D Yogev et al. JPGN Reports; 2024 https://doi.org/10.1002/jpr3.12083. Open Access! Manometric findings in children with eosinophilic esophagitis and persistent post-remission dysphagia
In this 10-year retrospective review (2013-2023), the authors reviewed children with EoE referred for high-resolution impedance manometry (HRIM) due to persistent dysphagia despite histologic healing (i.e., <15 Eos/hpf).
Key findings:
- Among a cohort of ~1500 children, only 4 patients met inclusion criteria: histologic remission (<15 eos/hpf) and absence of fibrostenotic features on endoscopic evaluation
- Thus, the estimated prevalence of post-remission dysphagia in this cohort was exceedingly rare (<0.05%).
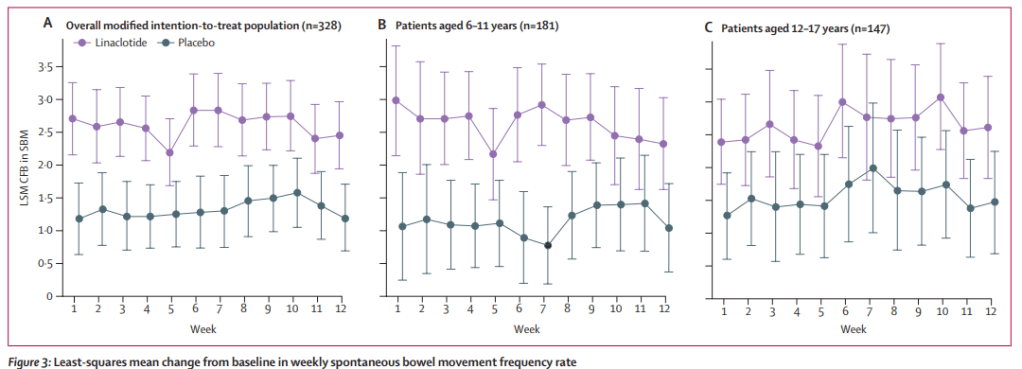
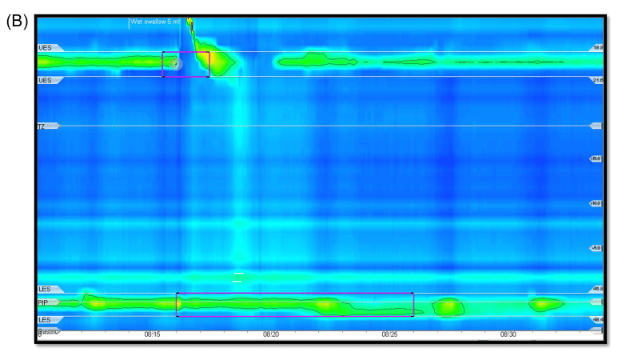
- On HRIM, all four patients had a hypomotile esophagus and abnormal bolus clearance. Lower esophageal sphincter integrated relaxation pressure values were normal in three patients and elevated in one. Two patients were diagnosed with ineffective esophageal motility, one with aperistalsis and one with achalasia type 1.
Discussion:
- “Endoscopic evaluation of fibrosis is somewhat limited as less than 50% of biopsies contain an adequate sample of the lamina propria for evaluation..[Also, these] patients did not undergo endoluminal functional lumen imaging (Endoflip) which has been recently shown to correlate with fibrotic changes of the esophagus in pediatric patients…Nonetheless, this case series highlights the fact that esophageal dysmotility can persist, even in the absence of endoscopic or histologic findings”
- The authors “did not study all EoE patients treated in our facility who had persistent dysphagia despite histologic remission of EoE, but rather explored just those who were referred for manometry. This methodology creates a risk of referral bias.”
My take: Though there is a referral bias due to the methodology, this study suggests that persistent dysphagia is rare in children who achieve EoE histologic remission. In addition, in those with significant dysphagia despite improvement in EoE, manometry is worthwhile.
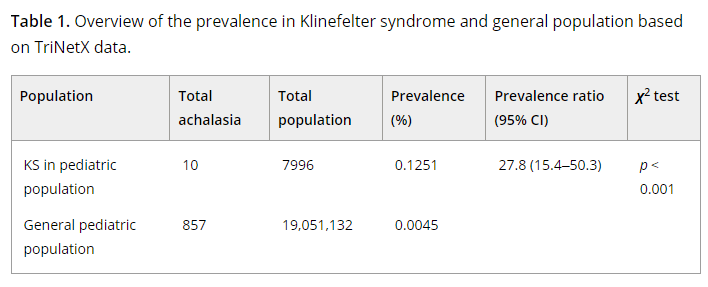
A related study suggests that achalasia may be more common in patients with Klinefelter syndrome, though still quite rare: L Miller et al. JPGN Reports 2024: https://doi.org/10.1002/jpr3.12084 Open Access! Achalasia in Klinefelter syndrome: A suspected pediatric case as well as prevalence analysis suggesting increased risk in this population
Related blog posts:
- Achalasia Frequent in Patients with Eosinophilic Esophagitis
- Practical Tips for Eosinophilic Esophagitis
- “Tug” Sign For Eosinophilic Esophagitis and EoE Bowel Sounds Tips
- Shorts: Hep E in Urine, Genetics in Autoimmune Enteropathy, EndoFlip Findings in EoE
- Dupixent Approved in Younger Children (15 kg+)
- Eosinophilic Esophagitis -Increasing Incidence and Emergence of Biologic Treatments
- NASPGHAN YouTube Video for Eosinophilic Esophagitis
- Increasing Burden of Eosinophilic Esophagitis
- Increasing Burden of Eosinophilic Esophagitis
- FLIP Patterns for Adults with Eosinophilic Esophagitis
- But How Well Does It Work in Theory and Eosinophilic Esophagitis Treatments
- Practical Guide to Dietary Therapy for Eosinophilic Esophagitis
- When to Use Dupilumab for Eosinophilic Esophagitis: Multispecialty Guidelines
- Managing Adrenal Insufficiency in Eosinophilic Esophagitis
- Landmark Dupilumab Study for Eosinophilic Esophagitis
- Achalasia -More Complexity to Pathophysiology?
- POEM vs Surgical Myotomy for Achalasia
- Achalasia -Updated Epidemiology | gutsandgrowth
- 4-14-4 Rule: More Biopsies Recommended for EoE