Here are some selected slides and notes from this year’s NASPGHAN’s postrgraduate course. There may be errors in omission or transcription on my part.
Link to the full NASPGHAN PG Syllabus 2019 (Borrowed with permission)
– Intestinal Inflammation Session
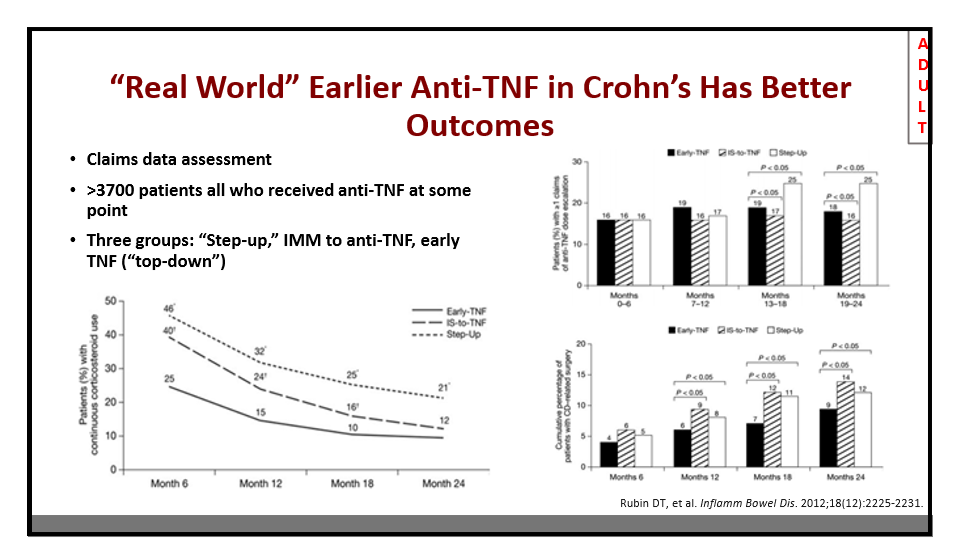
192 David T. Rubin, MD, University of Chicago Positioning the new IBD therapies: Merging experience with evidence
Some key points:
- Ustekinumab escalation can increase response. Optimization in CD patients with loss of response led to recapture of response in 69% of patients
