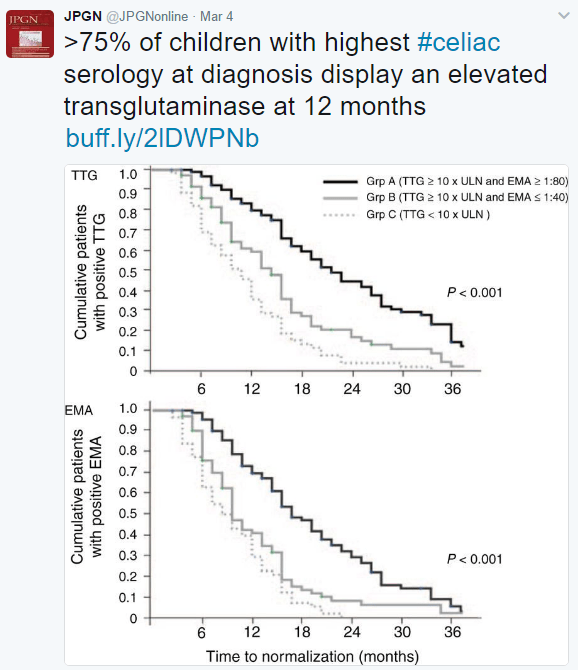
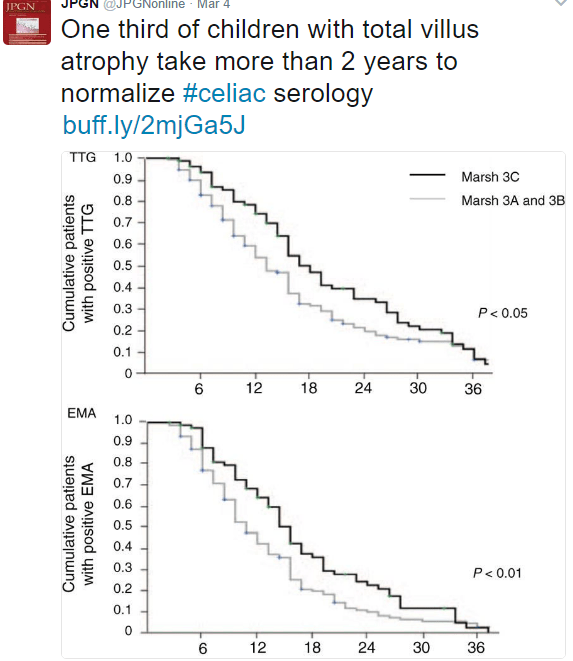
A recent study (JA Murray et al. Gastroenterol 2017; 152: 787-98) examined the effectiveness of latiglutenase for celiac disease. Latiglutenase (aka ALV003) is an oral medicine which is a mixture of two recombinant gluten-targeting proteases.
The concept of latigluenase is that a medicine that degrades the gluten protein could obviate the need for a gluten free diet. Unfortunately, in this study with 494 patients with celiac disease for at least 1 year, the medicine at various doses for 12 to 24 weeks was ineffective. There was no difference between the medicine and placebo with regard to villous height:crypt depth ratio, number of intraepithelial lymphocytes or serologic markers of celiac disease. Symptom scores increased in both the active treatment group and the placebo group. While this was a negative study, the authors did note some effect on symptom domains on higher dosing regimens. “This observation suggests that treatment with latiglutenase may affect symptoms before showing clinically meaningful effects on serologic and histologic end points.
A second study (RS Choung et al. Gastroenterol 2017; 152: 830-9) examined prevalence and morbidity of undiagnosed celiac disease in Olmstead County. After excluding patients with celiac disease, sera from 30,425 adults and 830 children were tested for tissue transglutaminase IgA antibody (tTG) and endomysial antibody (EMA). Case definition: patients were considered to have celiac serologically if tTG titer was 2.0 U/mL or greater with a positive EMA. The prevalence of celiac disease was 1.1% in adults and 1.0% in children. The majority of patients with celiac disease (>80%) have not received the diagnosis. By comparing those with positive celiac serology to matched controls (2 controls for each positive), the authors determined that undiagnosed celiac disease was associated with increased rates of hypothyroidism (OR 2.2) but no other significant morbidities. Median followup period was 6.3 years.
My take: A promising new therapy for celiac disease, latiglutenase, looks like it will not be effective and there are a lot of individuals with celiac disease who are unaware of their diagnosis.
Related blog posts:
- New Therapy for Celiac Disease: Larazotide Acetate
- Good Educational Two Minute Celiac Video | gutsandgrowth
- Are followup biopsies necessary in celiac disease: Looking beyond the Headline
- How Accurate is Serology at Predicting Mucosal Healing in Celiac Disease
- Drug Therapy for Celiac Disease | gutsandgrowth
- Why is Celiac Disease Becoming More Prevalent? | gutsandgrowth
- Benefits of Gluten–Free Diet for “Asymptomatic” Celiac | gutsandgrowth
- Closer followup for Celiac disease & pediatric … – gutsandgrowth
Disclaimer: These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.