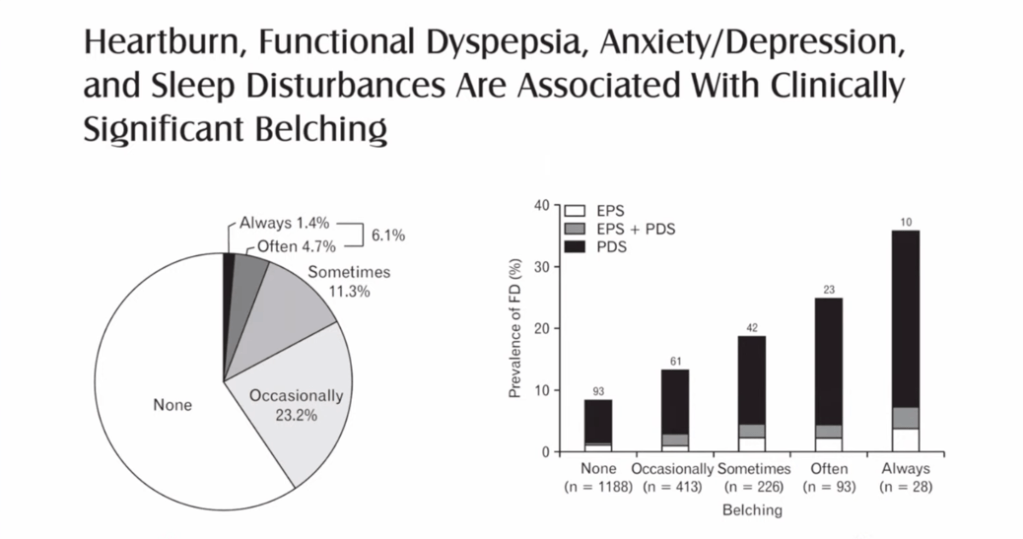
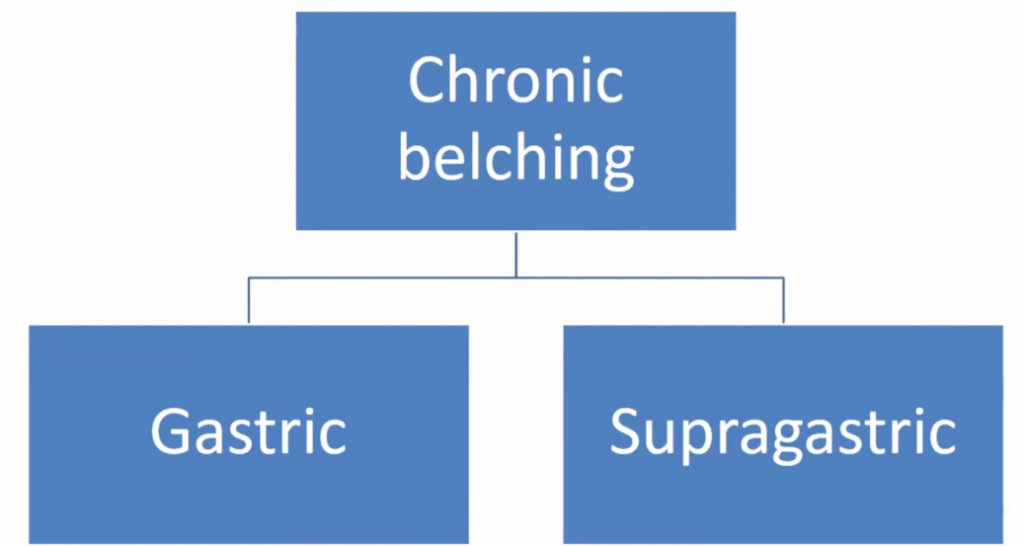
We had a brilliant lecture given to our group by Dr. Benjamin Gold. I have had the good fortune of getting to know Ben and working alongside Ben for more than 15 years. Most readers of this blog are very familiar with Dr. Gold who is a leader in our field.
My notes below may contain errors in transcription and in omission.
Guidelines:



