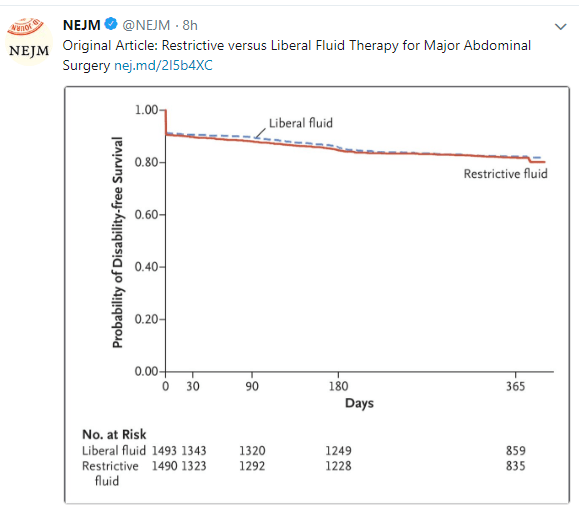
A new report from an ESPGHAN-Led Expert Group (N Thapar et al. JPGN 2018; 66: 9991-1019) provides detailed recommendations for pediatric intestinal pseudo-obstruction (PIPO). In addition, this report serves as an excellent self-assessment of your vision. If you can read figure 1, which has some incredibly tiny font size, then your vision is fantastic.
Full Link: “Paediatric Intestinal Pseudo-obstruction: Evidence and Consensus-based Recommendations From an ESPGHAN -Led Expert Group”
Aside from that snarky comment, the report offers a great deal of useful advice.
- After obstruction has been excluded, the authors recommend that patients should undergo a basic laboratory evaluation (including CBC, CMP, ESR/CRP, Celiac serology, Cortisol, Thyroid testing, Metabolic tests [urine organic acids, ammonia, lactate]) and to consider more extensive evaluation.
- If primary, rather than secondary, PIPO is suspected, the authors recommend neurogastroenterology evaluation.
Subsequently, the authors review management: potential medications (Table 6), enteral feeds, gastrostomy and ileostomy, and in more than 80% then need for parenteral nutrition. At the time of therapeutic procedures, it is recommended to obtain full-thickness biopsies to further characterize the PIPO.
Clinical features which distinguish pediatric chronic intestinal pseudo-obstruction (CIPO) from adult CIPO are listed in Table 2. These include the following:
- Frequent urologic involvement in pediatric CIPO which is rare in adults with CIPO.
- Dilated bowel loops are commonly absent (~40%) in pediatric CIPO in the neonatal period and universal in adult cases.
- Unlike in adults, there is a high risk of colonic and small bowel volvulus in pediatric CIPO and malrotation is evident in ~30% of pediatric CIPO (rarely seen in adults).
- Also, in pediatrics, fabricated cases are more commonly encountered.
Intestinal transplantation should be considered in patients with PIPO who develop life-threatening complications associated with TPN or poor quality of life/high morbidity.
Pictures below from yesterday’s Peachtree Road Race and previous T-shirts from previous years.




Disclaimer: These blog posts are for educational purposes only. Specific dosing of medications/diets (along with potential adverse effects) should be confirmed by prescribing physician/nutritionist. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.