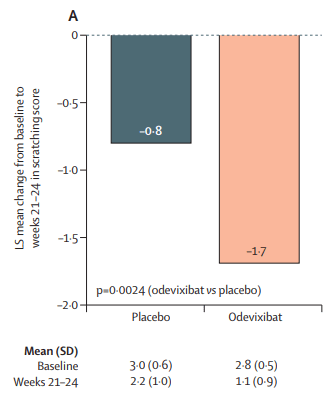
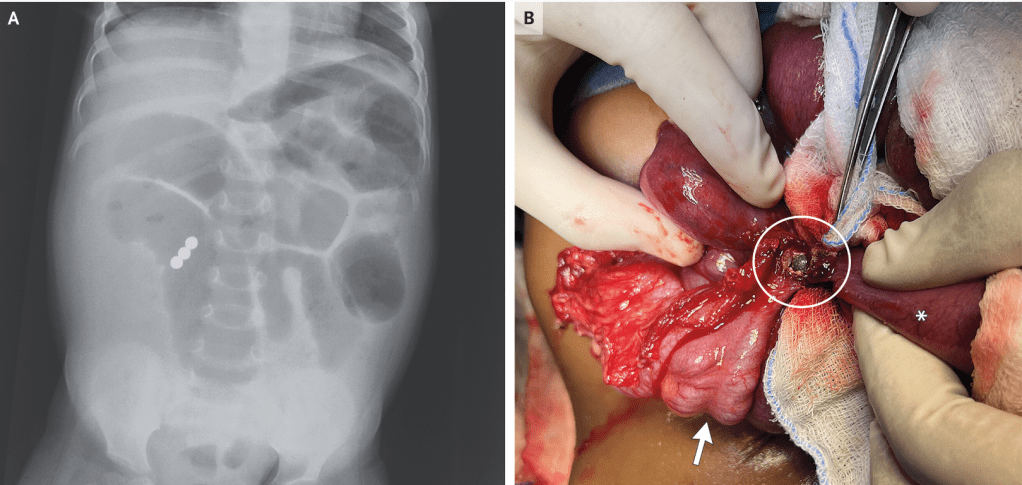
EP Kirk et al. NEJM 2024; 391: 1877-1889. Nationwide, Couple-Based Genetic Carrier Screening
One of the drawbacks with genetic screening has been identifying too many problems. Everyone has pathogenic mutations but many do not have effective treatments; in addition, many may be inconsequential if your partner does not share a similar genetic mutation (for autosomal recessive conditions). Furthermore, counseling everyone with genetic mutations is time-intensive. With this background, it is easy to see why a couple-based testing approach makes sense. In this study of an Australian genetic carrier screening program, couples had testing of at least 1281 genes with more than 750 serious diseases as part of the Mackenzie’s Mission project. Variants of uncertain significance were not reported.
Key findings:
- An estimated 45.9% of those invited to participate underwent reproductive genetic carrier screening
- 9107 couples completed screening, and 175 (1.9%) were newly identified as having an increased chance of having a child with a genetic condition for which we screened
- 180 (2.0%) were known, before participation in the study, to have an increased chance of having offspring with at least one genetic condition (due to family history or consanguinity)
- Of the 45 couples (25.7%) in which the female partner was pregnant when they received the result, 29 (64%) decided to have the fetus genetically tested, and 24 of these couples received normal test results. Of the 5 couples whose pregnancy was affected, 4 elected to end the pregnancy
- Of the 130 couples in which the female partner was not pregnant at the time of genetic testing, 95 (73.1%) indicated an intention to use in vitro fertilization with preimplantation genetic testing for monogenic conditions.
- Overall, 134 of 175 couples (76.6%) with a newly identified increased chance chose to alter their reproductive plans
Discussion Points:
- “92.7% of participants carried at least one pathogenic or likely pathogenic variant. With this prevalence and in this framework, reporting individual carrier results would be a burden on health systems. For example, one study showed a median time of 64 minutes to provide genetic counseling regarding results of reproductive genetic carrier screening.36“
- “Decisional regret was generally low,32 a finding that suggests that most participants valued the information provided by the screening.”
- 42% of couples had increased risks for diseases that would not have been identified using a previous list of 113 genes recommended by The American College of Medical Genetics and Genomics (ACMG).38 “As genomic testing technology advances, it is becoming possible to screen larger numbers of genes, and thus, any gene that meets criteria for inclusion, such as criteria that pertain to the severity of associated disease and to technical capability,1 can be screened.”
- “We limited reporting to variant combinations predicted to result in a serious childhood-onset condition or a condition in which early intervention can improve prognosis…Variants known to result in a mild condition or an adult-onset condition were not generally reported unless early intervention would improve prognosis. This approach aimed to minimize uncertain and ambiguous information and provide results that had high clinical usefulness.”
My take: Given the improvement in cost of genetic screening, this type of approach is quite practical and would not result in every family having to meet with a genetic counselor.
Related blog posts:
- Prenatal Testing, Statistics, and Life-Altering Decisions
- Noninvasive prenatal testing and why statistics matter