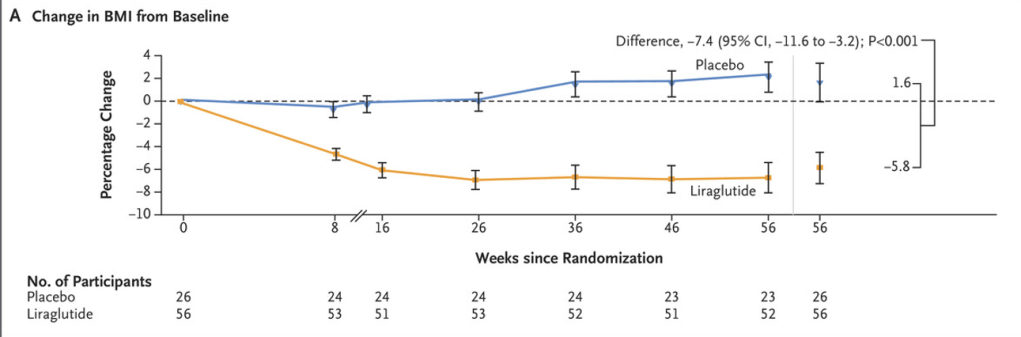
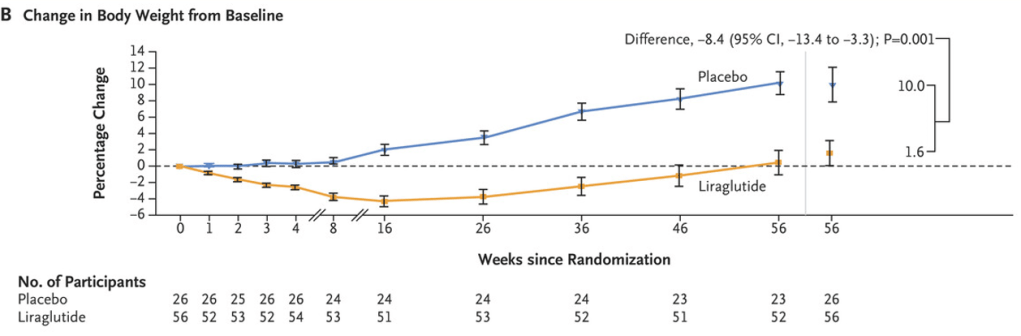
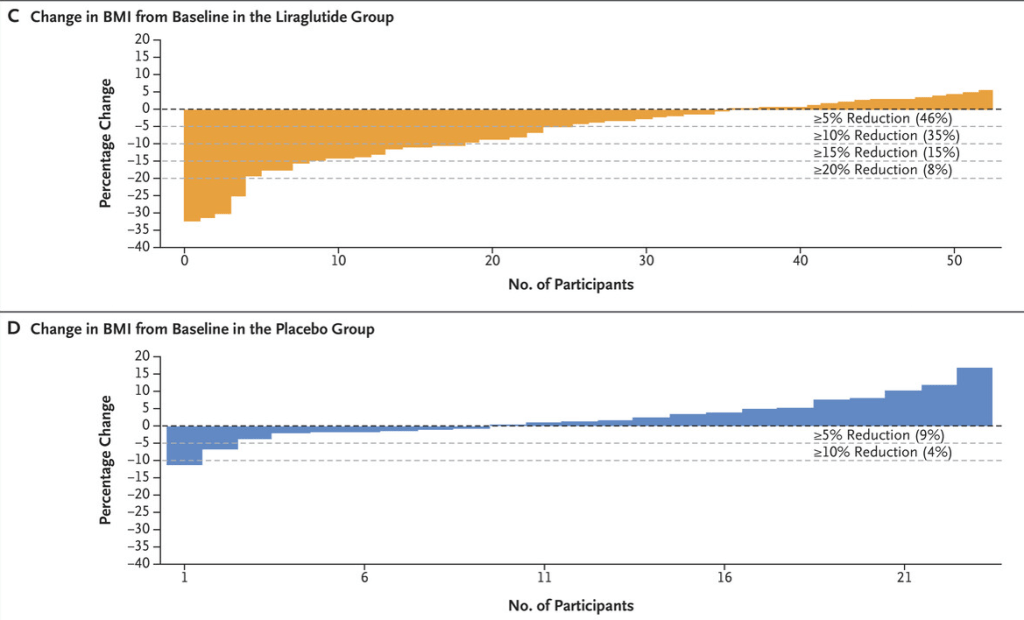
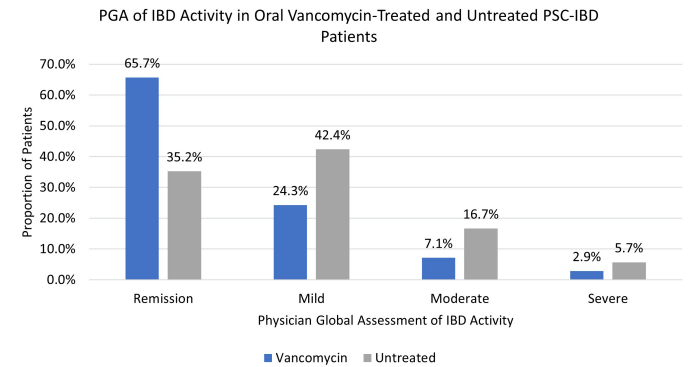
M Xue et al. Clinical Gastroenterology and Hepatology, Volume 22, Issue 9, 1889 – 1897.e12. Open Access! Environmental Factors Associated With Risk of Crohn’s Disease Development in the Crohn’s and Colitis Canada – Genetic, Environmental, Microbial Project
Methods: The authors prospectively followed 4289 healthy first-degree relatives (FDRs) of patients with CD from the Crohn’s and Colitis Canada – Genetic, Environmental, Microbial (CCC-GEM) project. Regression models identified environmental factors associated with future CD onset and their association with pre-disease biological factors, including altered intestinal permeability measured by urinary fractional excretion of lactulose to mannitol ratio (LMR); gut inflammation via fecal calprotectin (FCP) levels; and fecal microbiome composition through 16S rRNA sequencing.
Key findings:
- Over a 5.62-year median follow-up, 86 FDRs developed CD
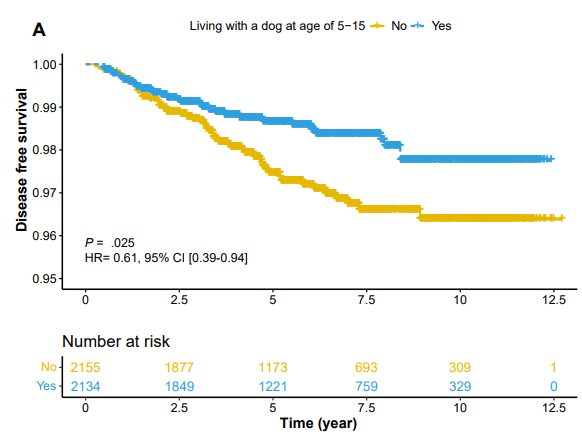
- Living with a dog between ages 5 and 15 (hazard ratio [HR], 0.62; P = .034) was associated with decreased CD risk
