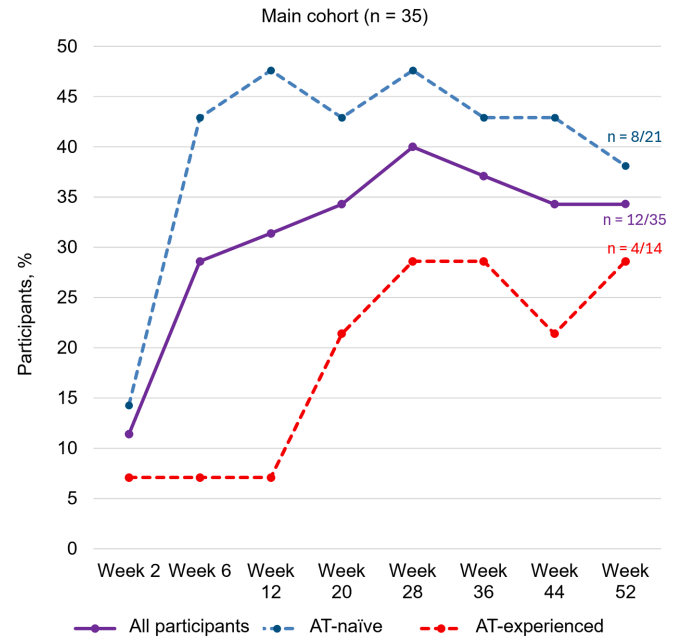
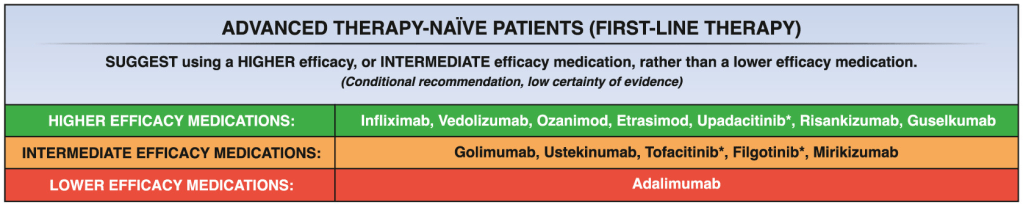
J Blackwell et al. Gastroenterol 2026; 170: 452-455: Open Access! A Normal UCEIS Is Zero: A Score Divided by a Common Language
“The index uses a Likert scale of vascular pattern, bleeding, erosions and ulcers, with a total score intended to reflect severity of disease. When the index was first published, the baseline score for each descriptor was 1, meaning that a normal vascular pattern, no bleeding, and no erosions or ulcers scored 3. However, when the index was subsequently validated, the values attributed to each descriptor were rebased to 0, to improve clinical utility…This means that a completely normal flexible sigmoidosopy scored 0 rather than 3, and the worst activity of UC ever seen by the investigators (compared with a visual analogue scale 0 to 100) was 8 rather than 11…
The UCEIS has since been shown to predict the need for escalation of medical treatment and has recently been incorporated into a validated prognostic clinical index to predict response to intravenous steroids among patients with acute severe UC.10,11 Accurate scoring is therefore essential, with direct implications for clinical decision making.”
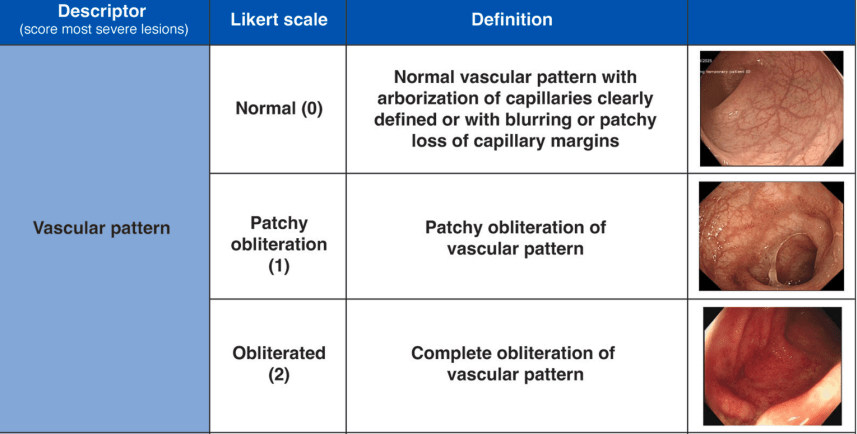

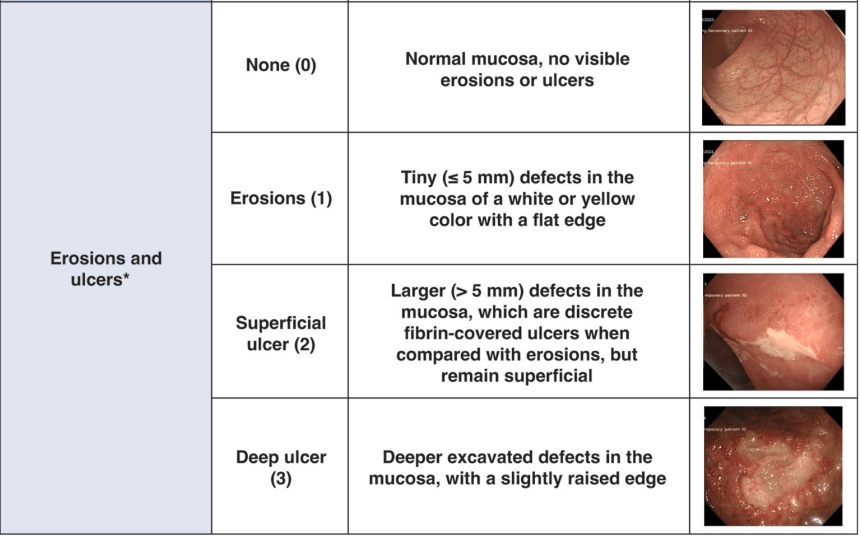
My take: The UCEIS is more detailed than the Mayo score. However, I expect that before long artificial intelligence will be able to review images and give a more consistent interpretation of the severity of endoscopic findings than either of these scoring systems.
