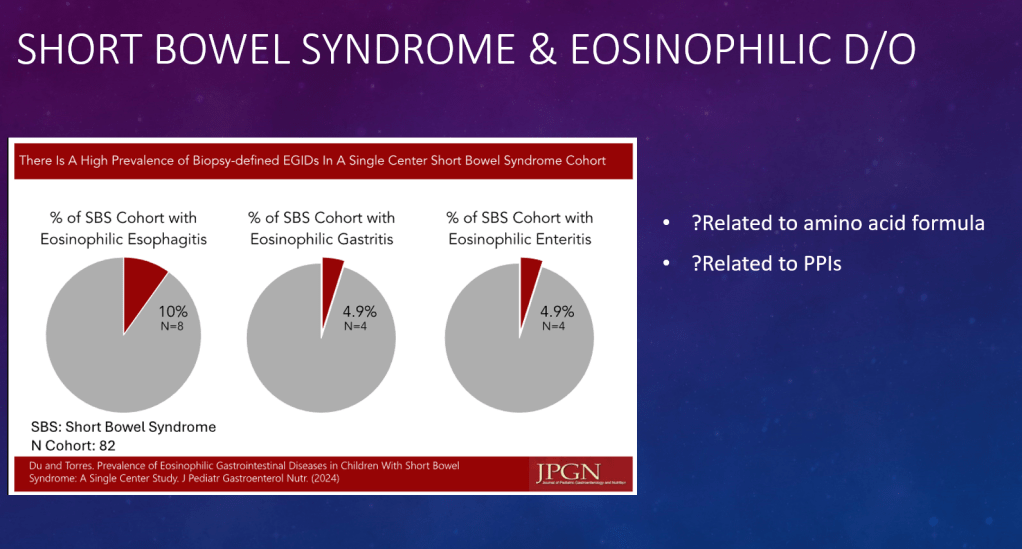
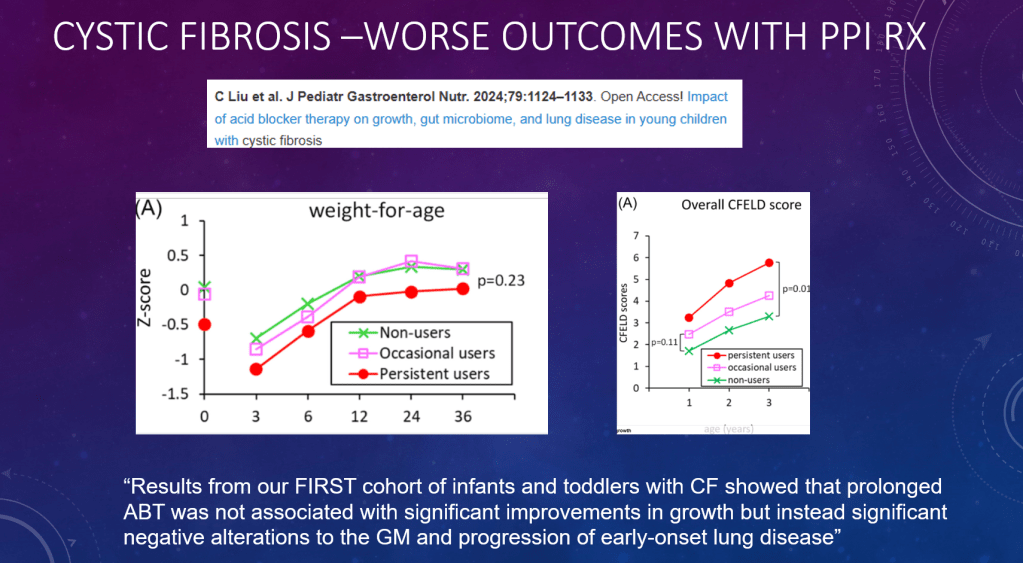
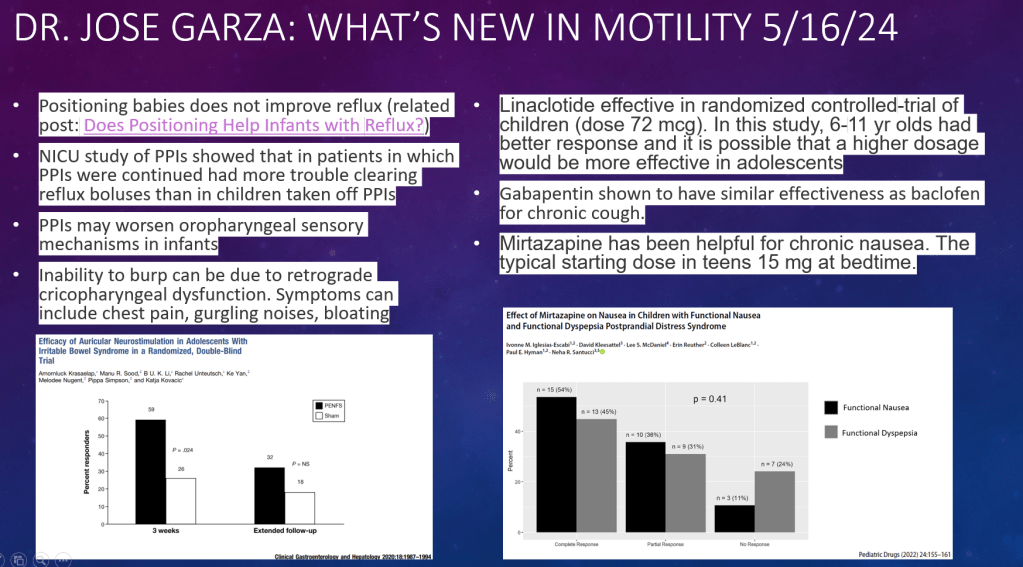
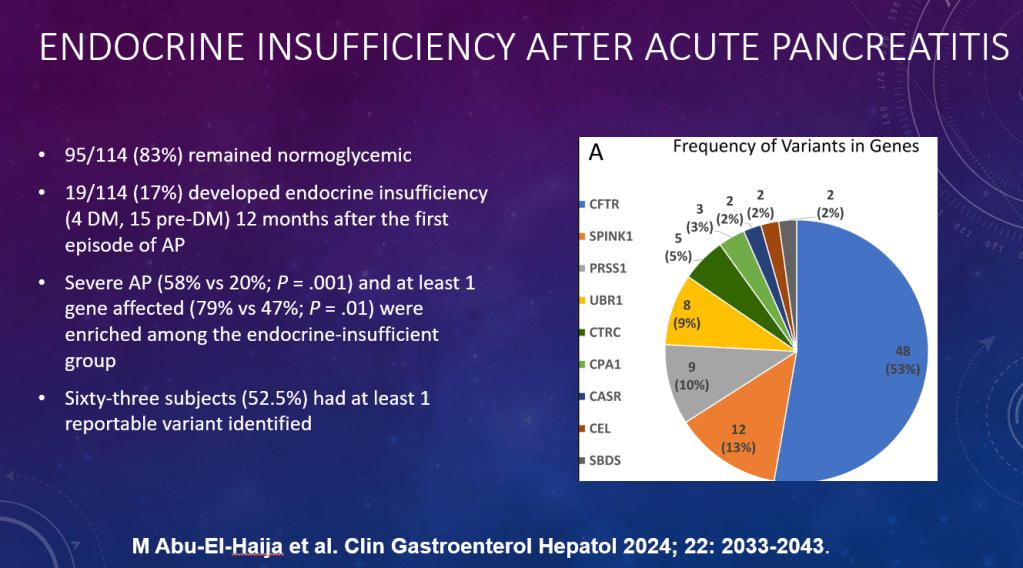
DR Shores et al. J Pediatr Gastroenterol Nutr. 2025;81:158–161. Navigating the blame game: Exploring necrotizing enterocolitis, preterm nutrition, and the ramifications of a formula shortage
Key points from this commentary:
- “Recent lawsuits involving preterm infant formula manufacturers in the United States (Abbott and Mead Johnson) … [a] formula shortage crisis … might ensue if these formulas are pulled from the market”
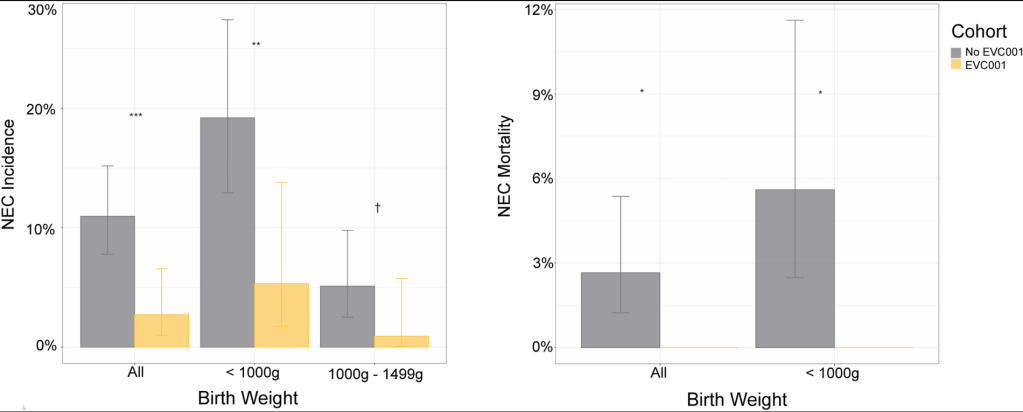
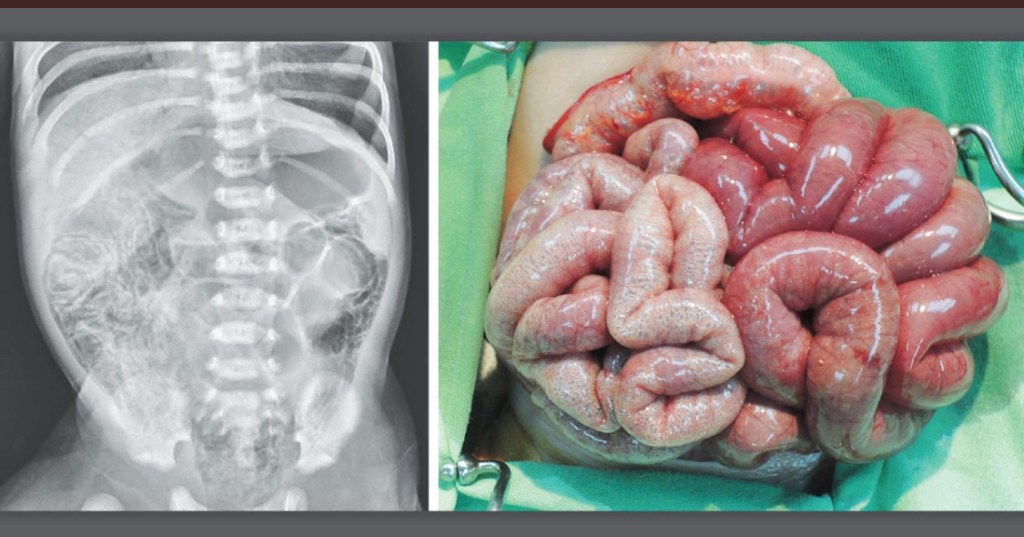
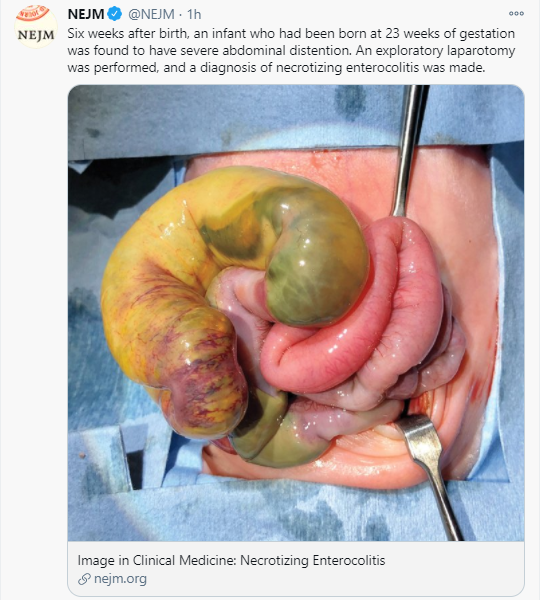
- “Breast milk has long been associated with protection against NEC, possibly due to bioactive molecules that maybe protective. However, infants fed exclusively breastmilk can still develop NEC, highlighting a multifactorial etiology. Mother’s own breast milk provides the best protection against NEC and is the ideal primary form of nutrition for preterm infants…pasteurized donor breast milk provides some protection against NEC and should be offered if mother’s own breast milk is not available”
- “Although preterm formula does not protect against NEC, it is the healthcare standard, assuming all medical comorbidities are taken into consideration for individualized care…preterm infant formulas that are specifically formulated to provide the calories, protein and micro-nutrient needs of preterm infants”
- “Placing all the blame for the development of NEC on preterm formula manufacturers is not supported by science and is thus unfounded”
- “For infants without access to mother’s own or donor breast milk, specialized formulas remain critical to their nutrition and survival….Vilifying formula companies risks provoking further shortages of essential specialized formulas, which could directly harm the very babies we all aim to protect”
Related blog posts:
- Proliferation of Formula Lawsuits In Necrotizing Enterocolitis
- Where Will We Be Without Formulas for Premature Babies?