- Tofacitinib –given black warning, will likely be used in more refractory patients
- May be able retry a previous therapy (Chicago protocol in slide below)
As an aside, while Dr. Rubin is an excellent speaker, my view is that there are so many terrific pediatric IBD specialists, I would favor having a pediatric IBD specialist give this talk at our postgraduate course. (Some might argue that adult IBD specialists would have more experience with emerging therapies.)

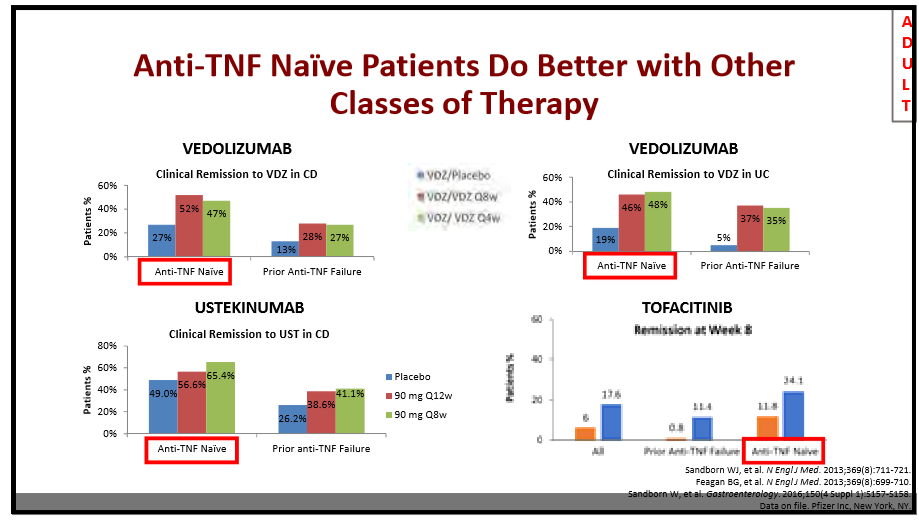
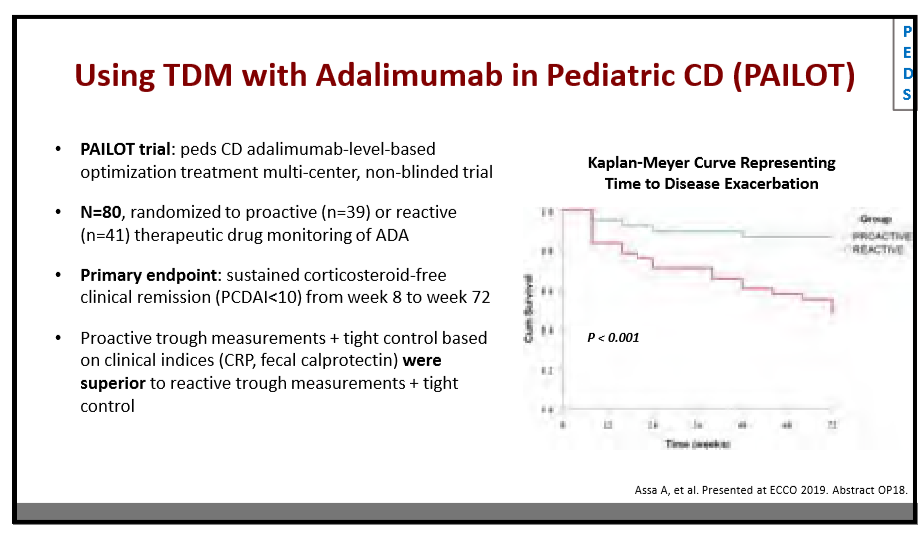
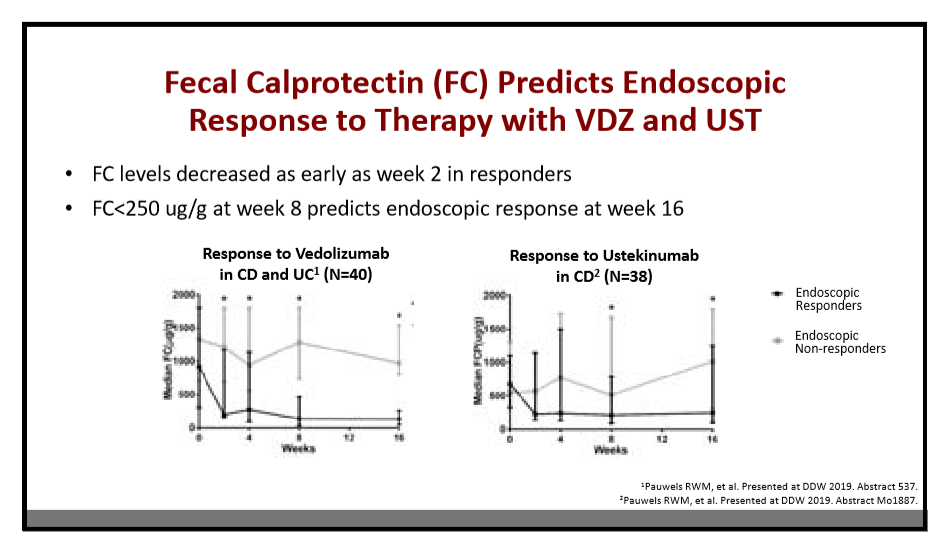


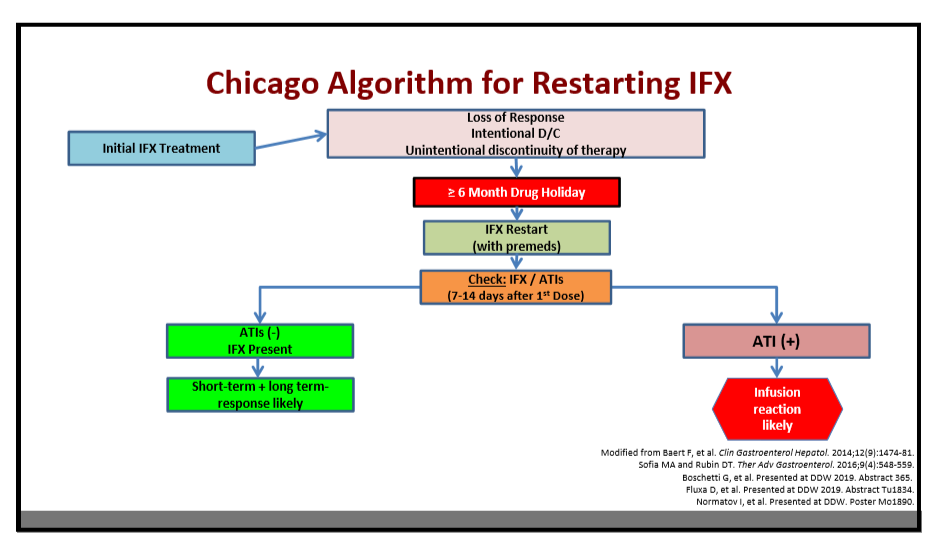

