes than those who received placebo
My take: While some probiotic strains have been shown to be helpful in some conditions (eg. antibiotic-associated diarrhea), this study indicates that probiotics are likely ineffective in altering the course of acute gastroenteritis.
Related blog posts:
- The Truth about Probiotics: Constipation Version
- Are Probiotics Effective in Changing the Microbiome?
- Lactobacillus rhamnosus GG Associated with Increased Necrotizing Enterocolitis in Observational Study
- How helpful are probiotics?
- Balanced summary of probiotics & Microbiome effects on brain
- Probiotics for Crohn’s Disease –No Beneficial Effects Noted | gutsandgrowth
- Probiotics For Fatty Liver Disease | gutsandgrowth
- “Low quality of evidence; strong recommendation” for Probiotics in Gastroenteritis
- Probiotics for Colic
- Do Probiotics Really Help Crying Infants? | gutsandgrowth
- Pushback on Probiotics
- One More Day Syndrome & Necrotizing Enterocolitis | gutsandgrowth
- Probiotics, Atopy, and Asthma | gutsandgrowth
Low Quality Evidence for IBS Dietary Therapy
A recent systematic review and meta-analysis (J Dionne et al. Am J Gastroenterol 2018; 113: 1290-1300) throws some shade on the effectiveness of dietary therapies for irritable bowel syndrome. Thanks to Ben Gold for this reference. The authors reviewed 1726 citations -only 9 were eligible for systematic review; two RCTs (n=111 participants) with gluten-free diet (GFD) and 7 RCTs (n=397) with low FODMAPs diet.
Key findings:
- A GFD was associated with reduced global symptoms compared with control interventions (RR=0.42, CI 0.11-1.55) which was not statistically significant. Thus, there is “insufficient evidence to recommend a GFD to reduce IBS symptoms.”
- A low FODMAP diet was associated with reduced global symptoms compared with control interventions (RR=0.69, CI 0.54-0.88). The three RCTs with rigorous control diets found the least magnitude of effect. Thus, the overall quality of the data was “very low” according to the GRADE criteria.
Given the limited data supporting dietary therapy for IBS, the authors caution that in those who are placed on a low FODMAPs diet, that after a 2-6 week trial, those who “fail to improve should not continue the diet. ”
Related blog posts:
- Another Study: Low FODMAP diet for IBS
- FODMAP diet -Real World Experience
- FODMAPs Advice From Harvard
- An Unexpected Twist for “Gluten Sensitivity” | gutsandgrowth
- Low-FODMAPs with or without Gluten-Free Diet in IBS …
- Mechanism for FODMAPs diet | gutsandgrowth
FDA IBD Workshop -Take-Home Points
From a previous blog lecture from Athos Bousvaros (NASPGHAN Postgraduate Course 2014)
Off-label does not equate to experimental
FDA Statement: The FD&C Act does not, however, limit the manner in which a physician may use an approved drug. Once a product has been approved for marketing, a physician may prescribe it for uses or in treatment regimens or patient populations that are not included in approved labeling. Such “unapproved” or, more precisely, “unlabeled” uses may be appropriate and rational in certain circumstances, and may, in fact, reflect approaches to drug therapy that have been extensively reported in medical literature.
Genotyping Still Matters with Hepatitis C
A recent study (R Esteban et al. Gastroenterol 20018; 155: 1120-7) evaluated the efficacy of sofosbuvir and velpatasvir in patients with hepatitis C genotype 3.
Overall, the study shows good efficacy of this regimen with and without ribavirin, though with higher SVR12 and lower relapse with the addition of ribavirin.
The difference in response was driven almost entirely based on whether there were pretreatment NS5A resistance-associated substitutions (RASs) present.
- In those with NS5A RASs the difference in response with added ribavirin compared to without was 96% vs 84%.
- In those without NS5A RASs the difference in response with ribavirin compared to without was 99% vs. 96%.
My take:
- If RAS testing is available and baseline Y93H is absent, then ribavirin is not likely needed
- Genotyping is still important. The associated editorial (pg 969-71) labeled genotype 3 ‘the problem child in the era of direct-acting antivirals.” That is, there are still differences in treatment recommendations based on HCV genotype.
Related blog posts:
A Bunch of Data on Vedolizumab
DE Yung et al. Inflamm Bowel Dis 2018; 24: 2327-38. This systematic review and meta-analysis of four studies “did not detect an increased risk of postoperative complications with preoperative vedolizumab” (VDZ). This study included 281 patients who received VDZ.
SC Ng et al. Inflamm Bowel Dis 2018; 24: 2431-41. The authors examined the frequency of opportunistic infection among 4 VDZ trials and postmarketing surveillance, accounting for ~114,000 patient-years of exposure. The most common infection was C difficile (0.5 per 100 patient-years); tuberculosis was reported at 0.1 per 100 patient years. This study showed “that the rate of serious opportunistic infections in patients receiving VDZ was low and most patients could continue VDZ treatment.”
SL Gold et al. Gastroenterol 2018; 155: 981-2. This clinical image showed a case of Henoch-Schonlein Purpura (HSP) that developed in a 53 year receiving VDZ.
E Shmidt et al. Inflamm Bowel Dis 2018; 24: 2461-7. This retrospective review of a prospectively maintained IBD registry provides information of risk factors for VDZ loss of response and management. 444 patients out of 788 who received VDZ had a significant response.The majority of VDZ recipients 75) had failed prior anti-TNF Rx. Key points:
- Loss of response (LOR) at 6 months and 12 months was 20% and 35% respectively
- UC patients compared to Crohn’s disease (CD) patients were more likely to have LOR with R of 1.54.
- Shortening VDZ infusion interval from q8 weeks to q4-6 weeks recaptured response in 49% and led to remission in 18% of this cohort.
- LOR was more common (2-fold) among those who had a LOR to anti-TNF agent. Patients with primary nonresponse were less likely to have LOR with VDZ.
U Kopylov et al. Inflamm Bowel Dis 2018; 24: 2442-51. This retrospective multicenter study examined VDZ effectiveness among anti-TNF naive patients, n=184.
- For CD, 42/50 (82%) responded by week 14, and 32 (64%) were in clnical remission. At last followup (30-52 weeks), clinical remission was noted in 24/35 (69%)
- For UC, 116/134 (79%) responded at week 14 and 53 (40%) were in clinical remission. At last followup (30-52 weeks), 67% were in remission (69/103)
The authors conclude that VDZ is similarly efffective for anti-TNF naive CD and UC patients.
My take: These studies show that we still have a lot to learn about the effectiveness of VDZ as its use becomes more widespread.
Related blog posts:
- Vedolizumab and Extraintestinal Manifestations
- VICTORY consortium showing good results for vedolizumab
- Summary of latest information on Vedolizumab
- GI Care for Kids Data on Vedolizumab 2017
- Latest on Vedolizumab
- Pediatric Experience with Vedolizumab | gutsandgrowth
- Vedolizumab -another new IBD treatment | gutsandgrowth