- Bismuth-based quadruple therapy recommended when antimicrobial sensitivity testing (AST) is not available
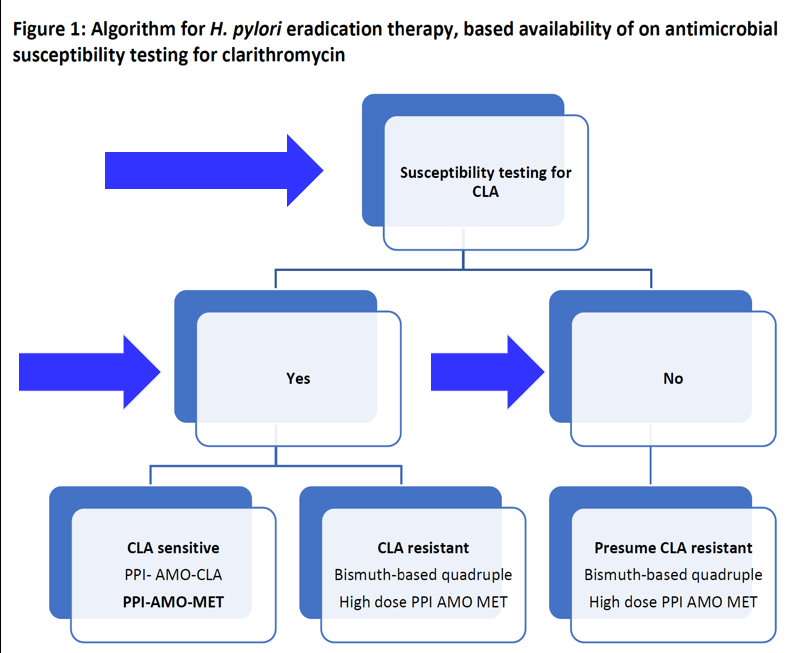
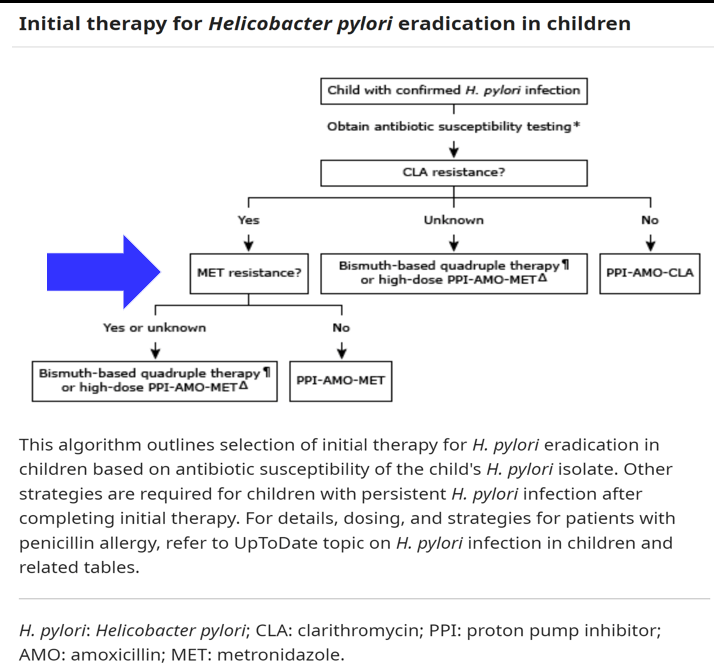


- Routine use of CLO test is NOT recommended during endoscopy
- Routine testing for H pylori is NOT recommended for children with recurrent abdominal pain

- Stool PCR testing is NOT recommended
- Test for cure should be done at 6-8 weeks after completion of treatment

During endoscopy at CHOA in which H pylori is suspected, complete a microbiology form and ask for a culture to arrange for resistance testing. Submit a sample (or multiple) in a sterile tube/cup. Completed results will include clarithromycin sensitivity. Additional testing for other antibiotic resistance can be requested subsequently. Testing can be done with paraffin block as well.
Related blog posts:
- Give the Right Dose (for H pylori) -It Works Better!
- Helicobacter Pylori Stool Susceptibility in Children –How Good Is It?
- Understanding FDA Approval of Vonoprazan-Based Therapies for Helicobacter Pylori
- Synergistic Dangers: Helicobacter Pylori and Cancer Genes
- AGA: Best Practice Advice for Refractory H pylori
- Treating Helicobacter Pylori Lowers The Risk of Gastric Cancer
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.