204 Anne Griffiths, MD, FRCPC, Hospital for Sick Children Immunosuppressive therapy in IBD: Can we de-escalate therapy?
- High rate of relapse when biologic therapy is stopped. Use of an immunomodulator may reduce the relapse rate when stopping an anti-TNF agent


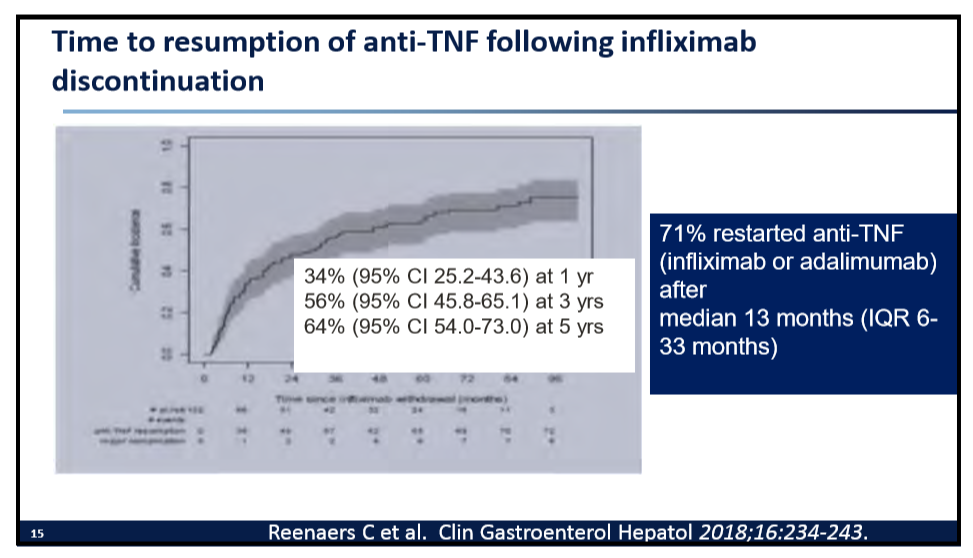

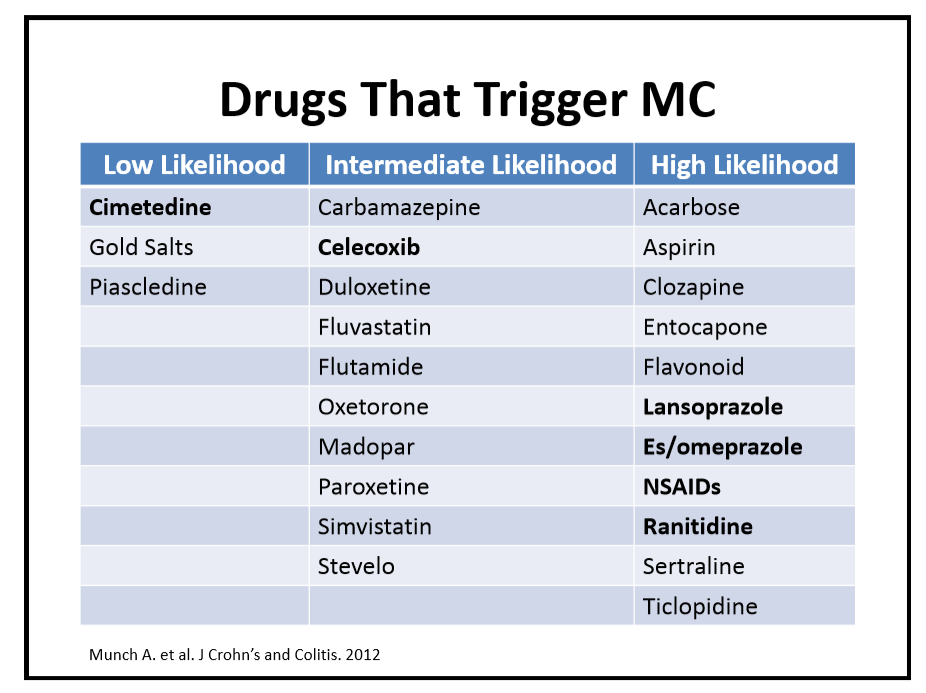
215 Stacy Kahn, MD, Boston Children’s Hospital When it is not IBD … rare forms of intestinal inflammation
- For patients with milder microscopic colitis, antidiarrheal agents can be given. For more severe disease, budesonide is effective.