Related blog posts:
- Comprehensive ACG Clinical Guidelines for Ulcerative Coliits (2025) (includes UCEIS and Mayo Score)
- Validated SEMA-CD Score For Crohn’s Disease
- Dr. Jennifer Lee: AI for Peds GI
- AI for GI
- Pediatric Guidelines for Ulcerative Colitis (Part 1) (ESPGHAN 2025)
- Dr. Joel Rosh: Positioning Therapies for Pediatric Ulcerative Colitis (2024)
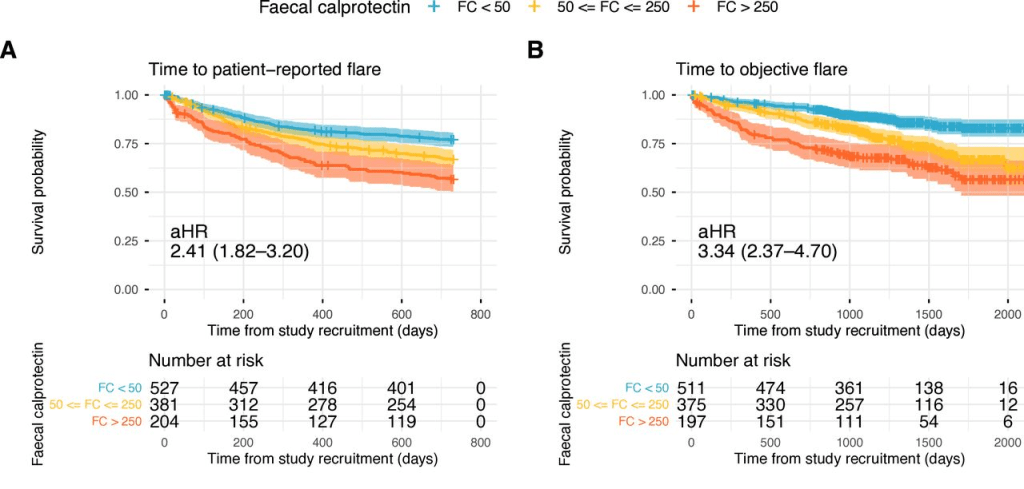
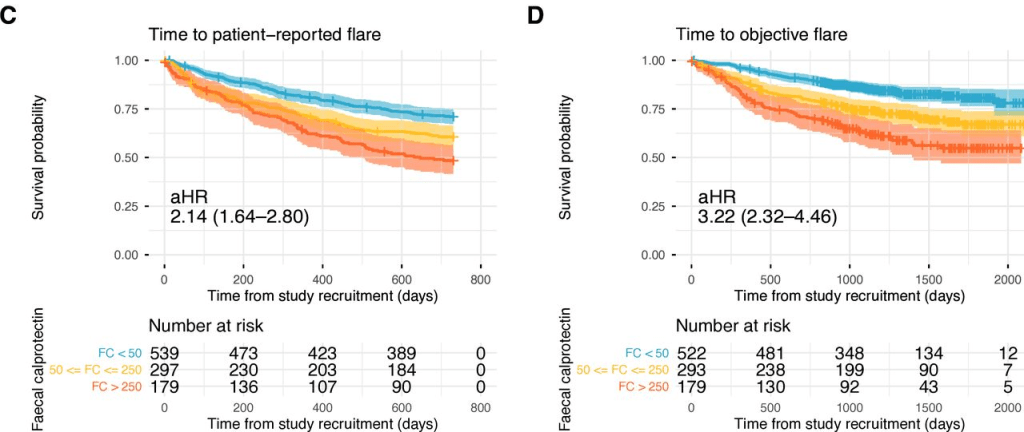
- Calprotectin Correlates with Disease Extent and Mucosal Healing in Ulcerative Colitis (2026)