DC Deza et al. AJG 2025. 120:p 2644-2659. Probiotics Prescribed With Helicobacter pylori Eradication Therapy in Europe: Usage Pattern, Effectiveness, and Safety. Results From the European Registry on Helicobacter pylori Management (Hp-EuReg) Thanks to Ben Gold for this reference.
Methods: Prospective European registry with 36,699 treatments were recorded, where 8,233 (22%) were prescribed with probiotics. The analysis of the effectiveness of probiotics was restricted to those receiving first-line therapy.
Key findings:
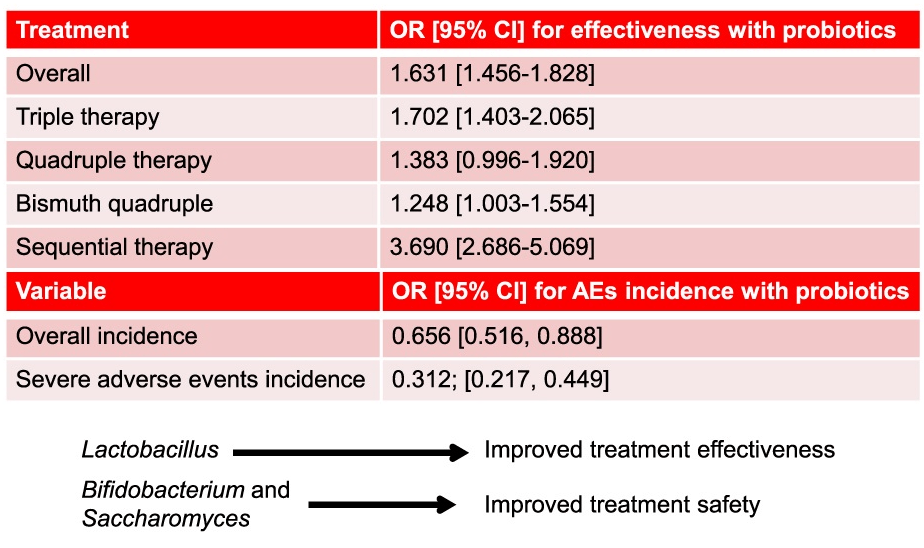
- Overall, the eradication rate was 90.6% with probiotics and 86.1% without probiotics.
- With quadruple therapy, the eradication rate was 93.1% with probiotics and 89.3% without probiotics
Discussion Points:
- “The prevalence of H pylori infection remains close to 45% in the European population”
- The population receiving probiotics may have been expected to have a higher risk of eradication failure (eg. antibiotic resistance and more prior treatments) and/or higher expectations of adverse effects
- This was NOT a randomized study which limits a more definitive conclusion on the effectiveness of adding a probiotic
My take: In highly-motivated families, probiotics may be worthwhile as part of an H pylori eradication regimen. This could, of course, necessitate changes in nomenclature. Would quadruple therapy with probiotics be called 5-drug (quintuple) treatment?
Related blog posts:
H pylori:
- 5 Rights and H pylori Treatment and H Pylori Study Methods
- Dr. Benjamin Gold: 2024 Pediatric H pylori Guidelines (Part One)
- Dr. Benjamin Gold: 2024 Pediatric H pylori Guidelines (Part 2)
- Give the Right Dose (for H pylori) -It Works Better!
- Next-Generation Treatment for H Pylori
Probiotics: