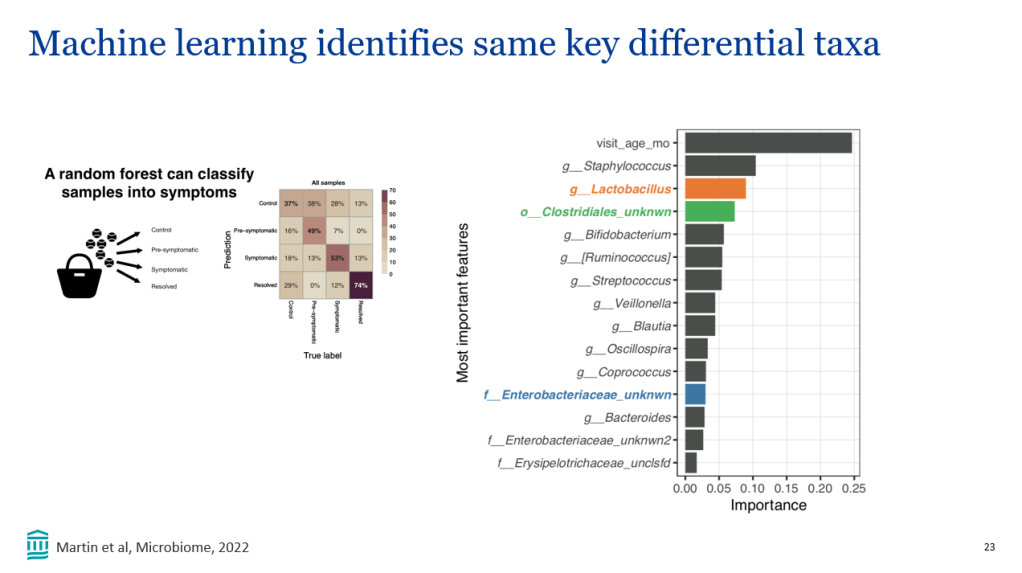
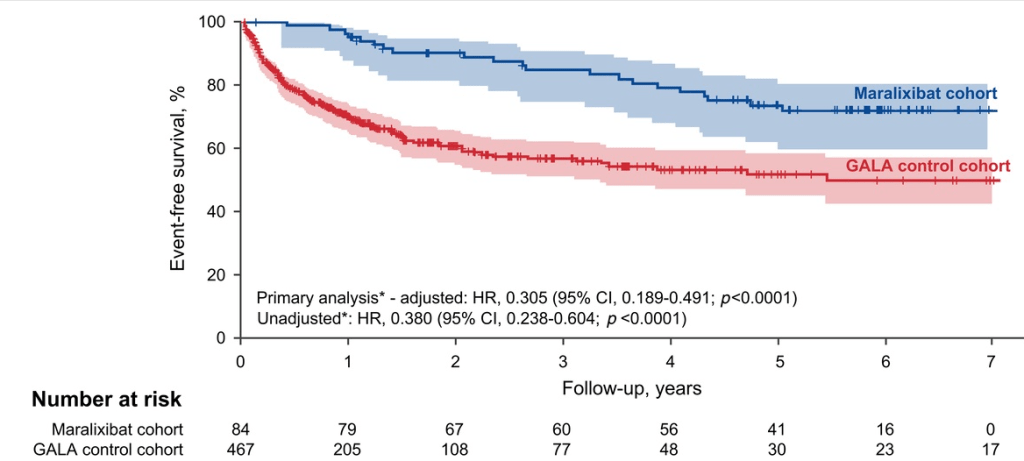
BE Hansen et al. Hepatology 2024; 79: 1279-1292. Open Access! Event-free survival of maralixibat-treated patients with Alagille syndrome compared to a real-world cohort from GALA
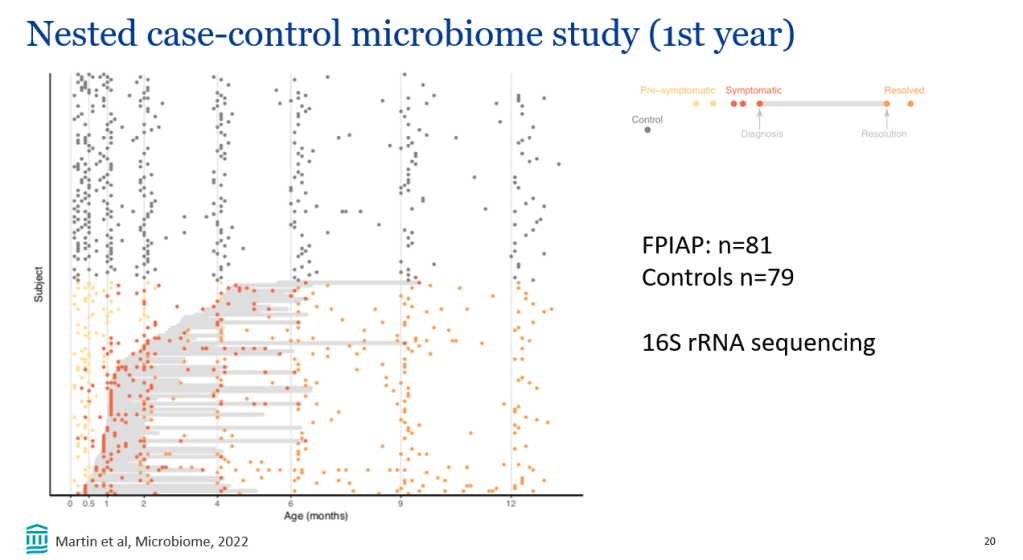
This study compared “6-year outcomes from maralixibat trials with an aligned and harmonized natural history cohort from the Global ALagille Alliance (GALA) study.”