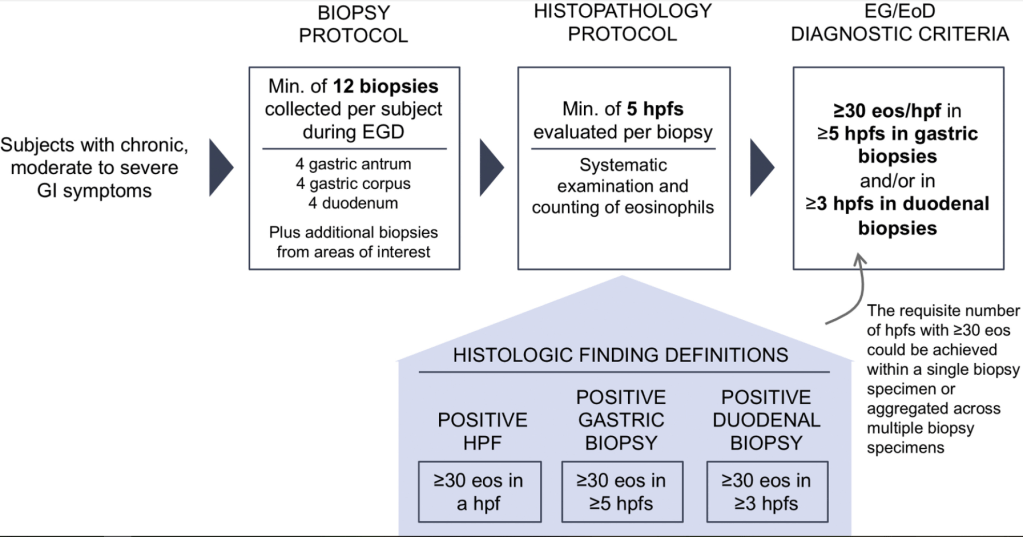
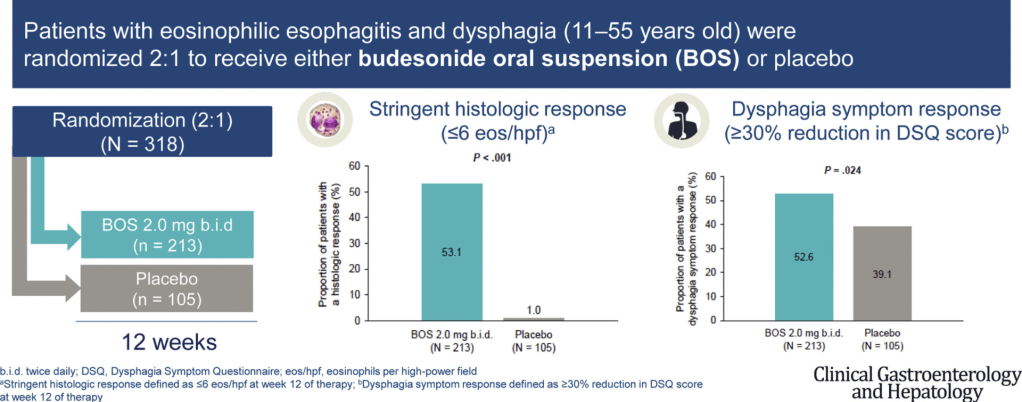
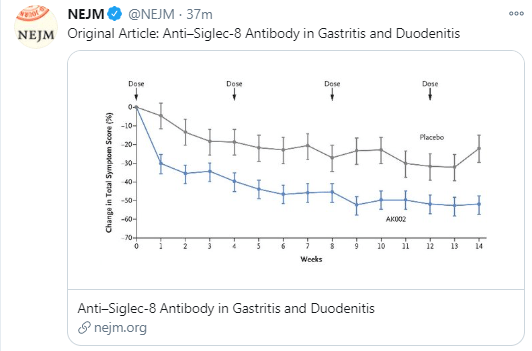
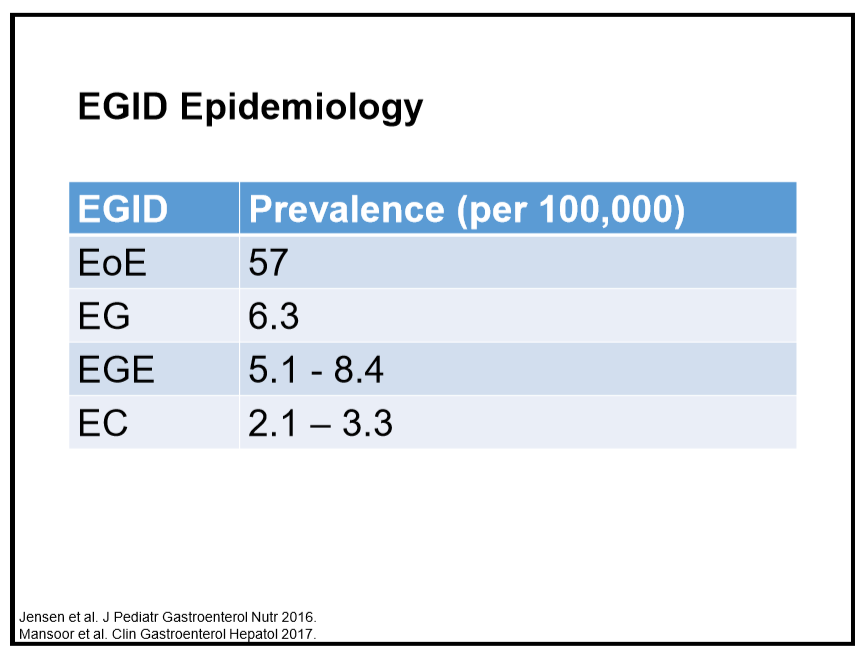
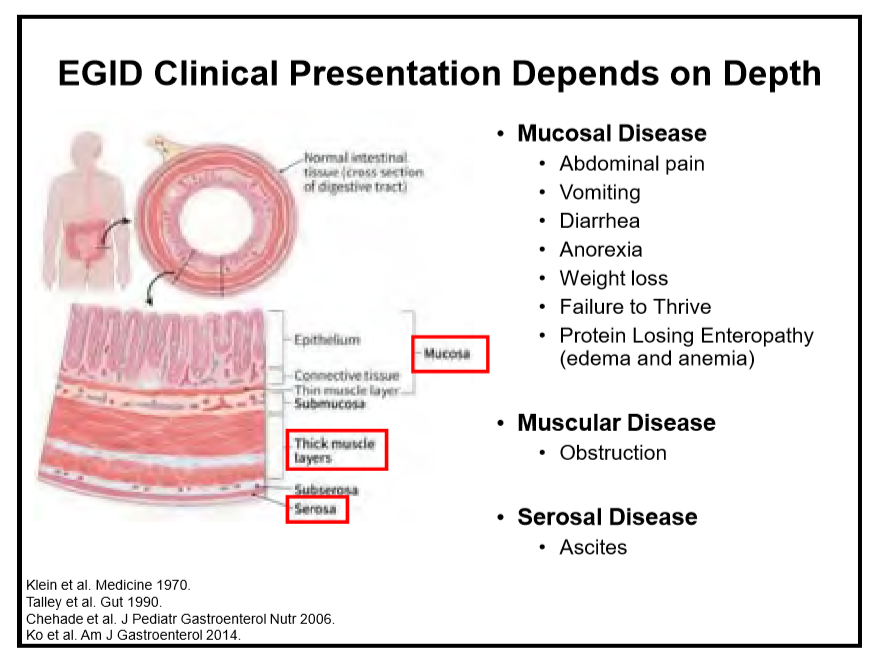
223 Edaire Cheng, MD, UT Southwestern Medical Center Eosinophilic inflammation beyond the esophagus