- Living with a dog was associated with reduced LMR, altered relative abundance of multiple bacterial genera, and increased Chao1 diversity.
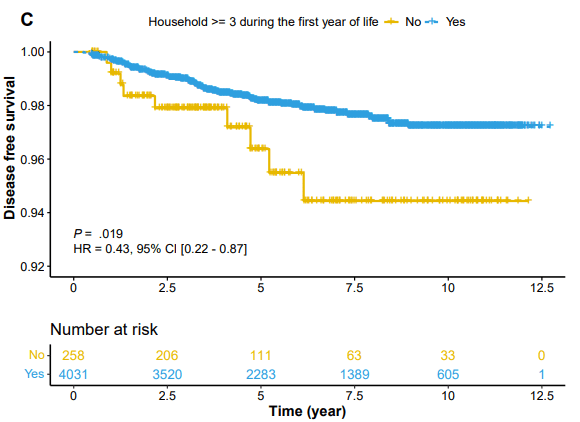
- Living with a large family size in the first year of life (HR, 0.43; P = .016) was associated with decreased CD risk
- Having a bird at the time of recruitment (HR, 2.78; P = .005) was associated with an increased CD risk (though there were relatively few FDRs with birds at baseline, n=136)
- Limitations: questionnaire-based assessments of environmental exposure can be subject to recall bias
My take (borrowed from authors): “Our findings contribute to the growing evidence supporting the potential health benefits of exposure to pets, particularly dogs, as a potential preventive strategy for individuals at risk of developing CD.” Having a dog during childhood may reduce the later risk of CD by ~40%.
Related blog posts:
- Risk Factors for Inflammatory Bowel Disease: Ultra-Processed Food (Part 1)
- Ultraprocessed Food and the Risk of Inflammatory Bowel Disease
- Risk Factors for Inflammatory Bowel Disease: Antibiotics (Part 2)
- Early Antibiotics -Minimal Risk for Crohn’s Disease
- Passive Smoking and Worsening Crohn’s Disease
- Emigration -One Way to Acquire Inflammatory Bowel Disease | gutsandgrowth