Based on a quick review, some the data appears to overlap a recent report in the same journal: RJ Sokol et al. Hepatology 2023; 78: 1698-1710. Open Access! Predictors of 6-year event-free survival in Alagille syndrome patients treated with maralixibat, an ileal bile acid transporter inhibitor (See blog post: Six Year Data for IBAT Inhibitor Treatment for Alagille Syndrome).
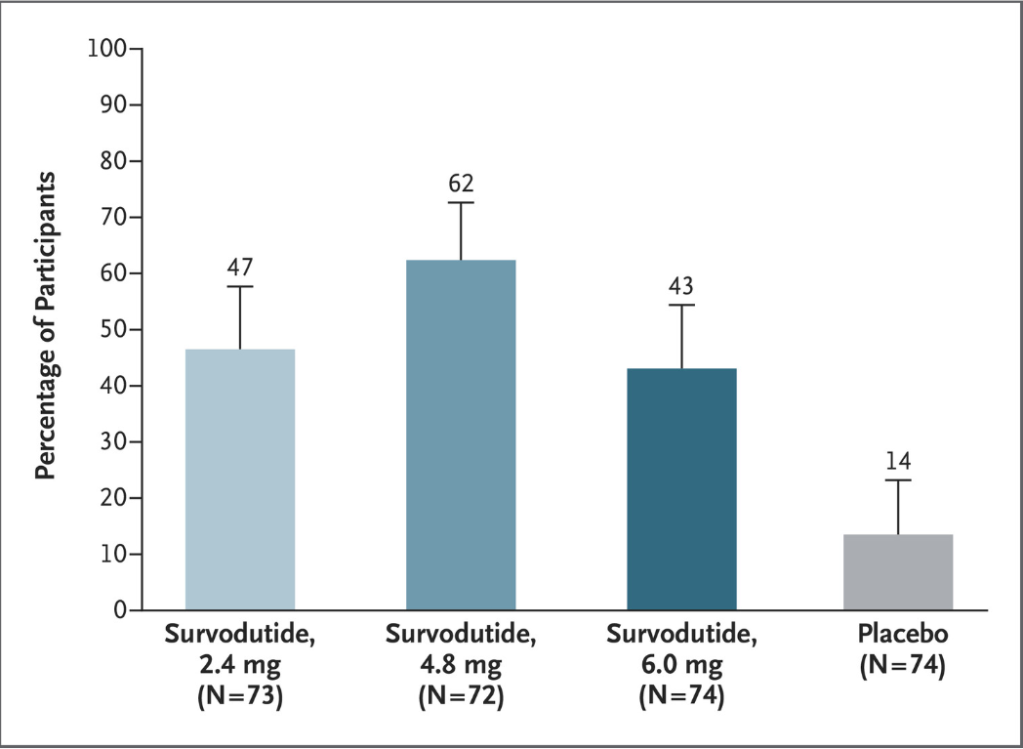
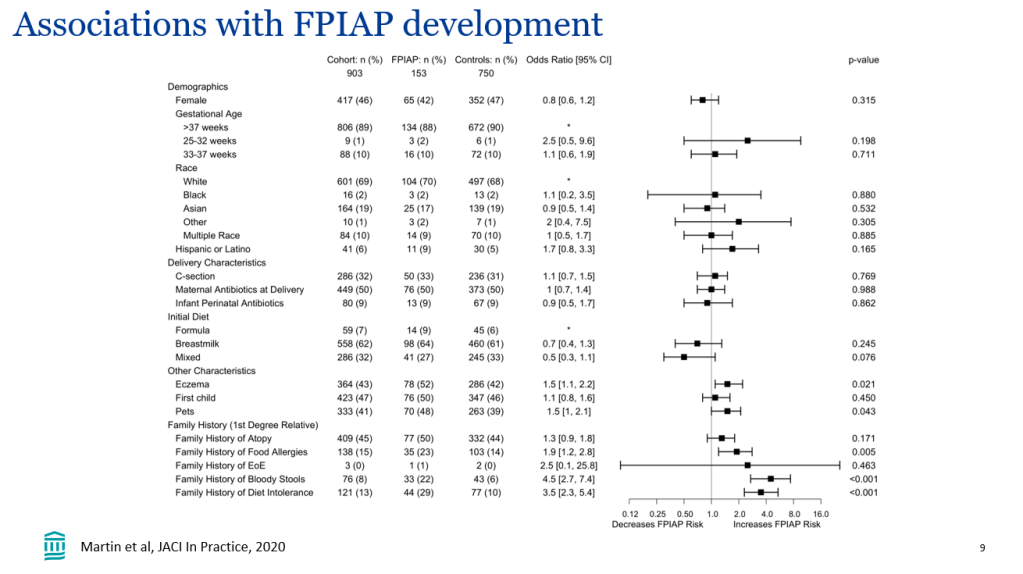
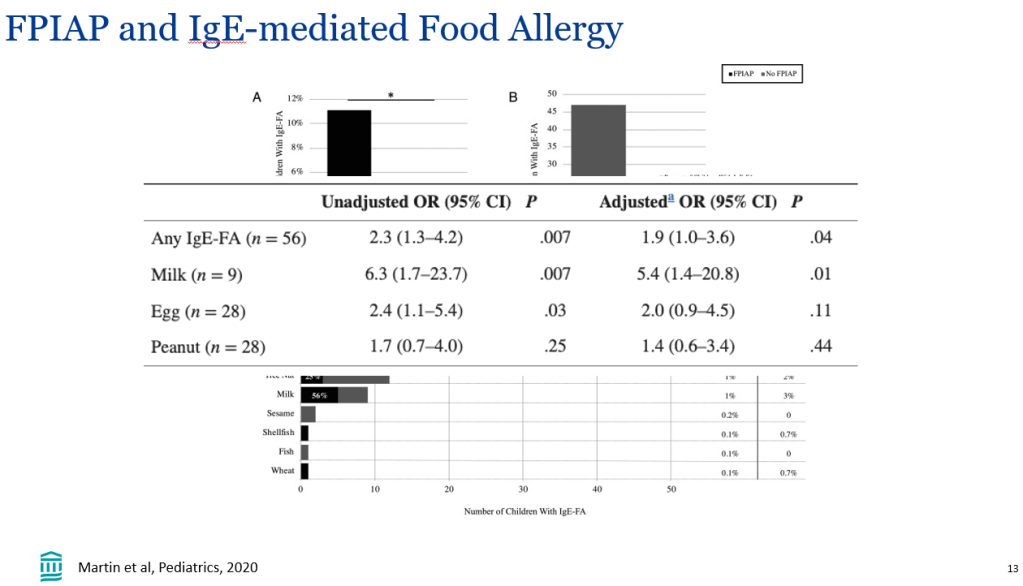
In the current study, “event-free survival, defined as the time to first event of manifestations of portal hypertension (variceal bleeding, ascites requiring therapy), surgical biliary diversion, liver transplant, or death, was analyzed by Cox proportional hazards methods.”
