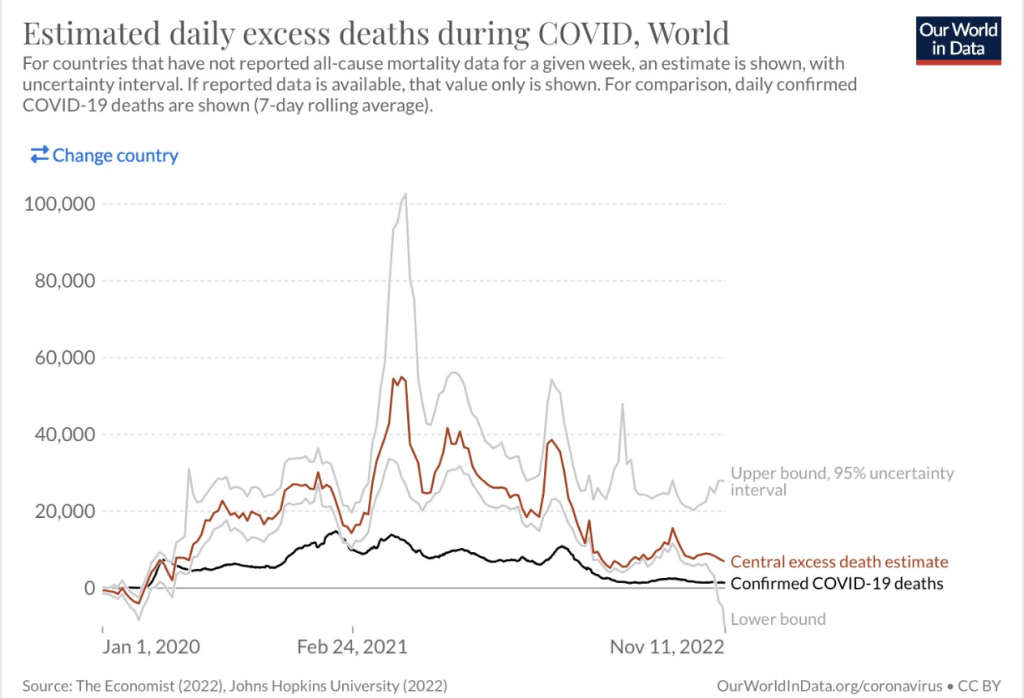
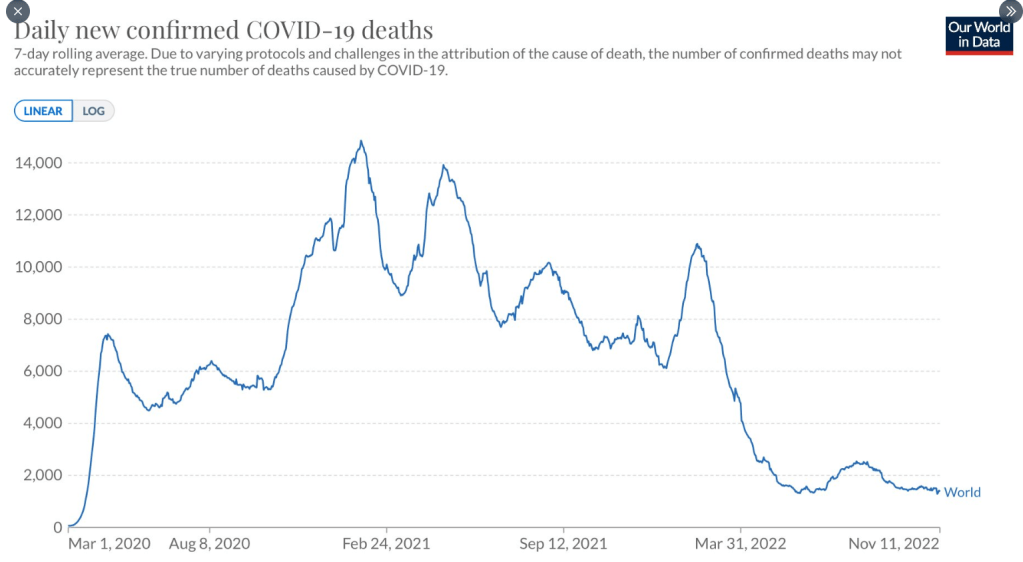
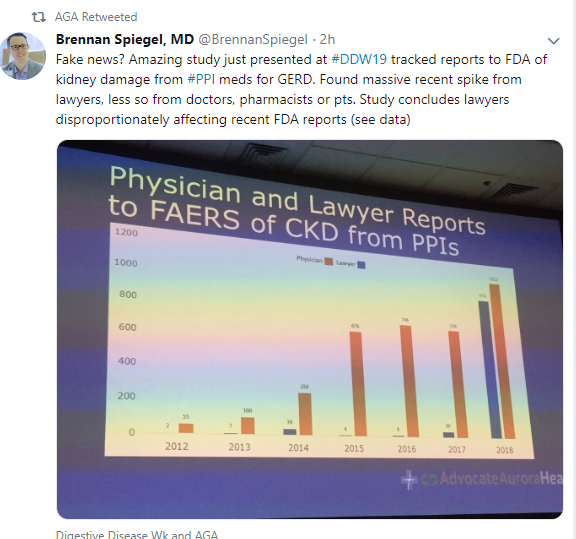
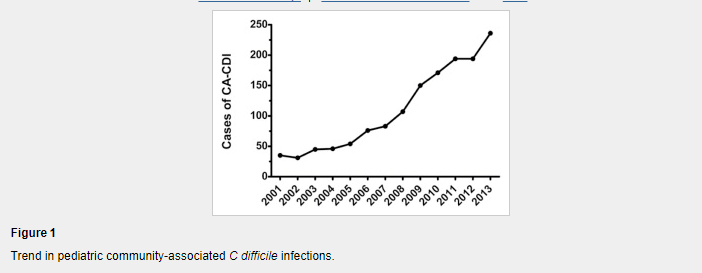
The most recent information and perspective on proton pump inhibitors and kidney disease:
From AGA: More Data on PPI Use and Kidney Disease
An excerpt:
The most recent study related to PPIs and CKD was a meta-analysis by Wijarnpreecha et al. presented at the American Society of Nephrology annual meeting and published in Digestive Diseases and Sciences. They found that any use of PPIs was associated with a 33 percent relative increase in risk for CKD/ESRD whereas no such risk was seen with H2RAs.
Talking to Your Patients
- Inform patients that, while this study does raise some concern about long-term PPI use and the potential contributions to kidney disease, the study does not show that PPI use causes kidney disease. No decisions should be made in haste as a reaction to this study. A brief explanation of the meta-analysis may also be helpful.
- Reassure patients that the benefits of using PPIs often outweigh the possible risks. Let them know that you prescribed a PPI for a clear-cut indication, in the lowest possible dose, and for an appropriate period of time (lowest dose, shortest time).
From the published abstract:
Results: five studies (three cohort studies and two case-control studies) with 536,902 participants met the eligibility criteria and were included in the meta-analysis. We found that individuals with PPIs use had significantly increased the risk of CKD or ESRD when compared with non-PPIs users (pooled RR of 1.33, 95% CI, 1.18-1.51). There was no publication bias of overall included studies assessed by the funnel plots.
