R Bolia et al. J Pediatr Gastroenterol Nutr. 2025;81:262–265. Open Access! Impact of occult spinal abnormalities on clinical outcomes in children with refractory constipation
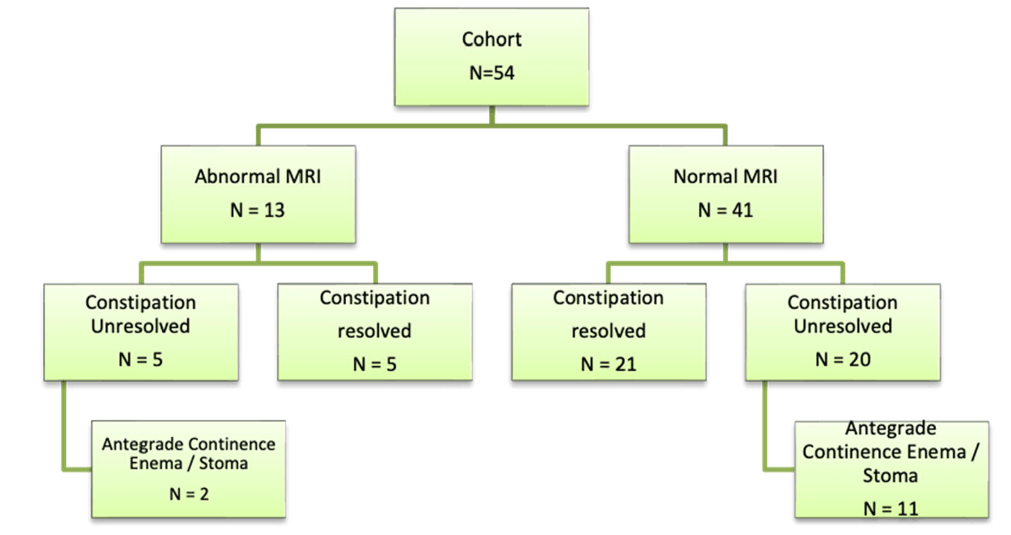
Methods: This was a retrospective review of records of children (n=54, median age 9 years) who underwent MRI of spine between January 2021 and December 2023 for evaluation of refractory constipation (RC). RC was defined as constipation not responding to optimal conventional treatment for at least 3 months. Conventional treatment included—education, disimpaction (if required), osmotic and stimulant laxatives, timed‐toileting and biofeedback.
Key findings:
- Thirteen children (24%) had an abnormal MRI. Findings included—syringomyelia‐8,sacral canal meningeal cyst‐2, filum terminale lipoma‐1,spina bifida occulta‐1 (SBO‐1), and Schmorl’s node‐1. None of these patients had a tethered cord
- Only one patient with a Chiari malformation and syrinx required a neurosurgical intervention. The surgery did not improve his constipation
- On a median follow‐up duration of 677 (range181–1240) days, constipation resolved in 48% (n = 26) of the entire cohort
- There was no difference in the number of patients or time to constipation resolution between those with and without abnormal MRI respectively
My take: In children with refractory constipation who do not have abnormal cutaneous/abnormal neurological exam, an MRI is unlikely to be helpful.
Related blog posts:
- Position Paper: Pediatric Refractory Constipation Management
- Diagnostic tests hardly ever help patients poop
- ACE report -10 year effectiveness
- Is There a Residual Impact of a Tethered Cord on Colonic Motility
- Retrospective Study on Botulinum Toxin for Refractory Constipation in Children
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition