K Karrento et al. JPGN 2023; 77: 347-353. Percutaneous Electrical Nerve Field Stimulation for Drug-Refractory Pediatric Cyclic Vomiting Syndrome
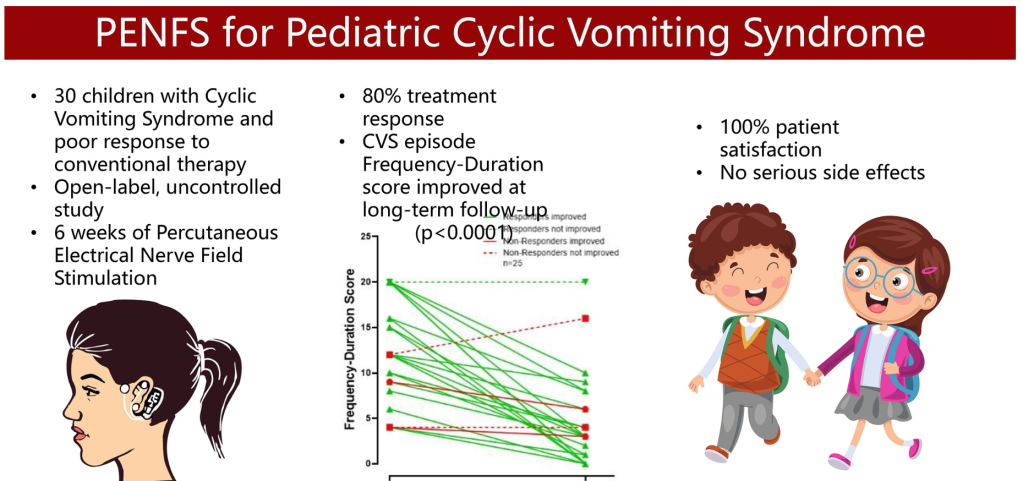
In this prospective study with 30 children with drug-refractory CVS, response was classified as ≥50% improvement in either frequency or duration of attacks at extended follow-up.
Key findings:
- At follow-up, 80% met criteria for treatment response with a median (IQR) response duration of 113 (61–182) days.
- At end of therapy, 66% and 55% patients reported global response of at least “moderately better” and “a good deal better,” respectively.
- There were no serious side effects.
Limitations: episodic nature of CVS, no control group, hx/o strong placebo control group and lack of validated assessment tool
My take: 1. As with the drug therapies with CVS, it is difficult to know how effective PENFS is for CVS. At the same time, its good safety profile makes this therapy an intriguing option in those not responding to more typical treatments. 2. The visual abstract is funny -mainly due to the two laughing kids holding hands. Though it would be even better if the person on the left was puking a lot instead of having a PENFS device.
Related blog posts:
- Abraham Lincoln’s Cyclic Vomiting Action Plan
- Cyclic Vomiting ED Protocol
- Topiramate -2nd Line Agent for Cyclic Vomiting Syndrome
- Does Stopping Cannabis Improve Cyclic Vomiting Syndrome?
- Aprepitant for CVS
- Getting In the Shower for Emetic Symptoms
- Costs/Yield of Diagnosing Cyclic Vomiting Syndrome
- Diet or drugs for cyclic vomiting syndrome