My take: (borrowed from the AGA) This is an association, not proof of a causal relationship. Patients who use PPIs differ at baseline than those who do not. For example, patients who use PPIs are more likely to have diabetes or hypertension than patients who do not use PPIs, and are more likely to use additional nephrotoxic medications. Large retrospective studies are unable to completely adjust for these baseline differences. These differences, rather than PPIs themselves, may explain the observed association.
Related study: DCF Klatte et al. Gastroenterol 2017; 153: 702-10. In this retrospective analysis with more than 100,000 new PPI users (Swedish cohort), PPI users (compared to H2 blocker users) had an increased risk for doubled levels of creatinine with a HR of 1.26, and an increased risk of end-stage renal disease with HR of 2.40. The risk of chronic kidney disease was increased with higher cumulative PPI exposures.
Related study: Effects of PPI on dementia –recent large study shows no association: H Taipale et al. The American Journal of Gastroenterology(2017) 112, 1802–1808 (2017) doi:10.1038/ajg.2017.196. (Thanks to Ben Gold for this reference. This study examined more than 70,000 Finnish patients with Alzheimer’s disease (AD) (2005-2011) and 280,000 controls. Results: PPI use was not associated with risk of AD with 3-year lag window applied between exposure and outcome (adjusted odds ratio (OR) 1.03, 95% confidence interval (CI) 1.00–1.05). Similarly, longer duration of use was not associated with risk of AD (1–3 years of use, adjusted OR 1.01 (95% CI 0.97–1.06); ≥3 years of use adjusted OR 0.99 (95% CI 0.94–1.04)). Higher dose use was not associated with an increased risk (≥1.5 defined daily doses per day, adjusted OR 1.03 (95% CI 0.92–1.14)).

Sunrise over the South Rim at the Grand Canyon