Key findings:
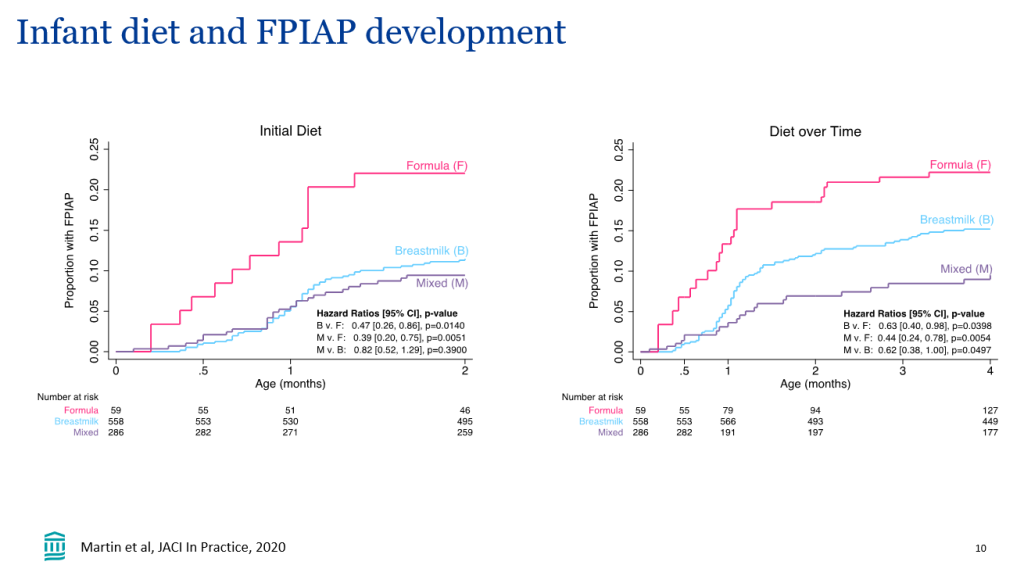
- Event-free survival in the maralixibat cohort (n=84) was significantly better than the GALA cohort (n=469) (HR, 0.305)
- Transplant-free survival showed similar results (aHR, 0.33)
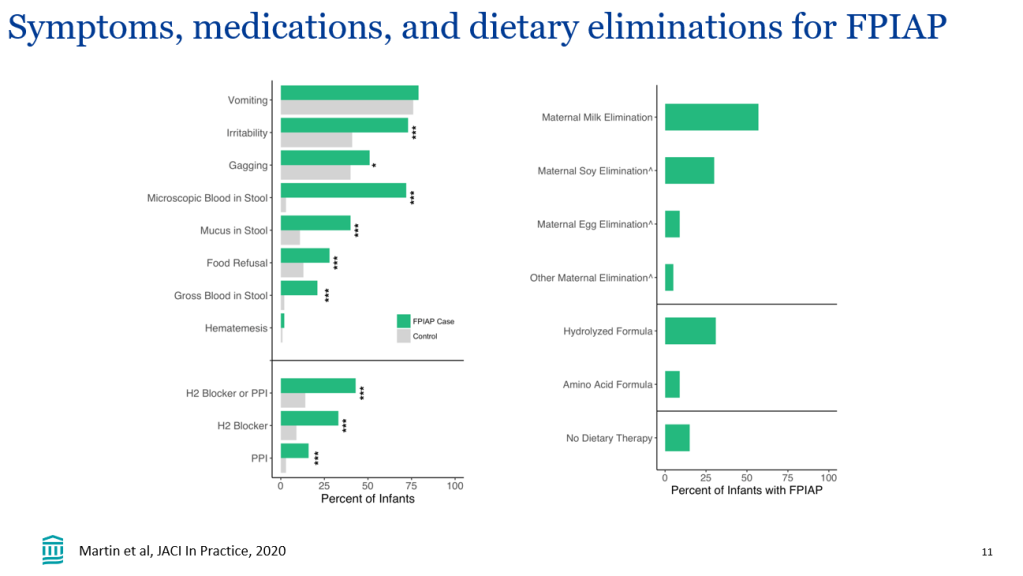
In their discussion, the authors note that much of the improvement in event-free survival is due to improvement in pruritus which is a main indication for liver transplantation. They speculate that improvement in event-free survival is also related to more broad-based clinical improvement (observed in ICONIC study), perhaps due to reduction in retained hepatic bile acids.
One of the limitations, reliance on a historical control, is discussed. “Historical control comparison is useful when there are ethical concerns regarding the recruitment of patients for long-term control arms requiring several years of study in life-threatening or debilitating diseases.”
My take: In this real-world comparison, Maralixibat, clearly was associated with improved outcomes. How much of this was due to relief of intractable pruritus and how much of this may be due to other biologic factors remains uncertain.
Related blog posts:
- Six Year Data for IBAT Inhibitor Treatment for Alagille Syndrome
- Lecture: IBAT Inhibitor for Alagille Syndrome
- GALA: Alagille Study
- Liver Briefs: HLH in Infancy, Maralixibat for Alagille Syndrome, Liver Disease Due to Inborn Errors of Immunity
- NASPGHAN Alagille Syndrome Webinar
- Intracranial Hypertension & Papilledema with Alagille Syndrome
- Explaining Differences in Disease Severity for Alagille Syndrome