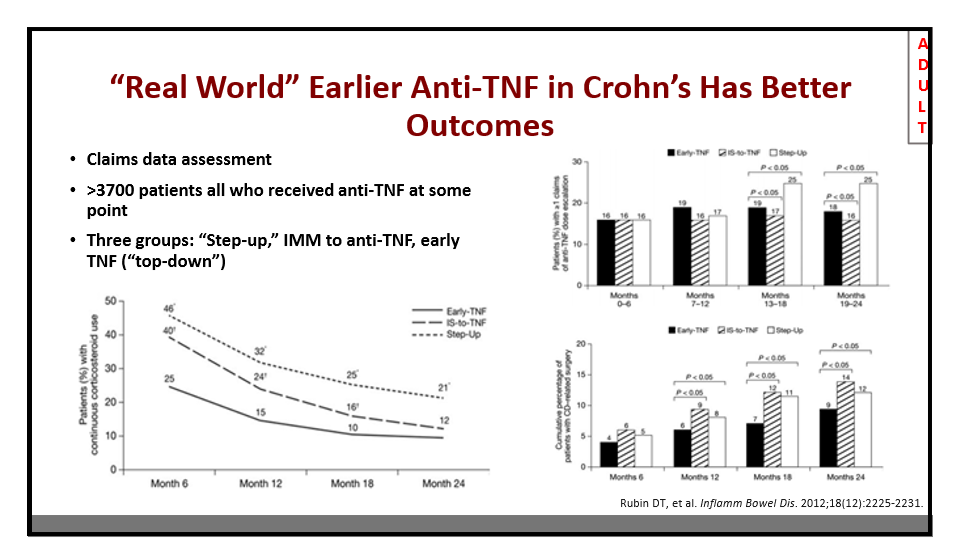
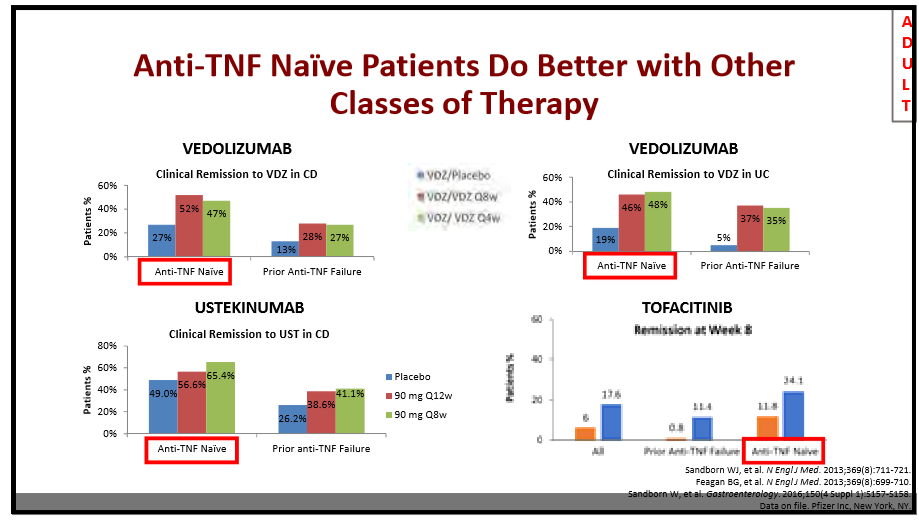
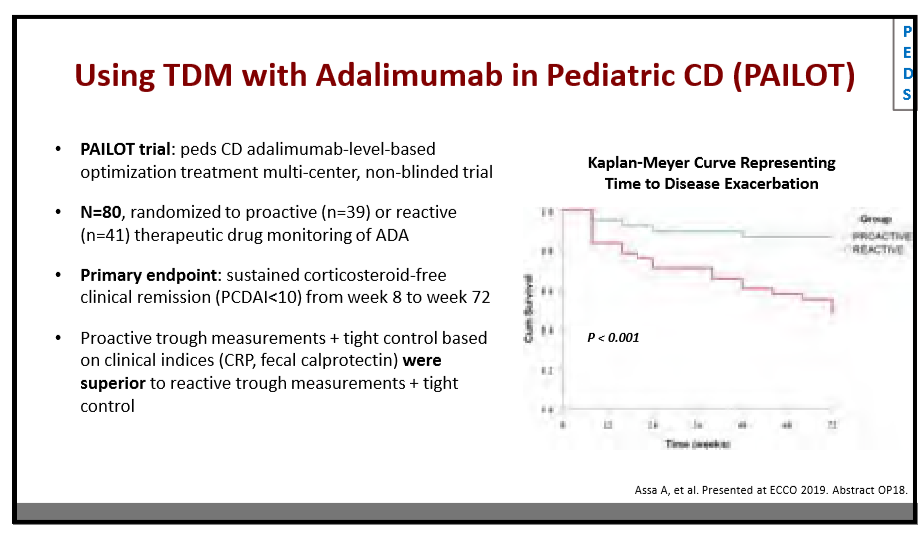
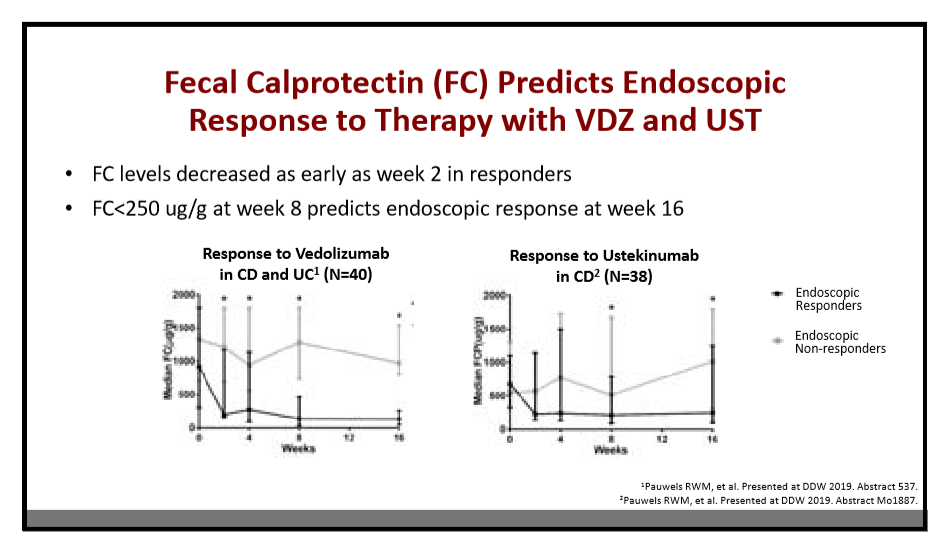
AH Everhov et al. Inflamm Bowel Dis 2022; 28: 1160-1168. Open Access! Probability of Stoma in Incident Patients With Crohn’s Disease in Sweden 2003-2019: A Population-based Study
In a nationwide Swedish cohort of 18,815 incident patients with a minimum 5 years of follow-up, 652 (3.5%) underwent formation of a stoma. The 5-year cumulative incidence of stoma formation was 2.5%, with no differences between calendar periods (2003–2006, 2007–2010, and 2011–2014).
RWM Pauweis et al. Clin Gastroentol Hepatol 2022; 20: 1671-1686. Open Access! Prediction of Relapse After Anti-Tumor Necrosis Factor Cessation in Crohn’s Disease: Individual Participant Data Meta-analysis of 1317 Patients From 14 Studies
C Ma. Clin Gastroentol Hepatol 2022; 20: 1668-1670. Associated editorial. Open Access! To Stop or Not to Stop? Predicting Relapse After Anti-TNF Cessation in Patients With Crohn’s Disease
This study captured data from 1317 patients (including 927 patients stopping infliximab and 390 patients stopping adalimumab) to develop risk prediction models. “The authors confirm many of the high risk, albeit rather intuitive, factors that are associated with the risk of relapse, including younger age, younger age at diagnosis, smoking, upper gastrointestinal tract involvement, longer disease duration, absence of concomitant immunosuppressant use, previous anti-TNF failure, and absence of clinical remission.”
The editorial notes that even in the lowest risk group, more than 20% had risk of relapse within 1 year; in addition, stopping therapy increases risk of not recapturing remission with restart of treatment. “Stopping anti-TNF therapy is a highly personalized treatment decision and is one that carries considerable