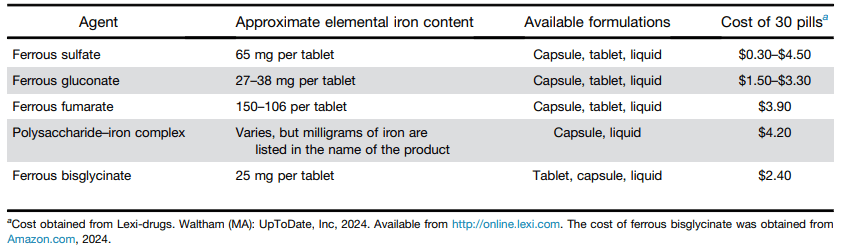
U Cucinotta et al J Pediatr 2024; https://doi.org/10.1016/j.jpeds.2023.113882. The Impact of Teduglutide on Real-Life Health Care Costs in Children with Short Bowel Syndrome
It is disappointing that in this study, the authors conclusions in the abstract state the following: “Treatment with teduglutide is associated with a significant reduction in the annual costs of HPN but still remains expensive because of the drug itself.”
Key findings from the study do NOT support this concclusion:
- In the treated group, the median total costs of home parenteral nutrition (HPN) significantly decreased after 1 (P < .001) and 2 years of treatment (P < .001) from 59,454 euros/year/patient to 43 885 euros/year/patient and 34,973 euros/year/patient, respectively
- When we compared patients treated and not treated, the total HPN costs/year/patient were similar at baseline (P = .6) but were significantly lower in the teduglutide-treated group after 1 (P = .006) and 2 years of treatment (P < .001)
- When we added the cost of teduglutide into the analysis, the total cost increased significantly in the treated group and remained much greater even after modeling a reduction in the cost of the drug to one-third the present cost and PN weaning (P < .001).
The study’s conclusion is like someone trying to tell me they saved money at a fancy restaurant if they just took the caviar off the bill. Guess what –it wasn’t less expensive!
My take: The conclusion from this article should be straight-forward: the costs were much greater in patients receiving teduglutide. It will remain more expensive even if the drug costs improve quite a bit. In addition, there are other additional costs of teduglutide if one follows the monitoring recommendations.
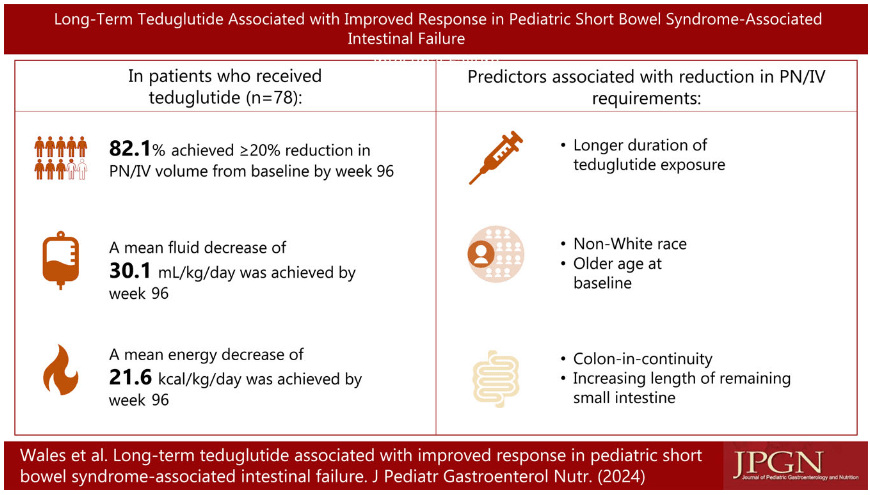
Related study: PW Wales et al. J Pediatr Gastroenterol Nutr. 2024;79:290–300. Long-term teduglutide associated with improved response in pediatric short bowel syndrome-associated intestinal failure. Safety concerns in this study (n=78 treated patients): out of 12 patients with positive antidrug antibodies, neutralizing activity was detected in four patients. There were no reported incidences of colorectal polyps
Related blog posts:
- Teduglutide-Induced Polyps
- Practical Intestinal Rehabilitation (Part 2)
- Is GLP2 Worth $300K per Year? | gutsandgrowth