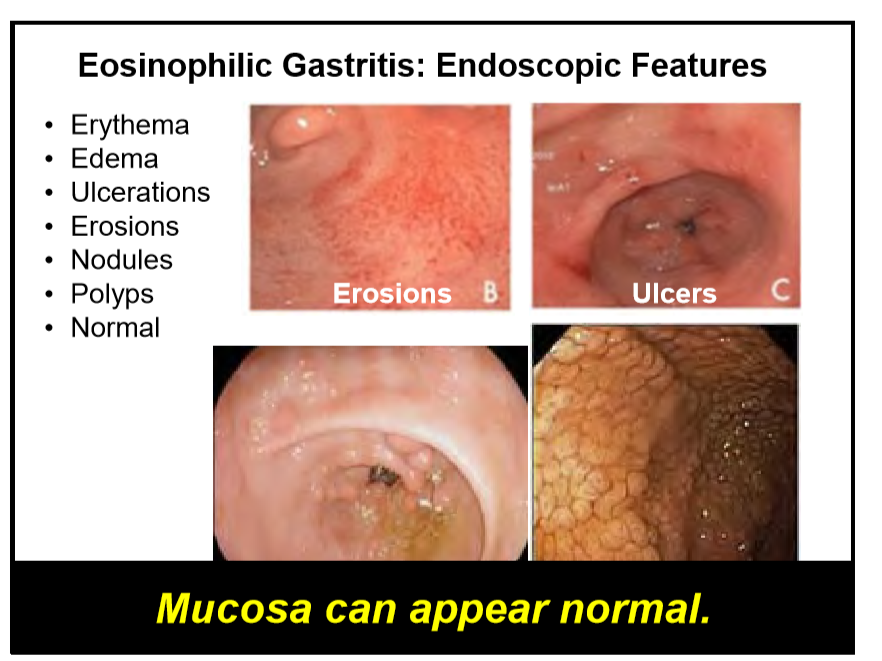
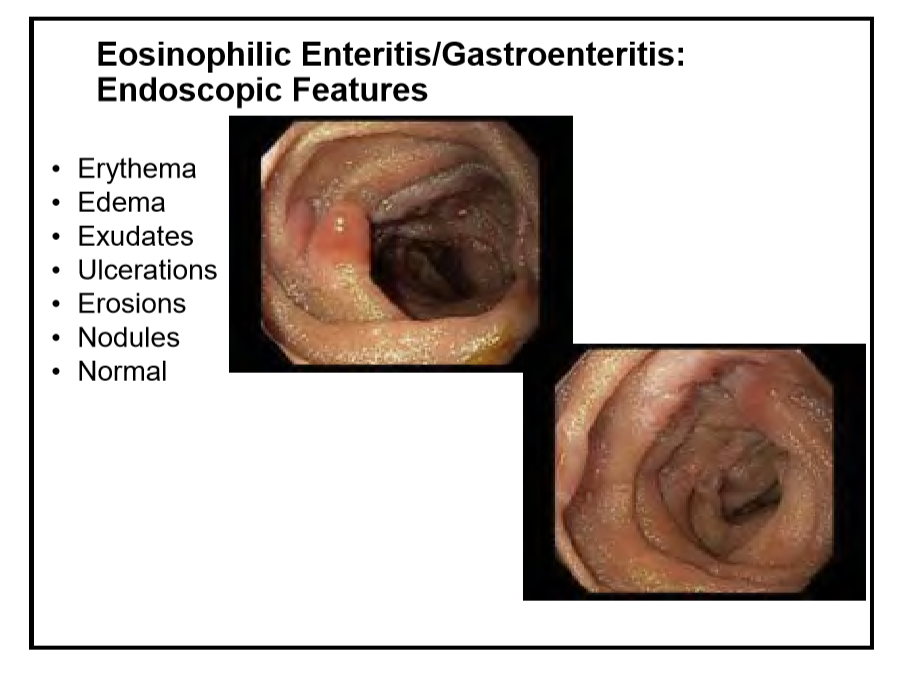
Disclaimer: NASPGHAN/gutsandgrowth assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. The discussion, views, and recommendations as to medical procedures, choice of drugs and drug dosages herein are the sole responsibility of the authors. Because of rapid advances in the medical sciences, the Society cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. Some of the slides reproduced in this syllabus contain animation in the power point version. This cannot be seen in the printed version.