risks…therapeutic discontinuation of TNF antagonists should be reserved for the very small minority of patients who are in deep remission, have a strong desire to stop treatment, have no (or very few) characteristics of high-risk CD, can tolerate a substantial disease flare, and are fully informed of the risks of therapeutic withdrawal.”
Related blog posts:
- What Happens When Infliximab is Stopped in Patients in Deep Remission Plus One
- “Do Not Stop Anti-TNF Medications in Children with IBD When They Are Working”
- Biologics in Children with Very Early Onset Inflammatory Bowel Disease
- What happens when anti-TNF therapy is stopped
WJ Sandborn et al. Clin Gastroenterol Hepatol 2022; 20: 1821-1830. Open Access! Efficacy and Safety of Extended Induction With Tofacitinib for the Treatment of Ulcerative Colitis
Graphical abstract below shows that 52.2% of patients who did not achieve clinical response to 8 weeks’ treatment with tofacitinib 10 mg BID in the induction studies achieved a clinical response following extended induction (delayed responders). At Month 12 of OCTAVE Open, 70.3%, 56.8%, and 44.6% of delayed responders maintained clinical response and achieved endoscopic improvement and remission, respectively. Corresponding values at Month 36 were 56.1%, 52.0%, and 44.6%.
My take: By extending the treatment induction to 16 weeks to determine response (rather than 8 weeks), the authors showed that 75% of patients with ulcerative colitis in the initial cohort respond to tofacitinib.
Related blog posts:
- Increased Risk, Increased Reward (possibly) with Tofacitinib
- IBD Shorts: Tofacitinib Safety, Vedolizumab for EIM
- AGA Guidelines: Moderate to Severe Ulcerative Colitis
- FDA Slaps Restrictions on JAK Inhibitors Over Serious Safety Risks (2021)
- A New FDA Warning for Tofacitinib (2021)
- FDA Warning on Tofacitinib (2019)
- “Tofacitinib: A Jak of All Trades”